230
Brief Communications
Age-Induced Decreases in the Messenger RNA
Coding for the Sarcoplasmic Reticulum
Ca2'-ATPase of the Rat Heart
L.M.Z. Maciel, R. Polikar, D. Rohrer, B.K. Popovich, and W.H. Dillmann
Downloaded from http://circres.ahajournals.org/ by guest on September 30, 2016
Age-associated slowing of cardiac relaxation related to the decline in the Ca2' pump function
of cardiac sarcoplasmic reticulum (SR) has been previously described. It is unclear if the
decreased Ca2' pump function results from a lower amount of Ca2+-ATPase protein or a
decreased pumping activity of the enzyme. To determine if these alterations could be mediated
by changes in the amount of the protein itself, the level of the messenger RNA (mRNA) coding
for the Ca2'-ATPase of the SR of Fischer rat hearts (4- and 30-month-old rats) were
quantitated with a Northern blotting technique. We observed that the levels of SR Ca2+-ATPase
mRNA were 60% lower in old rats as compared with young rats, suggesting that a quantitative
reduction in the levels of the corresponding protein could occur during aging to explain the
delayed diastolic relaxation documented in old animals as opposed to a change in the specific
activity of this enzyme. The thyroid hormone responsiveness of SR Ca2'-ATPase mRNA has
been previously established. We have found in this study that the thyroxine levels were
consistently lower in old rats; however, this difference was relatively small (4.3 ±0.7 and 3.1±0.8
gg/dl [mean± SD], respectively, in young and old rats). In addition, no age-induced decrease
in 3,5,3'-triiodothyronine levels was observed, suggesting that the aging process itself may
be responsible for the changes in SR Ca2'-ATPase mRNA levels. (Circulation Research
1990;67:230-234)
P rolonged duration of contraction is one of the
most prominent and well-documented effects
of aging on the mechanics of cardiac muscle
in humans' as well as in animals.2-5 Previous studies
have suggested that this prolonged contraction is due
to an age-associated slowing of cardiac relaxation,
because age does not seem to change the speed of
tension development by heart muscle during isometric contraction.2,6
In the heart, contraction and relaxation are dependent on the Ca21 concentration in the cytoplasm. Ca21
concentrations are changed by translocation across
the sarcoplasmic reticulum (SR) and sarcolemmal
membrane systems, and any modification in this process related to aging could affect changes in myocardial contraction and relaxation. This possibility is
supported by reports7,8 showing that the rate of Ca21
uptake is reduced in isolated SR vesicles prepared
from senescent rat hearts. These in vitro results reflect
From the Department of Medicine, University of California,
San Diego, San Diego, Calif.
Supported by a fellowship from the Conselho Nacional de
Desenvolvimento Cientifico e Tecnol6gico (CNPq) and National
Institutes of Health grant HL-25022.
Address for reprints: W.H. Dillmann, MD, UCSD Medical
Center, 225 Dickinson Street (H-811-C), San Diego, CA 92103.
Received March 7, 1989; accepted February 6, 1990.
a decreased Ca2' pump function of the SR, that is, a
decreased activity of the SR Ca2'-dependent ATPase
enzyme. It is currently unclear whether the decreased
Ca2' pump function results from a lower amount of
Ca2'-ATPase protein or a decreased pumping activity of an unchanged number of pump units. Because
the complementary DNA (cDNA) coding for Ca2'dependent ATPase of the SR has recently been
cloned,9 we used Northern blot analysis to investigate
the influence of aging on the level of SR Ca2'ATPase messenger RNA (mRNA).
Materials and Methods
Animals
Two groups of male Fischer 344 rats purchased
from the National Institute of Aging, Bethesda, Md.,
were used in this study. One group was composed of
nine animals aged 4 months and the other of nine
rats aged 30 months.
Studies of the same rat strain have shown that in
these animals adult physiological competence as
measured by maturity of the nervous and endocrine
functions is reached around 6 months of age in males.
Aging as measured by coat deterioration, incidence
of tumors, and increased mortality begins at 22
months of age. The average life span is approximately
Maciel et al Age and Cardiac Sarcoplasmic Reticulum Ca2'-ATPase
28 months, with the maximum being 33 months.l0
The two ages chosen in this study are, therefore,
representative of young adult and old rats. All animals were free of grossly visible disease and received
a standard block diet and water ad libitum.
Animals were anesthetized with sodium pentobarbital, blood was collected from the tail, and after
clotting and centrifugation the sera were stored at
-20° C for further analysis. After blood collection,
the chest was opened, and the heart was removed,
rinsed in ice-cold 0.9% sodium chloride, and immediately used for RNA preparation.
Downloaded from http://circres.ahajournals.org/ by guest on September 30, 2016
RNA Preparation
Total RNA was prepared by the proteinase Kphenol procedure previously described.'1 Briefly,
each ventricle was homogenized in a Waring blender
(New Hartford, Conn.) in the presence of proteinase
K. The homogenate was then extracted once with
equal volumes of phenol and chloroform. After this,
the RNA was precipitated with ethanol and suosequently with 3 M sodium acetate; a final ethanol
precipitate yielded total RNA with a ratio of absorbance at 260 nm to absorbance at 280 nm of 1.8:2.
The RNA was translationally active. Equal RNA
recovery was obtained from the heart of old and
young rats yielding 1 mg RNA/g heart wet wt.
Northem-Blot Analysis
Each animal's RNA was processed separately. A
total of 15 ,gg RNA was denatured in the presence of
2.2 M formaldehyde and 50% deionized formamide
at 650 C for 10 minutes and was separated through
electrophoresis in 0.8% agarose gels made of 20 mM
MOPS (pH 7.4), 5 mM sodium acetate, 1 mM
EDTA, and 2.2 mM formaldehyde. The positions of
28S and 18S mRNAs were marked by staining with
acridine orange in 10 mM sodium phosphate buffer
(pH 6.5) and 1.1 M formaldehyde. The ratio of 28S to
18S RNA was about 2:1. The RNA was transferred
to a Nytran membrane (Schleicher & Schuell, Inc.,
Keene, N.H.) in the presence of 10x standard saline
citrate (SSC) (1 x SSC is 300 mM sodium chloride, 30
mM sodium citrate, pH 7.0).
The membrane was air dried for 20 minutes,
baked at 800 C in a vacuum oven, and prehybridized
at 420 C for 10-12 hours in a solution containing 5 x
SSPE (1 x SSPE is 150 mM sodium chloride, 0.01 M
sodium dihydrogen phosphate, 0.001 M EDTA),
20 x Denhardt's solution (1 x Denhardt's solution is
0.02% Ficoll, 0.02% polyvinylpyrrolidone, and
deionized bovine serum albumin), 25 ,g/ml
sheared herring sperm DNA, 0.1% sodium dodecyl
sulfate (SDS), and deionized formamide at a final
concentration of 50%.
Labeling Probes
Ca`+-ATPase cDNA was labeled by using a nick
translation kit (Bethesda Research Laboratories,
Gaithersburg, Md.) according to the manufacturer's
instructions. The specific activities obtained were
231
about 1x 108 cpm/,g DNA. [`P]dNTPs (3,000 Ci/
mmol) were purchased from Amersham, Arlington
Heights, Ill. The Ca'+-ATPase cDNA is a 2.3kilobase pair rat cDNA clone corresponding to the 3'
end of slow SR Ca2'-ATPase. The rat Ca2'-ATPase
cDNA clone was obtained by screening a rat heart
cDNA library cloned into the AZAP vector (Stratagene Inc., La Jolla, Calif.) with a cDNA clone of the
3' end of the rabbit skeletal muscle SR Ca2+-ATPase
kindly provided by D. MacLennan, Toronto,
Canada.9 The rat heart SR Ca2+-ATPase cDNA
clone was verified by nucleotide sequencing.12
An unidentified cDNA clone (C5) of 1,000 base
pairs cloned into the vector pT7/T3-18 (Bethesda
Research) was isolated in this laboratory by D.
Rohrer and used as a reference for equivalent RNA
loading. This plasmid was also labeled by nick translation to a specific activity of 1x108 cpm/,ug. All
probes were purified on a Bio-Gel A-Sm column
(Bio-Rad, Richmond, Calif.).
After hybridization, the filter was washed twice
with 2x SSC and 0.1% SDS at room temperature for
20 minutes, twice with lx SSC and 0.1% SDS at
550 C for 30 minutes, once with 0.2x SSC and 0.1%
SDS at 55° C for 30 minutes, or 0.1 x SSC and 0.1%
SDS at 500 C for 30 minutes for Ca2-ATPase or C5.
A myosin light chain (MLC2) cDNA probe, kindly
provided by Dr. K. Chien,13 was simultaneously
hybridized with the Ca2-ATPase probe on an additional Northern blot made with RNA from five
individual young and five individual old rat hearts.
Autoradiograms were obtained after exposure to
Kodak XAR films (Rochester, N.Y.) at -70° C
baked by intensifying screens.
Relative mRNA ratios were determined by scanning densitometry of the autoradiograms and corrected for loading differences by using C5 and MLC2,
respectively, When probed simultaneously as a standard. It was assured that a linear relation existed
between dpm film darkening and scanning signal.
Radioimmunoassay Measurement of Serum Total
Thyroxine and 3,5,3'-Thiiodothyronine
Total thyroxine (T4) and 3,5,3'-triiodothyronine (T3)
levels in unextracted serum were measured by using a
commercial radioimmunoassay kit (Monobind, Costa
Mesa, Calif.). Hormone measurements for a given
experiment were all performed in the same assay.
Statistics
The results are expressed as mean+SEM and
were compared between the two groups by Student's t test to unpaired data. The level of significance was set at p<0.05.
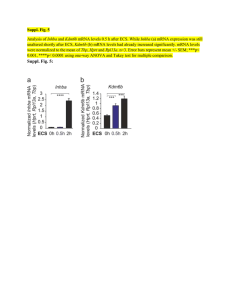
Results
The steady-state levels of total cardiac Ca2-ATPase
mRNA of 30- and 4-month-old Fischer 344 rats are
compared in Figure 1A. An equal amount of total
RNA obtained from young and old rat hearts was
loaded per lane and checked by acridine orange
232
Circulation Research Vol 67, No 1, July 1990
A
B
100
80
60
28 s
2
40ti
young
X tt 41 8 S
Downloaded from http://circres.ahajournals.org/ by guest on September 30, 2016
FIGURE 1. Northern-blot analysis of total RNA from old (O)
and young (Y) rat heart ventricle (15 gg) probed with slow
sarcoplasmic reticulum Ca2'-A TPase complementary DNA
(cDNA) (panel A). The same Northem blot was probed with
reference CS cDNA (panel B). 0, 30-month-old rat; Y
4-month-old rat.
staining. The autoradiogram shows that the mRNA
level of SR Ca'+-ATPase mRNA is significantly
lower in the aged as compared with the young rat
heart (Figure lA).
The same Northern blot was probed with C5
cDNA (Figure iB). No difference was observed in
mRNA levels between the two groups by the C5
cDNA probe. The level of C5 mRNA was used to
correct for slight differences in RNA loading.
The quantification of total Ca'+-ATPase mRNA
levels by densitometric scanning of autoradiograms
shows a 60% decrease in the mRNA coding for SR
Ca'+-ATPase from old rats (Figure 2). These data
indicate that the level of the mRNA coding for slow
SR Ca2-ATPase significantly decreases with age
(p=0.0002). Furthermore, a second Northern blot
containing RNA from five individual young and five
individual old rats was probed simultaneously with
SR Ca'z-ATPase and MLC2 cDNA probes. The ratio
of SR Ca2-ATPase mRNA over MLC2 mRNA in
young versus old rats revealed a 4.7-fold age-induced
decrease. These data are shown in Table 1.
Serum T4 concentrations in aged male rats were
significantly lower than those observed in young rats
(Table 2). However, no appreciable changes in serum
levels of T3 were documented between the two
groups. In addition, a significantly positive correlation between the levels of mRNA for Ca"2-ATPase
evaluated by the relative density and the serum levels
of T4 was observed for all animals grouped together
(r=0.69, p<0.001). However, the decrease of T4
levels in old animals is relatively small (4.3+0.7
old
RNA TYPE
FIGURE 2. Total RNA from 18 individual rat hearts was
quantitated on Northern blots by using the complementary
DNA probe described above, and the autoradiographic signals
for Ca2'-A TPase messenger RNA (mRNA) were corrected for
loading differences with CS mRNA as a nonchanging standard.
The autoradiograms were quantified by densitometric scanning.
Ca2'-ATPase mRNA levels in young rats were normalized to
100%. Values in old rats are expressed as the percentage of
young mRNA levels and represent mean±SEM. Young, 4month-old rats (n=9); old, 30-month-old rats (n=9).
versus 3.1±0.8 gg/dl). No correlation of SR Ca2'ATPase mRNA levels to T3 levels was observed.
Discussion
Aging could be considered as a process of gradual
disorganization of complex physiological systems
responsible for maintaining homeostasis and it may
result, at least in part, from the impairment of key
cellular functions. The results of this study demonstrate that the mRNA coding for the cardiac SR
Ca2'-ATPase decreases markedly and selectively
with age when compared with other mRNAs, including the mRNA coding for MLC2. Our findings provide additional support for previous observations that
showed a decrease in the activity of Ca2'-ATPase of
the SR and the resulting reduction in the rate of
calcium uptake by the myocardial SR of old
animals.7,8 These changes could constitute the biochemical basis for the slower rate of relaxation
observed in the heart muscle with aging.
The age-dependent reduction in the levels of this
specific mRNA should result in lower levels of the
corresponding protein. However, there are no studies
published so far that have quantified the Ca2'-ATPase
protein itself. Our findings are thus consistent with
decreased calcium pump function caused by
decreased synthesis of Ca2'-ATPase rather than a
change in the specific activity of this enzyme.
Several other recent studies have also described
the effect of age on the expression of specific mRNA
species.14 Most of the studies have shown decreasing
values of different specific mRNA with aging.14 However, one of the mRNA species that has been studied
in detail is albumin. Recently, Horbach et a115
showed that the cell-free synthesis of albumin by rat
liver increased 35% between 12 and 24 months of
age, and the levels of albumin mRNA increased
approximately 80% between these two ages.
Maciel et al Age and Cardiac Sarcoplasmic Reticulum Ca2'-ATPase
233
TABLE 1. Quantification of Sarcoplasmic Reticulum Ca2'-ATPase and Myosin Light Chain Messenger RNA Levels by
Densitometric Scanning
Old rats
Densitometric
Animal
1
3
Young rats
Densitometric
reading
Animal
reading
5.800
2
0.785
3.329
0.692
4
3.893
5
6
0.891
7
5.872
8
0.366
9
2.178
10
1.748
0.896±0.23*
4.214±0.717
Mean±SEM
The signals and autoradiograms of Northern blots were quantitated by densitometric scanning. The values from these
readings are indicated. SR Ca2+-ATPase, sarcoplasmic reticulum calcium ATPase; MLC2, myosin light chain; SR
Ca2+-ATPase/MLC2, ratio of these two messenger RNAs.
SR Ca2+-ATPase/MLC2
*p<0.01.
Downloaded from http://circres.ahajournals.org/ by guest on September 30, 2016
In addition, the present study confirmed earlier
observations of a reduction of serum T4 levels with
aging although circulating T3 concentrations remain
relatively unchanged.16-'9 This observation is part of
a large amount of data supporting the concept that
major changes in thyroid function occur with aging in
rats, including a reduced thyroid uptake of radiolabeled iodide, a probable reduction in the thyroid
release of the hormone as well as changes in the
release of thyroid-stimulating hormone from the
pituitary, and a reduction in tissue responsiveness to
administered thyroid hormone.20
The marked influence that thyroid status has on
cardiac function is well established, especially as
related to the activity of the SR by changing the rate
of calcium transport across SR membranes as well as
the calcium-dependent ATP hydrolysis,21 and also, as
recently reported, by modifying the mRNA levels of
the slow Ca2-ATPase of the SR.22 In this study, the
marked influence of thyroid hormone on mRNA for
Ca2-ATPase was demonstrated by a rapid increase
in the levels of mRNA for this enzyme to T3 administration in hypothyroid rats.
In our investigation, a significant correlation
between the serum levels of T4 and the corresponding
relative levels of mRNA was observed when all animals were analyzed together. Even though our findings
are consistent with previous observations, suggesting a
functional relation between thyroid hormones and
Ca2'-ATPase activity, we should point out that the
magnitude of the difference of T4 levels between young
and old rats was relatively small and also that no
correlation was observed between mRNA and the
levels of T3, which is the most biologically active thyroid
TABLE 2. Effect of Aging on Serum Thyroxine and 3,5,3'-
Trilodothyronine Levels in Male Fischer 344 Rats
Age (months)
30 (n=9)
4 (n=9)
3.1±0.8
4.3±0.7*
T4 (,ug/dl)
67.3±10.6
68.6±5.6
T3 (ng/dl)
All values are mean± SD. n, Number of animals; T4, thyroxine;
T3, 3,5,3'-triiodothyronine.
Hormone
hormone. Although partial desensitization to circulating thyroid hormones with aging cannot be ruled out,
our data suggest, but do not prove, that the aging
process itself is the primary cause for the decrease in
the mRNA for Ca'+-ATPase. However, a contribution
of age-induced changes in the thyroid status and alterations in the SR Ca'+-ATPase mRNA levels by a
thyroid hormone-mediated mechanism cannot be
excluded.
In summary, our results indicate that the mRNA
for slow Ca'+-ATPase decreases with age in rats
and may lead to a decrease in the amount of the
Ca'+-ATPase protein itself. Further studies will be
necessary to determine whether the observed
changes in mRNA levels are dependent exclusively
on the aging process or if the thyroid status contributes to this alteration to some extent.
References
1. Harrison TR, Dixon K, Russell RO Jr, Bidwai BS, Coleman
HN: The relation of age to the duration of contraction,
ejection, and relaxation of the normal human heart. Am Heart
J 1964;67:189-199
2. Lakatta EG, Gerstenblith G, Angel CS, Shock NW, Weisfeldt
ML: Prolonged contraction in aged myocardium. J Clin Invest
1975;55:61-68
3. Rumberger E, Timmerman T: Age-changes of the forcefrequency relationship and duration of action potential in
isolated papillary muscles of guinea pig. Eur J Appl Physiol
Occup Physiol 1976;34:227-234
4. Frolkis VV, Bezrukov VV, Shevtchuk VG: Hemodynamics
and its regulation in old aged. Exp Gerontol 1975;10:251-271
5. Templeton GH, Willerson JT, Platt MR, Weisfeldt M: Contraction duration and diastolic stiffness in aged canine left
ventricle, in Kobayashi T, Sano T, Dhalla NS (eds): Recent
Advances in Studies on Cardiac Structure and Metabolism: Heart
Function and Metabolism. Baltimore, University Park, 1978, pp
169-173
6. Lakatta EG: Cardiac muscle changes in senescence. Annu Rev
Physiol 1987;49:519-531
7. Froehlich JP, Lakatta EG, Beard E, Spurgeon HA, Weisfeldt
ML, Gerstenblith G: Studies of sarcoplasmic reticulum function and contraction duration in young adult and aged rat
myocardium. J Mol Cell Cardiol 1978;10:427-438
8. Narayanan N: Differential alterations in ATP-supported calcium transport activities of sarcoplasmic reticulum and sarcolemma of aging myocardium. Biochim Biophys Acta 1981;
678:442-459
234
Circulation Research Vol 67, No 1, July 1990
Downloaded from http://circres.ahajournals.org/ by guest on September 30, 2016
9. MacLennan DH, Brandl CJ, Korczak B, Green NM: Aminoacid sequence of a Ca2' +Mg2`-dependent ATPase from rabbit muscle sarcoplasmic reticulum, deduced from its complementary DNA sequence. Nature 1985;316:696-700
10. Goldberg P: Cardiac function of Fischer 344 rats in relation to
age, in Kaldor G, DiBattista WJ (eds): Aging. New York,
Raven Press, Publishers, 1978, vol 6, pp 87-100
11. Dillmann WH, Barrieux A, Neeley WE, Green NM: Influence
of thyroid hormone on the in vitro translational activity of
specific mRNAs in the rat heart. J Biol Chem 1983;
258:7738-7745
12. Rohrer DK, Dillmann WH: Rat cardiac sarcoplasmic reticulum Ca2++Mg2+ ATPase: Characterization of cDNA clones,
and thyroid hormone responsiveness of Ca2' ATPase mRNA
in cultured neonatal myocytes (abstract). J Cell Biochem
1989;13(E):210
13. Henderson SA, Xu Y-C, Chien KR: Nucleotide sequence of
full length cDNAs encoding rat cardiac myosin light chain-2
(abstract). Nucleic Acids Res 1988;16:4722
14. Richardson A, Semsei I: Effect of aging on translation and
transcription, in Rothestein M (ed): Review of Biological
Research in Aging, Vol. 3. New York, Alan R. Liss, Inc, 1987,
pp 467-483
15. Horbach GJMJ, Princen HMG, Van Der Kroef M, Van
Bezooijen CFA, Yap SH: Changes in the sequence content of
albumin mRNA and in its translational activity in the rat liver
with age. Biochim Biophys Acta 1984;783:60-66
16. Pekary AE, Hershman JM, Sugawara M, Gieschen KI, Sogol
PB, Reed AW, Pardridge WM, Walfish PG: Preferential
release of triiodothyronine: An intrathyroidal adaptation to
reduced serum thyroxine in aging rats. J Gerontol 1983;
38:653-659
17. Chen HJ, Walfish PG: Effect of age and ovarian function on
the pituitary-thyroid system in female rats. J Endocntnol 1978;
78:225-232
18. Fisher DA, Dussault IH: Development of the mammalian
thyroid gland, in Griep RO, Astwood EB (eds): Handbook of
Physiology, Section 7: Endocrinology, Volume III, Thyroid.
Washington, DC, American Physiological Society, 1974,
pp 21-38
19. Frolkis VV, Verhikovskaya NV, Valueva GV: The thyroid and
age. Exp Gerontol 1973;8:285-296
20. Felicetta JV: The thyroid and aging, in Sowers JR, Felicetta
JV (eds): The Endocrinology ofAging. New York, Raven Press,
Publishers, 1988, pp 15-39
21. Suko J: The calcium pump of cardiac sarcoplasmic reticulum:
Functional alterations at different levels of thyroid state in
rabbits. J Physiol (Lond) 1973;228:563-582
22. Rohrer D, Dillmann WH: Thyroid hormone markedly
increases the mRNA coding for sarcoplasmic reticulum Ca2'
ATPase in the rat heart. J Biol Chem 1988;263:6941-6944
KEY WORDS * Ca2+-ATPase
age effects
* sarcoplasmic reticulum
Downloaded from http://circres.ahajournals.org/ by guest on September 30, 2016
Age-induced decreases in the messenger RNA coding for the sarcoplasmic reticulum
Ca2(+)-ATPase of the rat heart.
L M Maciel, R Polikar, D Rohrer, B K Popovich and W H Dillmann
Circ Res. 1990;67:230-234
doi: 10.1161/01.RES.67.1.230
Circulation Research is published by the American Heart Association, 7272 Greenville Avenue, Dallas, TX 75231
Copyright © 1990 American Heart Association, Inc. All rights reserved.
Print ISSN: 0009-7330. Online ISSN: 1524-4571
The online version of this article, along with updated information and services, is located on the
World Wide Web at:
http://circres.ahajournals.org/content/67/1/230
Permissions: Requests for permissions to reproduce figures, tables, or portions of articles originally published
in Circulation Research can be obtained via RightsLink, a service of the Copyright Clearance Center, not the
Editorial Office. Once the online version of the published article for which permission is being requested is
located, click Request Permissions in the middle column of the Web page under Services. Further information
about this process is available in the Permissions and Rights Question and Answer document.
Reprints: Information about reprints can be found online at:
http://www.lww.com/reprints
Subscriptions: Information about subscribing to Circulation Research is online at:
http://circres.ahajournals.org//subscriptions/