View presentation
advertisement

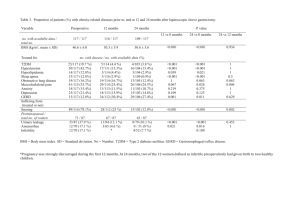
Economic Evaluation of Canagliflozin Versus Glimepiride and Sitagliptin in Dual Therapy With Metformin for the Treatment of Type 2 Diabetes in Italy R. Ravasio,1 P. Pisarra,2,* M. Comaschi3 1 Health Publishing & Services Srl, Milan, Italy; 2Janssen-Cilag S.p.A., Cologno Monzese, Milan, Italy; 3Diabetologia, Endocrinologia, Medicina Interna ICLAS, GVM Care & Research, Rapallo, Genoa, Italy. *Presenting author. ◾ Data from the 2013 Italian National Institute of Statistics statistical yearbook indicated that >3 million people (5.4% of the Italian population) have diabetes2; of these, ~90% have type 2 diabetes mellitus (T2DM)1 ◾ Lifestyle changes are initially recommended for the treatment of T2DM and remain the backbone of therapy, even after pharmacological intervention with metformin is initiated3,4 ◾ When additional glycaemic control is needed, other medications can be added to metformin3,5-7; however, some treatments have negative side effects (eg, weight gain, hypoglycaemia)3 ◾ Sodium glucose co-transporter 2 (SGLT2) inhibitors are a new class of medications for the treatment of adults with T2DM that lower blood glucose by increasing urinary glucose excretion (UGE) via an insulin-independent mechanism that is complementary to other classes of diabetes medications8 – Increased UGE also results in a mild osmotic diuresis and a net loss of calories in patients with T2DM, which may lead to weight loss and blood pressure reduction8 ◾ In contrast, dipeptidyl peptidase-4 (DPP-4) inhibitors (eg, sitagliptin) lower blood glucose but are not associated with weight loss or blood pressure reduction, and sulphonylureas (eg, glimepiride) are associated with weight gain and hypoglycaemia3 Genital Mycotic Infection Costs ◾ No cost was associated with genital mycotic infections, which were assumed to be self-managed with over-the-counter, antimycotic medications (eg, canacid) Projection Period – Canagliflozin 100 mg demonstrated non-inferiority in HbA1c lowering and canagliflozin 300 mg demonstrated superiority in HbA1c lowering as add-on to metformin versus either glimepiride or sitagliptin 100 mg at Week 5210,11 ◾ Sensitivity analyses were conducted to examine the uncertainty of the base case results per Italian guidelines for health care–related financial evaluations18 B. ◾ A literature search identified 1 article for glimepiride (Cefalu et al10) and 1 article for sitagliptin (Lavalle-González et al11) that were suitable for inclusion in the analysis ◾ Univariate analyses were conducted that varied the following parameters: – Costs and treatment setting of a hypoglycaemia episode ◾ Threshold analyses were also carried out on the following parameters: – Reduction in weight loss efficacy with canagliflozin ◾ In the add-on to metformin versus sitagliptin study, patients (N = 1,284) were randomised (2:2:2:1) to receive canagliflozin 100 or 300 mg, sitagliptin 100 mg, or placebo for 26 weeks; patients randomised to placebo switched to sitagliptin 100 mg during the 26-week extension period11 ◾ Baseline characteristics and a summary of the key results from both studies at Week 52 are shown in Table 1 Table 1. Clinical Inputs: Baseline Characteristics and Key Results at Week 52 Canagliflozin versus glimepiride10 Canagliflozin versus sitagliptin11 Canagliflozin Canagliflozin Canagliflozin Canagliflozin Sitagliptin 100 mg 300 mg Glimepiride 100 mg 300 mg 100 mg Baseline characteristics Number of patients Age, y Female, % Weight, kg BMI, kg/m2 HbA1c, % Duration of T2DM, y Results at Week 52 Change in HbA1c, % Total hypoglycaemia episodes,† % Mild to moderate‡ Severe Genital mycotic infections, % Change in weight % kg 483 56.4 48 86.9 31.0 7.8 6.5 –0.82* 485 55.8 50 86.6 31.2 7.8 6.7 –0.93* 482 56.3 45.0 86.5 30.9 7.8 6.6 –0.81 368 55.5 52.7 88.8 32.4 7.9 6.7 –0.73* 367 55.3 55 85.4 31.4 7.9 7.1 –0.88* 366 55.5 53.0 87.7 32 7.9 6.8 –0.73 5.6 4.9 34.2 6.8 6.8 4.1 5.2 0.4 4.3 0.6 31.1 3.1 6.5 0.3 6.8 0.0 3.8 0.3 8.9 11.1 1.7 8.4 6.5 1.9 –4.2 –3.7 –4.7 –4.0 1.0 0.7 –3.8 –3.3 –4.2 –3.7 –1.3 –1.2 BMI, body mass index. *Canagliflozin 100 mg demonstrated non-inferiority in HbA1c lowering and canagliflozin 300 mg demonstrated superiority in HbA1c lowering versus either glimepiride or sitagliptin 100 mg at Week 52. †Includes documented hypoglycaemia episodes from the clinical trial, defined as biochemically documented episodes (ie, glucose levels ≤3.9 mmol/L) with or without symptoms and severe episodes (ie, those requiring the assistance of another individual or resulting in seizure or loss of consciousness). ‡Includes documented hypoglycaemia episodes from the clinical trial that were not considered severe. ◾ Based on a conservative hypothesis of therapeutic equivalence, the models estimate the economic impact associated with the introduction of canagliflozin in the treatment of T2DM compared to the most common and widely used oral antidiabetic drugs (sulphonylureas) and those of the latest generation (DPP-4 inhibitors) ◾ The comparison of canagliflozin versus glimepiride and sitagliptin was assessed in the context of a cost-minimisation analysis (CMA) that considered only direct health care costs (eg, medicinal products, hospitalisations, specialist medical care, glycaemic self-monitoring, hypoglycaemic events) associated with the treatment of T2DM and body weight changes in patients with T2DM12 Use of Health Care Resources and Value Analysis Diabetes Medication Costs ◾ Diabetes medication costs were calculated based on dosages used in the studies; the average daily costs for canagliflozin 100 and 300 mg, sitagliptin 100 mg, and glimepiride were €1.34, €1.99, €1.28, and €0.118, respectively13 Weight Changes ◾ The relationship between weight change and health care costs was obtained from a study in patients with T2DM, where each 1% loss in body weight significantly decreased health care costs associated with T2DM management by 3.6% (P <0.05) and each 1% weight gain was not related to a statistically significant increase in treatment costs12 Patient Costs ◾ An Italian national survey estimated the total annual cost incurred by a patient with T2DM to be €2,756 (€814 for medications, €1,569 for hospitalisations, and €373 for specialist medical care)14 ◾ For this analysis, a total cost of €2,585 was used, with the cost of antidiabetic medications (€171) excluded to avoid a double-counting error Hypoglycaemia Costs ◾ It was assumed that a hypoglycaemia episode could be treated at home, with access to emergency care or hospitalisation if needed15 – No cost was associated with a hypoglycaemia episode treated at home (assumed 86% of mild-tomoderate episodes15) – The cost for emergency care was €133.89 based on the national fees associated with health care services provided to the patient16 (assumed 14% of mild-to-moderate episodes and 29.8% of severe episodes15) 5 10 Glimepiride 15 20 25 30 2,600 2,300 5 10 ◾ Canagliflozin 100 and 300 mg were associated with a lower annual cost per patient than sitagliptin 100 mg (Table 2) – The cost savings with canagliflozin 100 and 300 mg were due primarily to the differences in costs related to a greater loss in body weight; cost differences associated with other components (eg, management of hypoglycaemia, SMBG) were negligible Medications for diabetes Other medications Hospitalisations Specialist medical care Hypoglycaemia (mild to moderate) Hypoglycaemia (severe) Hypoglycaemia self-monitoring Total per treated patient 20 25 30 €43.05 €487.76 €724.36 €465.92 €545.78 €1,331.77 €534.20 €1,303.53 €643.00 €1,569.00 €555.04 €1,354.36 €545.78 €1,331.77 €612.91 €1,495.57 €316.60 €309.89 €373.00 €321.97 €316.60 €355.54 Average costs per patient (€) Canagliflozin 300 mg p = 39.8% 3,200 2,900 2,600 2,300 0 5 10 15 20 25 30 35 40 Percent reduction in the number of SMBG measurements with glimepiride Note that p represents the percent reduction at which canagliflozin is no longer cost-saving versus glimepiride. Figure 2. Threshold analysis results for (A) reduction in weight loss efficacy with canagliflozin and (B) reduction in patient treatment costs. Canagliflozin Canagliflozin Canagliflozin Canagliflozin Sitagliptin 100 mg 300 mg Glimepiride 100 mg 300 mg 100 mg €724.36 Canagliflozin 100 mg 2,000 Canagliflozin versus sitagliptin €487.76 Glimepiride 3,500 A. Sitagliptin 100 mg 3,300 Canagliflozin 100 mg Canagliflozin 300 mg p = 4.2% 3,200 3,100 3,000 2,900 2,800 2,700 0 5 10 15 20 25 30 Percent reduction in weight loss efficacy with canagliflozin €0.97 €0.81 €5.83 €1.22 €1.27 €0.72 €8.40 €12.55 €63.13 €5.51 €0.00 €5.54 €94.18 €94.18 €470.89 €94.18 €94.18 €94.18 €2,785.46 €2,979.52 €3,167.90 €2,820.05 €3,013.96 €3,030.38 Sensitivity Analyses B. ◾ Threshold analyses showed that canagliflozin 100 or 300 mg continued to represent a cost-saving option versus glimepiride, unless the number of SMBG measurements was reduced substantially (>39.8%; Figure 1) ◾ Threshold analyses confirmed the base case results that canagliflozin 100 mg was associated with cost savings versus sitagliptin 100 mg at all values for reduction in weight loss efficacy with canagliflozin and reduction in costs for managing a patient with T2DM, while uncertainty was greater for the comparison of canagliflozin 300 mg and sitagliptin 100 mg (Figure 2) – Canagliflozin 300 mg provided cost savings compared with sitagliptin 100 mg unless weight loss efficacy with canagliflozin was reduced by >4.2% or patient treatment costs were reduced by >6.1% Canagliflozin 300 mg 3,200 p = 6.1% 2,900 2,600 2,300 2,000 0 5 10 15 20 25 30 Percent reduction in patient treatment costs Note that p represents the percent reduction at which canagliflozin is no longer cost-saving versus sitagliptin. DISCUSSION ◾ This CMA identifies canagliflozin 100 or 300 mg as a cost-saving treatment option compared with glimepiride and sitagliptin 100 mg for the treatment of patients with T2DM inadequately controlled with metformin Canagliflozin versus sitagliptin Canagliflozin Canagliflozin Canagliflozin Canagliflozin Sitagliptin 100 mg 300 mg Glimepiride 100 mg 300 mg 100 mg €2,979.52 Canagliflozin 100 mg – Results from the present CMA, conducted from the Italian perspective, are consistent with cost-efficiency data from the United States, which demonstrated lower annual costs for treatment with canagliflozin 300 mg versus sitagliptin 100 mg19 Table 3. Sensitivity Analysis Results Base case €2,785.46 Costs for genital mycotic infection treatment One canacid package, Italian €2,787.07 National Health Service Costs for hypoglycaemia hospitalisation ICD-9-CM 250.3 and 250.8 €2,787.91 secondary diagnosis ICD-9-CM 250.3 and 250.8 main €2,787.03 and secondary diagnosis Costs for emergency care of hypoglycaemia –25% €2,785.17 +25% €2,785.74 Hypoglycaemia setting Mild to moderate: 100% treated in €2,791.42 emergency care facilities Severe: 100% treated in emergency €2,788.79 care facilities Sitagliptin 100 mg 3,500 ◾ Sensitivity analyses confirmed the base case results, which indicated that canagliflozin 100 and 300 mg were associated with lower costs versus glimepiride or sitagliptin 100 mg (Table 3) Univariate analysis 15 Percent reduction in patient treatment costs ◾ Canagliflozin 100 and 300 mg were associated with a lower annual cost per patient compared with glimepiride and, therefore, resulted in cost savings (Table 2) Cost items Canagliflozin 300 mg 2,900 0 Base Case Canagliflozin versus glimepiride Canagliflozin 100 mg 3,200 – Reduction in the number of SMBG measurements associated with glimepiride Canagliflozin versus glimepiride Study Design 2,800 2,000 – Decrease in treatment costs for a patient with T2DM Table 2. Base Case Results Clinical Inputs ◾ In the add-on to metformin versus glimepiride study, patients (N = 1,450) were randomised (1:1:1) to receive canagliflozin 100 or 300 mg or glimepiride over 52 weeks10 2,900 3,500 Average costs per patient (€) ◾ Based on the availability of head-to-head clinical trials with canagliflozin and their use in Italian clinical practice, 1 sulphonylurea (glimepiride) and 1 DPP-4 inhibitor (sitagliptin) were selected for this financial evaluation 3,000 Sensitivity Analyses Average costs per patient (€) Clinical Data and Treatments 3,100 Percent reduction in weight loss efficacy with canagliflozin – Costs for diabetes medications were higher with canagliflozin 100 and 300 mg compared with glimepiride; these higher costs were completely offset by cost differences related to a reduction in body weight (ie, other medications, hospitalisations, and specialist medical care) and a reduction in costs associated with the management of SMBG METHODS 3,200 0 RESULTS ◾ To determine the cost savings associated with canagliflozin 100 or 300 mg compared with either glimepiride or sitagliptin 100 mg for a patient with T2DM inadequately controlled with metformin, from the perspective of the Italian National Health Service Canagliflozin 300 mg 2,700 C. OBJECTIVE Canagliflozin 100 mg 3,300 ◾ The projected period for this analysis was 12 months (ie, 52 weeks), consistent with the duration of the 2 clinical studies – Costs for the treatment of a genital mycotic infection ◾ Canagliflozin, an SGLT2 inhibitor, has been associated with improvements in glycaemic control, body weight and blood pressure reductions, and a low risk of hypoglycaemia across studies in patients with T2DM who were on various background therapies, including metformin9 Glimepiride A. Average costs per patient (€) ◾ Surging obesity, an aging population, and increasingly sedentary lifestyles have led to a progressive worldwide increase in the observed prevalence of diabetes1 Figure 1. Threshold analysis results for (A) reduction in weight loss efficacy with canagliflozin, (B) reduction in patient treatment costs, and (C) reduction in the number of SMBG measurements with glimepiride. Average costs per patient (€) INTRODUCTION Self-monitoring of Blood Glucose Costs ◾ The costs associated with self-monitoring of blood glucose (SMBG) were calculated by attributing a value to the daily average usage of reaction strips and finger pricks per Italian guidelines (an average of 2 per day for patients treated with glimepiride and an average of 3 per week for patients treated with sitagliptin and canagliflozin),7 and an average national cost calculated based on regional reimbursement prices, when available €3,167.90 €2,820.05 €3,013.96 ◾ Cost savings per patient treated with canagliflozin versus glimepiride were attributable to all the cost items being considered in this analysis (ie, weight loss, reduced risk of hypoglycaemia, lower number of SMBG measurements) €3,030.38 ◾ Cost savings per patient with canagliflozin versus sitagliptin 100 mg primarily resulted from lower treatment costs associated with body weight reduction ◾ Sensitivity analyses generally support the base case findings €2,981.51 €3,168.20 €2,821.55 €3,015.13 €3,030.72 CONCLUSION €2,983.18 €3,186.34 €2,821.66 €3,013.96 €3,032.00 €2,981.86 €3,179.72 €2,821.08 €3,013.96 €3,031.42 ◾ This analysis demonstrates that canagliflozin 100 or 300 mg are likely to be the best cost-saving options compared with glimepiride and sitagliptin 100 mg in the treatment of patients with T2DM inadequately controlled with metformin in Italy, which may allow for more efficient allocation of resources available to the Italian National Health Service REFERENCES €2,979.25 €2,979.78 €3,166.13 €3,169.67 €2,819.71 €2,820.38 €3,013.64 €3,014.28 €3,030.17 €3,030.59 €2,984.50 €3,203.73 €2,827.56 €3,021.79 €3,034.79 €2,984.49 €3,192.93 €2,822.23 €3,013.96 €3,032.58 ICD-9-CM, International Classification of Diseases, 9th Revision, Clinical Modification. 1. International Diabetes Federation. IDF Diabetes Atlas. Brussels, Belgium: International Diabetes Federation; 2013. 2. Istituto Nazionale di Statistica. http://www.istat.it/it/archivio/ 107568. Accessed 30 April 2015. 3. Inzucchi SE, et al. Diabetes Care. 2015;38(1):140-149. 4. American Diabetes Association. Diabetes Care. 2015;38(suppl 1): S41-S48. 5. Cook MN, et al. Diabet Med. 2007;24(4):350-358. 6. Brown JB, et al. Diabetes Care. 2010;33(3):501-506. 7. Associazione Medici Diabetologi (AMD) - Società Italiana di Diabetologia (SID). http://www.standarditaliani.it/skin/ www.standarditaliani.it/pdf/STANDARD_2014_May28.pdf. Accessed 4 August 2015. 8. Chao EC, Henry RR. Nat Rev Drug Discov. 2010;9(7):551-559. 9. Rosenthal N, et al. Ann N Y Acad Sci. 2015. doi:10.1111/nyas.12852. 10. Cefalu WT, et al. Lancet. 2013;382(9896):941-950. 11. Lavalle-González FJ, et al. Diabetologia. 2013;56(12):2582-2592. 12. Yu AP, et al. Curr Med Res Opin. 2007;23(9):2157-2169. 13. Medico Facile. http://www.medicofacile.it/cerca_farmaci/ principi-attivi-farmaci-italia. Accessed 6 October 2015. 14. Osservatorio ARNO Diabete. https://osservatorioarno.cineca.org/diabete/doc/Rapporto_ ARNO_Diabete_2011.pdf. Accessed 4 August 2015. 15. Aglialoro A, et al. Il Giornale di AMD. 2013;16:433-438. 16. Ministero della Salute. http://www.crob.it/crob/files/docs/10/ 63/33/DOCUMENT_FILE_106333.pdf. Accessed 6 October 2015. 17. De Berardis G, et al. IsF. 2014;38(1). 18. Fattorre G. PharmacoEconomics – Italian Research Articles. 2009;11(2):83-93. 19. Ektare VU, et al. Am J Manag Care. 2014;20(10 suppl):S204-S215. ACKNOWLEDGEMENTS This analysis was made possible by an unconditional grant provided by Janssen-Cilag S.p.A., and was based on data from clinical studies funded by Janssen Research & Development, LLC. Medical writing support was provided by Alaina DeToma, PhD, of MedErgy, and was funded by Janssen Pharmaceutica NV. Canagliflozin has been developed by Janssen Research & Development, LLC, in collaboration with Mitsubishi Tanabe Pharma Corporation. – The average (weighted) cost per hospitalisation was €2,833 based on diagnosis-related group (DRG) tariffs, as identified in a study conducted by Fondazione Mario Negri Sud17 (assumed 70.2% of severe episodes15) POSTER PRESENTED AT THE INTERNATIONAL SOCIETY FOR PHARMACOECONOMICS AND OUTCOMES RESEARCH (ISPOR) 18TH ANNUAL EUROPEAN CONGRESS; 7-11 NOVEMBER 2015; MILAN, ITALY.