The intensity of spectral lines depends on:
advertisement
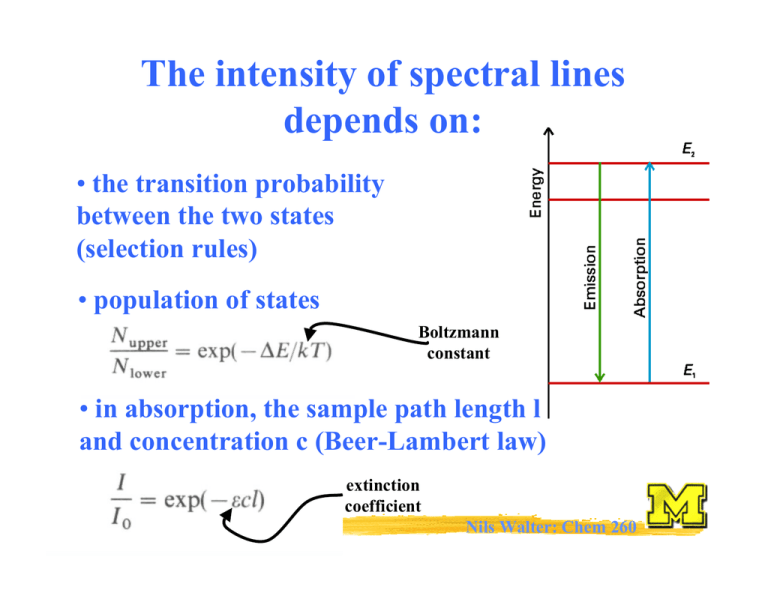
The intensity of spectral lines depends on: • the transition probability between the two states (selection rules) • population of states Boltzmann constant • in absorption, the sample path length l and concentration c (Beer-Lambert law) extinction coefficient Nils Walter: Chem 260 Rotational spectroscopy: The rotation of molecules z (IC) symmetric: IA=IB≠IC≠0 x (IA) y (IB) Rotor in 3-D linear: IA=0; IB=IC asymmetric: IA≠IB≠IC≠0 spherical: IA=IB=IC ≠0 Nils Walter: Chem 260 The moment of inertia for a diatomic (linear) rigid rotor r definition: r = r1 + r2 m1 m2 C(enter of mass) r1 Balancing equation: m1r1 = m2r2 Þ m1m2 2 r = µr 2 I= m1 + m2 r2 Moment of inertia: I = m1r12 + m2r22 µ = reduced mass Nils Walter: Chem 260 Solution of the Schrödinger equation for the rigid diatomic rotor h2 2 − ∇ Ψ = EΨ 2µ d d d 2 = dx 2 2 + dy 2 + 2 dz 2 (No potential, only kinetic energy) Þ EJ = hBJ(J+1); rot. quantum number J = 0,1,2,... D B= 4πI [Hz] Þ EJ+1 - EJ = 2hB(J+1) Þ microwave spectroscopy (GHz range) Nils Walter: Chem 260 Selection rules for the diatomic rotor: 1. Gross selection rule Light is a transversal electromagnetic wave Þ a molecule must be polar to be able to interact with light a polar rotor appears to have an oscillating electric dipole Nils Walter: Chem 260 Selection rules for the diatomic rotor: 2. Specific selection rule rotational quantum number J = 0,1,2,… describes the angular momentum of a molecule (just like electronic orbital quantum number l=0,1,2,...) and light behaves as a particle: photons have a spin of 1, i.e., an angular momentum of one unit and the total angular momentum upon absorption or emission of a photon has to be preserved Þ ∆J = ± 1 Nils Walter: Chem 260 Okay, I know you are dying for it: Schrödinger also can explain the ∆J = ± 1 selection rule Transition dipole moment µ fi = ò Ψ f µΨi dτ initial state final state Þ only if this integral is nonzero, the transition is allowed (for ∆J = ± 1) ; if it is zero, the transition is forbidden Nils Walter: Chem 260 The angular momentum 1 2 E = Iω 2 rotational frequency P = Iω classical rotational energy and with Þ E J = hBJ ( J + 1) D B= 4πI Þ P = 2 EI classical angular momentum mJ J=1 P = J ( J + 1) D But P is a vector, i.e., has magnitude and direction Nils Walter: Chem 260