5. Atomic Radiation Processes
advertisement

5. Atomic radiation processes
Einstein coefficients for absorption and emission
oscillator strength
line profiles: damping profile, collisional broadening, Doppler
broadening
continuous absorption and scattering
1
2
A. Line transitions
Einstein coefficients
probability that a photon in frequency interval
in the solid angle
range
is absorbed by an atom in the energy level El with a
resulting transition El Eu per second:
dwabs (ν, ω, l, u) = B lu I ν(ω) ϕ(ν )dν
atomic property
~ no. of incident
photons
probability for
transition l u
dω
4π
probability for
absorption of
photon with
probability for
with
absorption
profile
Blu: Einstein coefficient for absorption
3
Einstein coefficients
similarly for stimulated emission
dwst (ν, ω, l , u) = Bul Iν (ω) ϕ(ν)dν
dω
4π
Bul: Einstein coefficient for stimulated emission
and for spontaneous emission
dwsp (ν, ω, l, u) = A ul ϕ(ν)dν
dω
4π
Aul: Einstein coefficient for spontaneous emission
4
dwst (ν, ω, l , u) = Bul Iν (ω) ϕ(ν)dν
dω
4π
dwabs (ν, ω, l, u) = B lu I ν(ω) ϕ(ν )dν
dω
4π
Einstein coefficients, absorption and emission coefficients
Iν
dF
dV = dF ds
Number of absorptions
& stimulated emissions
in dV per second:
nl dw abs dV, nu dwst dV
ds
absorbed energy in dV per second:
stimulated emission counted
as negative absorption
dEνabs = nl hν dw abs dV − nu hν dwst dV
and also (using definition of intensity):
dEνabs = κL
ν Iν ds dω dν dF
κL
ν =
hν
ϕ(ν) [ nl B lu − nuBul ]
4π
for the spontaneously emitted energy:
Absorption and emission coefficients are a
function of Einstein coefficients, occupation
numbers and line broadening
²L
ν =
hν
n A ϕ(ν )
4π u ul
5
κL
ν
hν
=
ϕ(ν) [ nl B lu − nuBul ]
4π
²L
ν =
hν
n A ϕ(ν )
4π u ul
Relations between Einstein coefficients
Einstein coefficients are atomic properties do not depend on thermodynamic
state of matter
We can assume TE:
B ν (T ) =
²L
Sν = νL = Bν (T )
κν
Aul
nuAul
n
= u
nl B lu − nu B ul
nl Blu − nnul B ul
gu − hν
nu
=
e kT
nl
gl
From the Boltzmann formula:
for hν/kT << 1:
gu
2ν2
kT
=
c2
gl
for T ∞
nu
gu
=
nl
gl
µ
¶
hν
1−
,
kT
¶
µ
hν
1−
kT Blu −
0
gu
gl
A
¡ ul
1−
hν
kT
2ν2
Bν (T ) = 2 kT
c
¢
B ul
gl Blu = guB ul
6
gu
2ν2
kT =
2
c
gl
µ
¶
hν
1−
kT Blu −
gu
gl
A
¡ ul
1−
hν
kT
¢
gl Blu = guB ul
B ul
Relations between Einstein coefficients
2ν 2
gu
kT
=
c2
gl
for T ∞
2 hν3
A ul gu
=
c2
B lu gl
Aul
µ
hν
1−
kT
¶
2 h ν 3 gl
=
B
c2 gu lu
2 h ν3
A ul =
Bul
c2
hν
ϕ(ν ) B lu
κL
=
ν
4π
²L
ν =
¶
µ
hν A ul kT
1−
kT B lu hν
·
¸
gl
nl −
n
gu u
hν
gl 2hν3
ϕ(ν) nu
Blu
4π
gu c2
Note: Einstein coefficients atomic
quantities. That means any
relationship that holds in a special
thermodynamic situation (such as T
very large) must be generally valid.
only one Einstein
coefficient needed
7
Oscillator strength
Quantum mechanics
The Einstein coefficients can be calculated by quantum
mechanics + classical electrodynamics calculation.
Eigenvalue problem using using wave function:
Hatom |ψ l >= El |ψl >
H atom
p2
+ Vnucleus + Vshell
=
2m
Consider a time-dependent perturbation such as an external
electromagnetic field (light wave) E(t) = E0 eiωt.
The potential of the time dependent perturbation on the atom is:
V (t) = e
N
X
i=1
E · ri = E · d
d: dipol operator
[Hatom + V (t)] |ψl >= El |ψ l >
with transition probability
∼ | < ψ l |d|ψ u > |2
8
Oscillator strength
hν
πe 2
B =
f
4π lu
mec lu
The result is
flu: oscillator strength (dimensionless)
classical result from electrodynamics
= 0.02654 cm2/s
Classical electrodynamics
electron quasi-elastically bound to nucleus and oscillates within outer electric field as E.
Equation of motion (damped harmonic oscillator):
ẍ + γ ẋ + ω20 x =
damping
constant
γ=
2 ω20 e2
3 mec3
2 2
=
8π e
3 me
ν20
c3
resonant
(natural)
frequency
e
E
me
ma = damping force + restoring force + EM force
the electron oscillates preferentially at resonance
(incoming radiation ν = ν0)
The damping is caused, because the de- and
accelerated electron radiates
9
Classical cross section and oscillator strength
Calculating the power absorbed by the oscillator, the integrated “classical”
absorption coefficient and cross section, and the absorption line profile are
found:
Z
integrated over the line profile
ϕ(ν)dν =
2
πe
cl
= nl σtot
κL,cl
ν dν = nl
me c
γ/4π
1
π (ν − ν0 )2 + (γ/4π)2
nl: number
density of
absorbers
σtotcl: classical
cross section
(cm2/s)
[Lorentz (damping) line profile]
oscillator strength flu is quantum mechanical correction to classical result
(effective number of classical oscillators, ≈ 1 for strong resonance lines)
hν
L
ϕ(ν ) nl B lu
κ
=
From
(neglecting stimulated emission)
ν
4π
10
Oscillator strength
hν
πe 2
ϕ(ν) B lu =
ϕ(ν) flu = σlu (ν )
4π
mec
flu =
absorption cross section;
dimension is cm2
1 mec
hν B lu
4π πe 2
Oscillator strength (f-value) is different for each atomic transition
Values are determined empirically in the laboratory or by elaborate numerical atomic physics calculations
Semi-analytical calculations possible in simplest cases, e.g. hydrogen
flu =
25
g
3/2
5
3
π l u3
¡1
l2
−
¢
1 −3
u2
g: Gaunt factor
Hα: f=0.6407
Hβ: f=0.1193
Hγ: f=0.0447
11
Line profiles
line profiles contain information on physical conditions of the gas and
chemical abundances
analysis of line profiles requires knowledge of distribution of opacity with
frequency
several mechanisms lead to line broadening (no infinitely sharp lines)
- natural damping: finite lifetime of atomic levels
- collisional (pressure) broadening: impact vs quasi-static approximation
- Doppler broadening: convolution of velocity distribution with atomic profiles
12
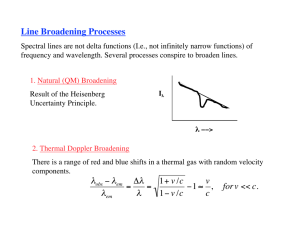
1. Natural damping profile
finite lifetime of atomic levels line width
NATURAL LINE BROADENING OR RADIATION DAMPING
t = 1 / Aul
Δ E t ≥ h/2π
(¼ 10-8 s in H atom 2 1): finite lifetime with respect to
spontaneous emission
uncertainty principle
Δν1/2 = Γ / 2π
Δλ1/2 = Δν1/2λ2/c
line broadening
e.g. Ly α: Δλ1/2 = 1.2 10-4 A
Γ/4π
1
ϕ(ν) =
π (ν − ν0 )2 + (Γ/4π)2
Lorentzian profile
Hα: Δλ1/2 = 4.6 10-4 A
13
Natural damping profile
resonance line
excited line
natural line broadening is important for strong lines (resonance
lines) at low densities (no additional broadening mechanisms)
e.g. Ly α in interstellar medium
but also in stellar atmospheres
14
2. Collisional broadening
radiating atoms are perturbed by the electromagnetic field of neighbour atoms,
ions, electrons, molecules
energy levels are temporarily modified through the Stark - effect: perturbation
is a function of separation absorber-perturber
energy levels affected line shifts, asymmetries & broadening
ΔE(t) = h Δν = C/rn(t)
r: distance to perturbing atom
a) impact approximation: radiating atoms are perturbed by passing particles at distance r(t).
Duration of collision << lifetime in level lifetime shortened line broader
in all cases a Lorentzian profile is obtained (but with larger total Γ than only natural damping)
b) quasi-static approximation: applied when duration of collisions >> life time in level
consider stationary distribution of perturbers
15
Collisional broadening
n=2
linear Stark effect ΔE ~ F
for levels with degenerate angular momentum (e.g. HI, HeII)
field strength F ~ 1/r2
ΔE ~ 1/r2
important for H I lines, in particular in hot stars (high number density of free
electrons and ions). However , for ion collisional broadening the quasi-static
broadening is also important for strong lines (see below) Γe ~ ne
n=3
resonance broadening
atom A perturbed by atom A’ of same species
important in cool stars, e.g. Balmer lines in the Sun
ΔE ~ 1/r3
Γe ~ ne
16
Collisional broadening
n=4
quadratic Stark effect ΔE ~ F2
field strength F ~ 1/r2
ΔE ~ 1/r4
(no dipole moment)
important for metal ions broadened by e- in hot stars Lorentz profile with Γe ~ ne
n=6
van der Waals broadening
atom A perturbed by atom B
important in cool stars, e.g. Na perturbed by H in the Sun
Γe ~ ne
17
Quasi-static approximation
tperturbation >> τ = 1/Aul
perturbation practically constant during emission or absorption
atom radiates in a statistically fluctuating field produced by ‘quasi-static’ perturbers,
e.g. slow-moving ions
given a distribution of perturbers field at location of absorbing or emitting atom
statistical frequency of particle distribution
probability of fields of different strength (each producing an energy shift ΔE = h Δν)
field strength distribution function
line broadening
Linear Stark effect of H lines can be approximated to 0st order in this way
18
Quasi-static approximation for hydrogen line broadening
Line broadening profile function determined by probability function for electric field
caused by all other particles as seen by the radiating atom.
W(F) dF: probability that field seen by radiating atom is F
dF
ϕ(∆ν ) dν = W (F )
dν
dν
For calculating W(F)dF we use as a first step the nearest-neighbor approximation:
main effect from nearest particle
19
Quasi-static approximation –
nearest neighbor approximation
assumption: main effect from nearest particle (F ~ 1/r2)
we need to calculate the probability that nearest neighbor is in the distance
range (r,r+dr) = probability that none is at distance < r and one is in (r,r+dr)
Integral equation for W(r)
differentiating
differential equation
probability for no particle in (0,r)
relative probability for particle in shell
(r,r+dr) N: total uniform particle density
Differential equation
Normalized solution
20
Linear Stark effect:
Quasi-static approximation
mean interparticle
distance:
normal field strength:
define:
F
r0 2
β=
={ }
F0
r
note: at high particle density large F0
stronger broadening
from W(r) dr W(β) dβ:
Stark broadened line profile in the wings,
not Δλ-2 as for natural or impact broadening
21
Quasi-static approximation – advanced theory
complete treatment of an ensemble of particles: Holtsmark theory
+ interaction among perturbers (Debye shielding of the potential at distances >
Debye length)
Holtsmark (1919),
Chandrasekhar (1943, Phys. Rev. 15, 1)
2β
W (β) =
π
Z
∞
−y 3/2
e
y sin(βy)dy
0
Ecker (1957, Zeitschrift f. Physik, 148, 593 & 149,245)
4/3
2βδ
W (β) =
π
number of particles
inside Debye sphere
Mihalas, 78
2
g(y) = y 3/2
3
Z
Z
y
T 1/2
D = 4.8
cm
ne
4π 3
δ=
D N
3
∞
e−δg(y) y sin(δ 2/3 βy)dy
0
∞
(1 − z −1 sinz)z −5/2 dz
Debye length, field of ion vanishes
beyond D
number of particles
inside Debye sphere
22
3. Doppler broadening
radiating atoms have thermal velocity
Maxwellian distribution:
³ m ´3/2
m
2
2
2
P (vx , vy , vz ) dvx dvy dvz =
e − 2 kT (vx+ vy +vz ) dvx dvy dvz
2πkT
Doppler effect: atom with velocity v emitting at frequency
frequency :
v cos θ
ν0 = ν − ν
c
, observed at
23
Doppler broadening
Define the velocity component along the line of sight:
The Maxwellian distribution for this component is:
P (ξ) dξ =
1
π 1/2ξ 0
e
¡ ξ ¢2
−
ξ0
dξ
ξ0 = (2kT /m)1/2
ν0 = ν − ν
if v/c <<1
ξ
c
thermal velocity
if we observe at v, an atom with velocity
component ξ absorbs at ν′ in its frame
line center
∆ν = ν0 − ν = −ν
ξ
ξ
ξ
= −[(ν − ν0) + ν0 ] ≈ −ν0
c
c
c
24
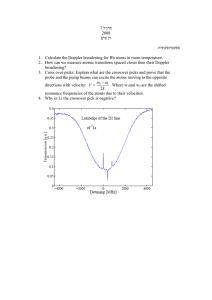
Doppler broadening
line profile for v = 0
ϕ R (ν − ν0 )
=⇒
profile for v ≠ 0
ξ
ϕR (ν0 − ν0) = ϕ(ν − ν0 − ν0 )
c
New line profile: convolution
ϕ new (ν − ν0) =
Z∞
−∞
ξ
ϕ(ν − ν0 − ν0 ) P (ξ) dξ
c
profile function in rest
frame
velocity distribution
function
25
Doppler broadening: sharp line approximation
ϕ new (ν − ν0) =
1
π1/2
Z∞
−∞
ξ ξ
ϕ(ν − ν0 − ν0 0 ) e−
c ξ0
¡ ξ ¢2
ξ0
dξ
ξ0
: Doppler width of the line
ξ0 = (2kT /m)1/2
thermal velocity
Approximation 1: assume a sharp line – half width of profile function <<
ϕ(ν − ν0 ) ≈ δ(ν − ν0 )
delta function
26
Doppler broadening: sharp line approximation
new
ϕ
(ν − ν0 ) =
1
π1/2
Z∞
−∞
δ(ν − ν0 − ∆νD
¡ ¢
ξ − ξξ0 2 dξ
)e
ξ0
ξ0
δ(a x) =
ϕnew(ν − ν0) =
1
π1/2
1
δ(x)
a
¶ ¡ ¢2
Z∞ µ
ξ
ν − ν0
dξ
ξ
e − ξ0
δ
−
∆νD
ξ0
∆νD ξ0
−∞
¡ ν−ν 0 ¢ 2
1
−
ϕ new (ν − ν0) = 1/ 2
e ∆ν D
π
∆νD
Gaussian profile –
valid in the line core
27
Doppler broadening: Voigt function
Approximation 2: assume a Lorentzian profile – half width of profile function >
Γ/4π
1
ϕ(ν) =
π (ν − ν0 )2 + (Γ/4π)2
1
a
ϕnew (ν − ν0 ) = 1/2
π
∆νD π
Z∞
−∞
2
³
∆ν
∆νD
e −y
dy
´2
− y + a2
a=
Γ
4π∆νD
¶
µ
1
ν
−
ν
0
ϕ new(ν − ν0 ) = 1/2
H a,
π
∆νD
∆νD
Voigt function (Lorentzian * Gaussian):
calculated numerically
28
Voigt function: core Gaussian, wings Lorentzian
normalization:
Z∞
H(a, v) dv =
√
π
−∞
usually α <<1
max at v=0:
H(α,v=0) ≈ 1-α
~ Gaussian
~ Lorentzian
Unsoeld, 68
29
Doppler broadening: Voigt function
Approximate representation of Voigt function:
³
H a,
ν −ν0
∆νD
´
≈e
≈
¡ ν−ν 0 ¢2
−
π
∆ν D
a
¡
¢2
1 /2 ν−ν 0
∆ν D
line core: Doppler broadening
line wings: damping profile
only visible for strong lines
General case: for any intrinsic profile function (Lorentz, or Holtsmark, etc.) – the
observed profile is obtained from numerical convolution with the different
broadening functions and finally with Doppler broadening
30
General case: two broadening mechanisms
two broadening functions representing
two broadening mechanisms
Resulting broadening function is
convolution of the two individual
broadening functions
31
4. Microturbulence broadening
In addition to thermal velocity: Macroscopic turbulent motion of stellar
atmosphere gas within optically thin volume elements. This is
approximated by an additional Maxwellian velocity distribution.
Additional Gaussian broadening function
in absorption coefficient
32
blueshift
5. Rotational broadening
If the star rotates, some
redshift
surface elements move
towards the observer and
some away
blueshift
redshift
Gray, 1992
to
observer
Rotational velocity at
equator: vrot = Ω•R
stripes of constant wavelength shift
Observer sees vrot sini
Intensity shifts in wavelength along stripe
33
Rotation and observed line profile
stellar spectroscopy uses mostly normalized spectra Fλ / Fcontinuum
integral over stellar disk
towards observer, also
integral over all solid
angles for one surface
element
observed stellar
line profile
34
Spring 2013
Fλ /Fcontinuum
M33 A supergiant
Keck (ESI)
U, Urbaneja,
Kudritzki, 2009,
ApJ 740, 1120
35
Rotation changes integral over stellar surface
stellar spectroscopy uses mostly normalized spectra Fλ / Fcontinuum
Gray, 1992
integral over stellar disk
towards observer
observed stellar
line profile
Correct treatment by numerical integral over
stellar surface with intensity calculated
by model atmosphere
36
Approximation: assume Pλ const. over surface
integral of Doppler shifted profile over
stellar disk and weighted by
continuum intensity towards obs.
continuum limb
darkening
37
Approximation: assume Pλ const. over surface
Rotational
broadening
function
Rotationally
broadened line
profile
convolution of original
profile with rotational
broadening function
38
Rotational broadening
Note: Rotational line broadening is not caused
by physical processes affecting the absorption
coefficient.
It is not the result of radiative transfer.
It is caused by the macroscopic motion of the
stellar surface elements and the Doppler-effect.
39
Rotational broadening profile function
G(x)
Unsoeld, 1968
40
Note: rotation does not
change equivalent width!!!
Rotationally broadened line profiles
v sini
km/s
Gray, 1992
41
observed stellar line profiles
v sini large
v sini small
Gray, 1992
42
Note: rotation does not
change equivalent width!!!
Rotationally broadened line profiles
v sini
km/s
Gray, 1992
43
observed stellar line profiles
θ Car, B0V
vsini~250 km/s
Schoenberner, Kudritzki et al. 1988
44
6. Macro-turbulence
The macroscopic motion of optically thick surface volume
elements is approximated by a Maxwellian velocity
distribution. This broadens the emergent line profiles in
addition to rotation
45
Rotation and macro-turbulence
rotation
rotation +
macro-turb.
5Å
Gray, 1992
46
B. Continuous transitions
1. Bound-free and free-free processes
absorption
hν + El Eu
photoionization - recombination
emission
hνlk
spontaneous
Eu El + hν
stimulated
hν + Eu El +2hν
(isotropic)
(non-isotropic)
bound-bound: spectral lines
bound-free
free-free
consider photon hν ≥ hνlk
κνcont
(energy > threshold): extra-energy to free electron
e.g. Hydrogen
R
1
mv2 = hν − hc 2
2
n
R = Rydberg constant = 1.0968 105 cm-1
47
b-f and f-f processes
Hydrogen
l continuum
Wavelength (A)
Edge
1 continuum
912
Lyman
2 continuum
3646
Balmer
3 continuum
8204
Paschen
4 continuum
14584
Brackett
5 continuum
22790
Pfund
48
b-f and f-f processes
κb−f
= nl σlk (ν )
ν
- for a single transition
- for all transitions:
f
κb−
ν
=
X
elements,
ions
for hydrogenic ions
for H:
X
nl σlk (ν)
l
σ lk (ν ) = σ0(n)
Gaunt factor ≈ 1
³ ν ´3
l
ν
gbf (ν )
Kramers 1923
Gray, 92
σ0 = 7.9 10-18 n cm2
νl = 3.29 1015 / n2 Hz
absorption per particle
49
b-f and f-f processes
Gray, 92
late A
σb-f
n (ν)
29
= 2.815 × 10
Z4
g (ν )
n5ν 3 bf
peaks increase with n:
hνn = E∞ − En = 13.6/n2 eV
=⇒ ν
−3
6
→n
late B
for non-H-like atoms no ν-3
dependence
peaks at resonant frequencies
free-free absorption
much smaller
50
b-f and f-f processes
Hydrogen dominant continuous absorber in B, A & F stars (later stars H-)
Energy distribution strongly modulated at the edges:
Balmer
Vega
Paschen
Brackett
51
b-f and f-f processes : Einstein-Milne relations
Generalize Einstein relations to bound-free processes relating photoionizations and
radiative recombinations
line transitions
σlu(ν) =
hν
ϕ(ν ) Blu
4π
gl Blu = guB ul
2 h ν3
A ul =
Bul
c2
stimulated b-f emission
b-f transitions
κb−f
ν
=
X
elements,
ions
X
i
"
σik(ν ) ni −
nenIon
1
gi
2g1
µ
h2
2πmkT
n∗i
¶3/2
i
e EIon /k T e−hν /kT
#
LTE occupation number
52
2. Scattering
In scattering events photons are not destroyed, but redirected and
perhaps shifted in frequency. In free-free process photon interacts with
electron in the presence of ion’s potential. For scattering there is no
influence of ion’s presence.
in general: κsc = ne σe
Calculation of cross sections for scattering by free electrons:
- very high energy (several MeV’s): Klein-Nishina formula (Q.E.D.)
- high energy photons (electrons): Compton (inverse Compton) scattering
- low energy (< 12.4 kEV ' 1 Angstrom): Thomson scattering
53
Thomson scattering
THOMSON SCATTERING: important source of opacity in hot OB stars
8π 2
8π e 4
−25
2
σe =
=
6.65
×
10
cm
r0 =
3
3 m2e c4
independent of frequency, isotropic
Approximations:
- angle averaging done, in reality: σe σe (1+cos2 θ) for single scattering
- neglected velocity distribution and Doppler shift (frequency-dependency)
54
Simple example: hot star -pure hydrogen atmosphere
total opacity
Total opacity
κν =
N
N
X
X
i=1 j= i+1
+
N
X
i=1
µ
¶
g
i
σline
(ν
)
n
−
nj
i
ij
gj
³
σik(ν ) ni − n∗i e −hν/k T
´
³
´
−hν /kT
+nenpσ k k(ν, T ) 1 − e
+ne σe
line absorption
bound-free
free-free
Thomson scattering
55
Simple example: hot star -pure hydrogen atmosphere
total emissivity
Total emissivity
N
N
2hν 3 X X line
gi
²ν = 2
σij (ν ) nj
c
gj
line emission
i=1 j =i+1
N
2hν 3 X ∗
+ 2
ni σ ik (ν)e −hν/k T
c
bound-free
i=1
2hν3
+ 2 nenpσk k(ν, T )e− hν/kT
c
free-free
+neσ e Jν
Thomson scattering
56
Rayleigh scattering – important in cool stars
RAYLEIGH SCATTERING: line absorption/emission of atoms and
molecules far from resonance frequency: ν << ν0
from classical expression of cross section for oscillators:
σ(ω) = fij σkl (ω) = fij
ω4
σe
(ω 2 − ω2ij )2 + ω 2γ 2
ω4
for ω << ωij
σ(ω) ≈ fij σe 4
ωij + ω 2γ 2
ω4
for γ << ωij
σ(ω) ≈ fij σ e 4
ωij
σ(ω) ∼ ω 4 ∼ λ−4
important in cool G-K stars
for strong lines (e.g. Lyman
series when H is neutral) the
λ-4 decrease in the far wings
can be important in the
optical
57
What are the dominant elements for the continuum opacity?
- hot stars (B,A,F) : H, He I, He II
- cool stars (G,K): the bound state of the H- ion (1 proton + 2 electrons)
only way to explain solar continuum (Wildt 1939)
The H- ion - important in cool stars
ionization potential = 0.754 eV
λion = 16550 Angstroms
H- b-f peaks around 8500 A
H- f-f ~ λ3 (IR important)
He- b-f negligible,
He- f-f can be important
in cool stars in IR
requires metals to provide
source of electrons
dominant source of
b-f opacity in the Sun
Gray, 92
58
Additional absorbers
Hydrogen molecules in cool stars
H2 molecules more numerous than atomic H in M stars
H2+ absorption in UV
H2- f-f absorption in IR
Helium molecules
He- f-f absorption for cool stars
Metal atoms in cool and hot stars (lines and b-f)
C,N,O, Si, Al, Mg, Fe, Ti, ….
Molecules in cool stars
TiO, CO, H2O, FeH, CH4, NH3,…
59
Examples of continuous absorption coefficients
Unsoeld, 68
Teff = 5040 K
B0: Teff = 28,000 K
60
Modern model atmospheres include
● millions of spectral lines (atoms and ions)
● all bf- and ff-transitions of hydrogen
helium and metals
● contributions of all important negative ions
● molecular opacities (lines and continua)
61
Concepcion 2007
complex atomic models for O-stars (Pauldrach et al., 2001)
62
NLTE Atomic Models in modern model atmosphere codes
lines, collisions, ionization, recombination
Essential for occupation numbers, line blocking, line force
Accurate atomic models have been included
26
elements
149 ionization stages
5,000 levels ( + 100,000 )
20,000diel. rec. transitions
4 106 b-b line transitions
Auger-ionization
recently improved models are based on Superstructure
Eisner et al., 1974, CPC 8,270
AWAP 05/19/05
63
64
Concepcion 2007
Recent Improvements on Atomic Data
• requires solution of Schrödinger equation
for N-electron system
• efficient technique:
R-matrix method in CC approximation
• Opacity Project
Seaton et al. 1994, MNRAS, 266, 805
• IRON Project
Hummer et al. 1993, A&A, 279, 298
accurate radiative/collisional data
to 10% on the mean
65
Confrontation with Reality
Photoionization
Nahar 2003, ASP Conf. Proc.Ser. 288, in press
Electron Collision
Williams 1999, Rep. Prog. Phys., 62, 1431
high-precision atomic data
66
Improved Modelling for Astrophysics
e.g. photoionization cross-sections for carbon model atom
67
Przybilla, Butler & Kudritzki 2001b, A&A, 379, 936
Red supergiants, NLTE model atom for FeI
Bergemann, Kudritzki et al., 2012
68
Red supergiants, NLTE model atom for TiI
Bergemann, Kudritzki et al., 2012
69
TiII
Bergemann,
Kudritzki et al., 2012
TiI
70
USM 2011
TiO in red supergiants
MARCS model atmospheres, Gustafsson et al., 2009
71
USM 2011
TiO in red supergiants
A small change in
carbon
abundances…
MARCS model
atmospheres
72
CO molecule in red supergiants
The Scutum RSG clusters
Davies, Origilia, Kudritzki et al., 2009
73
Rayner, Cushing, Vacca, 2009: molecules in Brown Dwarfs
74
Exploring the substellar temperature regime down to ~550K
Burningham et al. (2009)
Jupiter
T9.0 ~ 550K
T8.5 ~ 700K
75