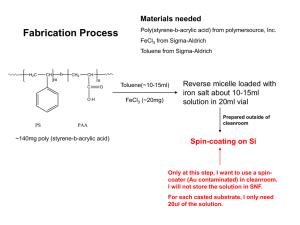
Technical Brief 2010 Volume 9
Cleanroom Operations
Background
Cleanrooms are defined as a
room or suite of rooms, in which
the concentration of airborne particles is maintained within established parameters and where other
factors are controlled to within
specified limits. These rooms are
designed to provide control of environmental factors including:
• Viable and non-viable airborne
particles
• Air flow patterns
• Temperature and humidity
• Air pressure differential
• Containment of hazardous
aerosols
Applications include the manufacture of sterile and non-sterile
pharmaceutical and biotech products, medical devices, and implants. These rooms are also used
to manufacture sensitive electronics. However, the requirements for
these latter applications are not
the objective of this article. GMP
requirements from the different
Boards of Health, including the
US Food and Drug Administration
(FDA), for sterile pharmaceutical,
biotech, medical devices and implants require the manufacture of
these products are performed in
clean environments that meet the
requirements of standards such as
ISO EN146441.
Compliance Requirements:
• FDA Guideline Sterile Drug
Products Produced by Aseptic
Processing2
• European Union (EU) GMP
Annex 13 Manufacture of Sterile
Medicinal Products
Other Helpful Documents:
• ISO EN14644-1, -2
• International Society for Pharmaceutical Engineering (ISPE4)
more restricted the air flow, the
In order to control contamination, more turbulence and this can
operators and those in charge of a cause undesireable particle
cleanroom need to be cognizant of movement.
sources of contamination. These • Filtration – In addition to the
HEPA filters commonly used in
include:
1. Facilities: walls, floors and cleanrooms, there are a number
ceilings; paint and coatings; spills of other filtration mechanisms
used to remove particles from
and leaks
gases and liquids used in the
2. People: skin flakes and oil; manufacture of pharmaceutical
cosmetics and perfume; spittle; products.
These filters are
clothing debris (lint, fibers, etc.); essential for providing effective
hair
contamination control.
3. Tool-generated:
friction
and • Cleanroom Garments – The
wear particles; lubricants and requirements for cleanroom
emissions; vibrations; brooms, garments will vary from location
mops, and dusters
to location. It is important
4. Fluids: particulates floating in air; to know the local cleanroom
bacteria, organics, and moisture; garment requirements. Gloves,
floor finishes or coatings; face masks and head covers
cleaning chemicals; plasticizers are standard in nearly every
(outgasses); water
cleanroom environment as well
5. Product-generated: glass flakes; as coveralls.
cleanroom debris; aluminum • Personnel - There are both
particles from vial caps
physical and psychological
when
humans
Key Elements of Contamination Control concerns
are present in cleanrooms.
• Cleanroom
Architecture
– Physical behavior like fast
Cleanrooms are designed to motion and horseplay can
achieve and maintain an airflow increase
contamination.
in which essentially the entire Psychological concerns like
body of air within a confined comfort, claustrophobia, strong
area moves with uniform velocity odors, and workplace attitude
along parallel flow lines. This air are important. Below are
flow is called laminar flow. The several ways people produce
contamination:
Table 1
◊ Behavior-- Rate of moveParticles/Minute (≥0.3 μ)
ment, sneezing and coughing
supplied in accordance with ventilation codes, to balance exhaust
air and to maintain specified pressures. Unless, otherwise specified, typical temperature range
for this kind of room is within the
range of 16 to 19 °C and relative
humidity of approximately 50% is
maintained. The type of equipment and number of people in
the room may dictate where in the
range you need to be to assure that
during production the operations
area is maintained at the right
temperature and humidity levels.
Only HEPA filtered air should
enter the cleanroom and the gowning areas. These modules are
available in fan assisted with fan
speed control and should operate at a velocity of 90 ± 10 fpm
or 0.45 ± 0.05 m/s. The location
of the HEPA filters and air return
grilles should create air movement
from the designated ‘clean zone’ to
the ‘less clean’ zones. Return air
grilles should be at a lower level to
aid in laminar flow requirements.
Air supply to the cleanroom
should provide a room air change
rate of >20 per hour. Air cleanliness will be enhanced by higher
air change rates. When the doors
are opened the supply air volume
should maintain an outward flow
of air.
People Activity
Sources of Contamination
Motionless (Standing or Seated)
100,000
Walking about 2 mph
5,000,000
Horseplay
100,000,000
Table 2
Maximum permitted
Old Fed Std 209E ISO EN14644 EU GMP Annex 1 particles per m3 equal to or
greater than tabulated size
Air Handling Requirements
Room air should be supplied
by an external air conditioning
system, preferably dedicated to
the facility. Partial recirculation
of room air is appropriate and this
allows for optimal energy utilization. Sufficient fresh air should be
◊ Attitude-- Work habits and
communication
between
workers
People are a major source of
contamination in the cleanroom
At rest
In operation
0.5 µ
0.5 µ
Maximum Microbiological
Active Air Action Levels
(cfu/m3)
100
ISO 5
Class A & B*
3,520
3,520
1
1,000
ISO 6
---
35,200
---
7
10,000
ISO 7
Class C
352,000
3,520,000
10
100,000
ISO 8
Class D
3,520,000 Not defined
100
*Requirements for particle counts during operation are different from class A, 35,200 particles at 0.5 µ
©2010 Particle Sciences, Inc. All rights reserved.
(Table 1). Notice the number of
particles produced per minute
during these activities.
Cleanroom Classifications
Cleanroom Classifications as
specified by different standards
are shown in Table 2. Non-sterile
products are typically produced in
an ISO 8 or EU Class D environment. Sterile products are required
to be filled and stoppered in an
ISO 5 or EU Class A environment.
Cleanroom Cleaning
As an example, here are some
recommended procedures for
cleaning a Class C or ISO 7 Area
cleanroom. These procedures are
simply guidelines, not standards
or rules. It is important to review
all cleaning procedures to be used
in a cleanroom with responsible
management. A detailed cleaning
schedule should be prepared for
every cleanroom.
Procedure
Good housekeeping and maintenance of the cleanroom and the
associated restricted areas are essential to assure quality. Cleaning
of an active cleanroom should be
performed daily. However, if the
room is not used daily, a different
schedule may be implemented,
but it should be cleaned after
every use. Improper cleaning of
the cleanroom can lead to contamination and compromise product quality. Proper selection of
equipment, cleaning agents and
cleaning materials is important
for proper cleaning. Only products that have proven cleanroom
performance records should be
considered for use. These products should be listed in appropriate policies or procedures and all
vendors should be informed about
the strict policies of how products are qualified. All procedures
should be strictly enforced. Below
are some examples of how to organize cleanroom cleaning. These
are guidelines for preparing work
procedures and schedules. Local
requirements must be included in
any cleaning program.
Equipment and Supplies – all supplies must meet the Class C or ISO
7 Area minimum requirements
• Cleaning
and
disinfecting
solutions
• Only garments approved for
• Cleanroom mops
the cleanroom should be worn
• Cleanroom vacuum cleaner
when entering.
(if allowed)
•
No cosmetics shall be worn in
• Cleanroom wipers
the cleanrooms. This includes:
• Cleanroom mop bucket and
rouge, lipstick, eye shadow,
wringer
eyebrow pencil, mascara, eye
Cleaning Tasks – frequency may
liner, false eye lashes, fingernail
vary depending upon local requirepolish, hair spray, mousse, or
ments
the heavy use of aerosols, after
• Cleaning of all work surfaces in
shaves and perfumes.
the controlled environment
• Only
approved
cleanroom
• Vacuuming (if allowed) of the
paper shall be allowed in the
floors and work surfaces
cleanroom.
• Emptying of appropriate trash • Gloves should not be allowed
and waste receptacles
to touch any item or surface
that has not been thoroughly
• Cleaning of the doors, door
cleaned.
frames and lockers in the prestaging area and gowning areas • Only approved gloves, pliers,
using the approved cleaning
tweezers should be used to
solution
handle product.
• Mop gowning area and cleanroom • All tools, containers and fixtures
floors
used in the cleaning process
should be cleaned to the
General Cleanroom Requirements
same degree as the cleanroom
Here is a list of general requiresurfaces.
ments recommended as a mini• No tool should be allowed to
mum for the successful operation
rest on the surface of a bench
of a cleanroom. All cleanroom peror table. It should be placed on
sonnel should be aware and follow
a cleanroom wiper.
these requirements at all times.
• Only
cleanroom
approved
• All personal items such as keys,
wipers are allowed to be used.
watches, rings, matches, lighters
The wipers must be approved
and cigarettes should be stored
for the class of cleanroom being
in the personal locker outside the
cleaned.
gowning room.
• All equipment, materials and
• Valuable personal Items such
containers introduced into a
as wallets may be permitted in
sterile facility must be subjected
the cleanroom provided they are
to stringent sterilization prior to
never removed from beneath the
entrance.
cleanroom garments.
• No one who is physically ill,
• No eating, smoking or gum
especially with respiratory or
chewing is allowed inside the
stomach disorders, may enter a
cleanroom.
sterile room.
Figure 1
NEGATIVE PRESSURE CLEANROOM FOR
HANDLING POTENT COMPOUNDS
Cleanroom
Z = P1
= Air Flow
Z = HEPA
Filtered Air
Gowning Area
Z = P2
3894 Courtney St., Bethlehem, PA 18017-8920, USA
Definitions
HEPA Filter - High Efficiency
Particulate Air Filter
Viable - a particle capable of
living, developing, or germinating
under favorable conditions, i.e.,
bacteria.
Non-viable - typically dust or liquid
particles
Airborne - carried by or through the
air
Cleanrooms for Handling Potent
Compounds
For handling potent compounds,
i.e., toxic materials, in addition to
the requirements described for a
cleanroom, precautions need to
be taken to avoid spreading the
toxic material to adjacent areas.
This may be achieved by maintaining a negative pressure differential between the cleanroom
and adjacent area so any hazardous powder or aerosol is contained
within the cleanroom, i.e., P3 ~
P2 > P1 (Figure 1). This is accomplished by designing the facility with HEPA filtered incoming
air and providing HEPA fil­tration
at the exhaust. Also providing a
higher flow through the exhaust
filters to assure proper flow, pressure differential and sufficient
air changes in order to meet the
required room classification; this
setup does not allow air from the
cleanroom to enter into adjacent
areas. Additionally, by providing
localized exhaust or incorporating isolators onto manufacturing
equipment, the control of aerosols
and dust from handling powders
is increased. Note that personnel
enter the cleanroom through the
gown­ing area whereas equipment
and materials are brought through
the air lock.
References
1. ISO EN 14644-1, -2, -5, -9
International Standards Organization Cleanroom Standards
2. FDA Guidance for Industry –
Sterile Drug Products produced by
Aseptic Processing – CGMP September 2004
3. EU GMP Annex 1- Manufacture of Sterile Medicinal Products
Air Lock
Z=P3
4. ISPE – International Society
for Pharmaceutical Engineers
Particle Sciences
Phone: +1 610 861 4701
Fax: + 1 610 861 4702
Email: info@particlesciences.com
www.particlesciences.com
Particle Sciences is a leading integrated
provider of formulation and analytic services
and both standard and nanotechnology
approaches to drug development and delivery.