FINDING THE VOLTAGE OF DIFFERENT TYPES OF POTATO
advertisement

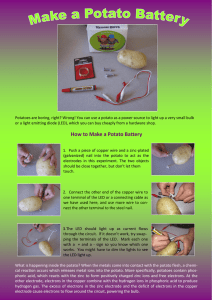
FINDING THE VOLTAGE OF DIFFERENT TYPES OF POTATO BATTERIES Samantha Dietrich Cary Academy ABSTRACT The purpose of the experiment was to find which form of potato, being used as a battery produces the most energy and lights an LED the best. It was hypothesized that the generic Idaho potato would produce the most energy and light an LED the best as it was the kind of potato most often used as a battery. A section of copper wire was stuck into the potato, then a zinc nail next to it. The metals were then attached to an LED via alligator-clip wires. To measure the voltage, the battery was hooked up to a digital multimeter. The voltage was found and recorded. This was done to all of the potatoes. The blue potato produced the most voltage at .946 V. The Idaho potato battery produced .819 V. The sweet potato battery produced .88 V. The baked potato battery produced .888 V. The French fry battery produced the least energy at .81 V. INTRODUCTION The purpose of this experiment was to find which form of potato, being used as a battery, produces the most energy and lights an LED the best. Typically, these are made by using a potato, and sticking a zinc plated nail into it, and sticking a copper wire segment next to it. Alligator clip wires are clipped to each of the metals. When the two loose ends of the wire are attached to a small light bulb or LED, it supposedly lights up. Potato batteries are electrochemical cells, in which chemical energy is converted into electrical energy through a spontaneous electron transfer (this occurs when an electron moves from an atom/ molecule into another atom/molecule. The potato separates the zinc (nail) and copper (penny) ions. If the zinc and copper had been touching in the potato, they only would have generated heat. With the potato between them, the electron transfer must occur in the copper wire, channeling the energy into the light bulb/LED. Electrochemical reactions most often involve an electron transfer. The passage of an electric current causes or accompanies them. In most cases, it involves an electron transfer between a solid and a liquid. Mostly, chemical reactions involve heat or light, but not another form of energy. There are some chemical reactions that produce electricity rather than heat, or light. The potato battery is a cell, and is made up of the electrodes, which electrons enter and exit from (the nail and the penny) and an electrolyte, which allow electrons to flow through them (the chemicals in the potato). Chemical reactions between these are what change the chemical energy to electric energy. The zinc plated nail becomes the negative side of the battery and the penny becomes the positive. Caused by a chemical reaction between the metal and the potato, electrons gather at the negative side of the battery, but are lost at the positive side. This causes some electrons from the negative side of the battery to move towards the positive side of the battery. The current creates enough energy to light up a small light bulb or LED. The potential difference between two places in a circuit or electric current is voltage. It can also be thought of as the amount of energy released when a charge moves from one place to another in a current. The greater the voltage, the more energy is released. If there is voltage between two places on a current, then electrons will flow through the current. A greater voltage makes a greater current. The greater the current, the more electrons move through the wire each second. The resistance of an object affects the amount of electrons that can move through the wire. Conductors, such as copper or iron, have a small enough resistance so that electrons can move through them. However, copper has a much lower resistance than iron, so more electrons can flow through it at a time. Insulators, on the other hand, have so much resistance that electrons cannot pass through them at all, Wood, cloth, and paper are all insulators. In the potato battery, the copper penny, the wire, the nail, and even the chemicals in the potato are conductors. If there was an insulator in between a wire and a penny in the battery, the light bulb would not light, because the electrons would not be able to get through the material to power the light bulb. The sweet potato was a sort of reddish, orangeish, brown color, and pointy on both ends. It was rough and scratched. It was 14.5 cm, and had a mass of 312 g. The normal Idaho potato was brown, long, and had round ends. It felt rough, hard and had dips in the skin. It was 16.5 cm and had a mass of 448.1 g. The blue potato was purplish brown, round, rough and hard. It had a few blemishes in its skin. It was 6 cm, and had a mass of 89 g. The baked potato was dark brown, long and round on both ends. It was rough, and soft. The dips in the skin were the same as the ones the normal Idaho potato had. It smelled like French fries, and was 13.5 cm. The French fries were light brown, long rectangular prisms. They were soft and soggy. It was hypothesized that the normal Idaho potato would produce the most energy as a battery and light an LED the best, because it is the most often used to make a potato batteries. The sweet potato and the blue potato may have less of the chemical in them that produces electricity when it reacts with the copper and zinc. The baked potato and the French fries were cooked, and liquid turns to steam when heated up, so some of the chemical may have left them when they were cooked. MATERIALS AND METHOD Copper wire segment Zinc plated nail Baked potato French fry Sweet potato Normal Idaho potato Blue potato Red LED Multimeter Alligator clip wires First the zinc plated nail was stuck into the potato, and then the copper wire segment was stuck next to it. Alligator clip wires were clipped to each the zinc plated nail and the copper wire segment. The loose ends of the wires were clipped to the red LED to see if it lit. Then the Alligator clip wires were taken off of the nail and the copper wire. A digital multimeter was used to measure the amount of voltage the potato battery produced. This was repeated for each of the potato variations. The control of the experiment was the Idaho potatoes. The independent variable was the kind of potato used. The dependent variable was the amount of volts produced. Controlled variables included type of wire, type of electrolyte, and type of LED used. RESULTS AND DISCUSSION Amount of Volts Produced (V) 1 0.95 0.9 0.85 0.8 0.75 0.7 Idaho Blue Sweet Baked French Fry Type of Potato The Idaho potato battery produced .819 V. The blue potato battery produced .946 V. The sweet potato battery produced .88 V. The baked potato produced .888 V. The French fry produced .810 V. The maximum amount of volts produced in the experiment was the blue potato at .946 V. The minimum amount of volts produced was the French fries at .810. CONCLUSION The hypothesis was that the normal Idaho potato would produce the most energy and light an LED the best, but it was not confirmed. The Idaho potato battery produced only .819 V. The blue potato produced much more, .946 V, the most energy produced from any of the other potato batteries. None of the potato batteries could actually light an LED anyways, so it was an incorrect hypothesis. The reason this might have happened is that the way that the potato battery produces electrical energy is by way of a chemical reaction between the electrolytes and the chemicals in the potato. Since the potato doesn’t have much liquid in it, the chemical may not be present as much as in something like a lemon, which is also often used as a battery. The blue potato seemed to have more liquid in it than the Idaho potato, so it would produce more energy. The experiment could be improved by using even more types of potatoes, such as mashed potatoes, to see if it makes a difference. Future experiments that could be performed are using citrus fruits instead of potatoes, as such fruits as lemons, limes, and oranges have more liquid, and therefore more chemical reaction, then the potatoes. The experiment could also be done on different fruit juices to find if that could also work. REFERENCES California Science Center. "Lemon light." California Science Center. California Science Center. 2013. Web. January 28, 2014. Champagne, Andrew. Electricity and Magnetism. Austin: Holt, Rinehart, and Winston, 2007. Print. Kidsworld. "How Potato Batteries Work." Kidzworld. 2014. Web. January 27, 2014. Robert Lamb. "Lemon Batteries and Potato power." How Stuff Works. How Stuff Works Inc.2014. Web. January 27, 2014.