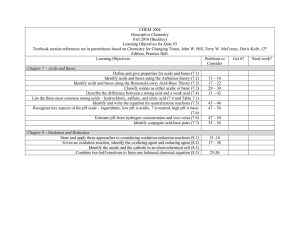
Review Exercises Chapter 11 a._____________________
advertisement

Review Exercises Chapter 11 1. List 3 properties of water that are unique: a._____________________ b. ____________________ c._____________________ 2. The water molecule has a ____________ shape and as a result it is involved in _______________bonding. 3. Hydrogen bonding occurs when atoms of each of the following 3 elements is bonded to hydrogen (H): a.__________ b.___________ c.____________ 4. Hydrogen bonding in water occurs because the partially positive _____ atom(s) are attracted to the partially negative _______ atom(s). 5. Mixtures can be either __________________, when two substances are not soluble in one another, or _________________, when two substances are soluble in one another. Solutions are ______________mixtures. 6. Identify each of the following as a homogeneous (HO) or a heterogeneous (HE) mixture: a. vinegar____________ b. white wine_____________ c. orange juice______________ d. stainless steel________________ e. air__________________ f. oil and vinegar_____________ 7. The solubility of substance “A” in water is 25 g per 100 g of water. Identify each of the following as saturated (s), or unsaturated (u): a. 50 g of “A” in 100 g of water________________ b. 2.5 g of “A” in 10 g of water _______________ c. 10 g of “A” in 100 g of water_______________ d. 50 g of “A” in 200 g of water________________ 8. Indicate whether the following substances are soluble (S) or insoluble (I) in water: a. KBr________________ b. ethyl alcohol, a polar substance_______________ c. carbon tetrachloride________________ d. diethyl ether, a nonpolar substance_____________ 9. Soap has an ionic end and a nonpolar end. The __________end dissolves oils and greases, the__________dissolves in water. 10. Based on the charges of the ions, would you expect each of the following ionic compounds to be soluble (S) or insoluble (I) in water? a. CaCO3________________ b. NaNO3_________________ c. Li2SO4_________________ d. AlPO4__________________ 11. If a solution is said to have a concentration of 56% by mass this means that for every __________ g of solution there are ____________g of solute. 12. Most ionic compounds_____________their solubility in water when the temperature increases. All gases ______________their solubility in water when the temperature increases. 13. Which of the following substances are expected to be electrolytes (E) or nonelectrolytes (NE)? a. Ba(NO3)2______________ b. ethyl alcohol_____________ c. sucrose (table sugar)______________ d. water_______________ e. HCl_________________ f. NaOH___________________ g. NH3_____________________ 14. Complete the following for the electrolytes listed. Make sure you differentiate between strong and weak electrolytes: a. HBr H2O b. KOH H2O c. HF H2O d. NH3 H2O 15. Which compounds ionize in water: a.__________________ b. _________________ c.__________________ 16. As the atmospheric pressure decreases the boiling point of a substance _____________. The boiling point of a substance at 1 atm of pressure is called the ______________ boiling of the substance. 17. If a solute is dissolved in a solvent, the boiling point of the solvent _______________ and the melting point _________________. 18. Classify each of the following acids and bases as strong acids (SA), strong bases (SB), weak acids (WA), or weak bases (WB): a. HCl_______________ b. NaOH______________ c. HF________________ d. HC2H3O2_____________ e. NH3________________ f. H2SO4_______________ g. Ca(OH)2____________ 19. Solutions with a pH of 7 are ___________, pH greater than 7 are ___________ and pH less than 7 are ______________. 20. List two properties each for acids and for bases: a. acids: b. bases: _______________________________________________________ _______________________________________________________ 21. What are two reasons why salts are important in your diet? a.______________________________ b.______________________________ 22. Predict the product of the following neutralization reactions: a. RbOH + HNO3 b. H2SO4 + KOH Answers: 1. high boiling point, high specific heat, high latent heat, lower density for solid than for liquid. 2. V-shape, hydrogen bonding. 3. O, F, N 4. hydrogen, oxygen 5. heterogeneous, homogeneous, heterogeneous. 6. HO, HO, HE, HO, HO, HE 7. S, S, U, S 8. S, S, I, I 9. nonpolar, polar 10. I,S,S,I 11. 110, 56 12. increase, decrease 13. E, NE, NE, NE, E, E, E 14. a. HBr H+(aq) + Br -(aq) b. KOH K+(aq) + OH-(aq) c. HF H+(aq) + F -(aq) d. NH3 NH4+(aq) + OH-(aq) 15. Acids, bases, soluble salts. 16. decrease, normal 17. increases, decreases 18. SA, SB, WA, WA, WB, SA, SB 19. neutral, basic, acidic 20. Acids: sour, litmus turns red, release H2 gas when react with water. Bases; bitter, litmus turns blue, slippery 21. Provide electrolytes and source of certain elements needed in the diet. 22. a. RbOH + HNO3 RbNO3 + H2O b. 2KOH + H2SO4 K2SO4 + 2H2O