WATER MANAGEMENT GOALS salinity and sodicity aspects
advertisement

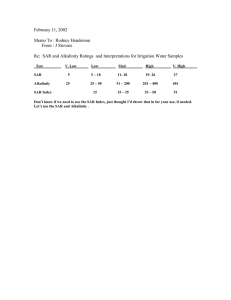
WATER MANAGEMENT GOALS:
salinity and sodicity aspects
• Profitable crop yields
ECiw and ECe
• Maintain or improve soil physical properties.
EC/SAR
• Consider environmental impacts (N, P, TDS,
pesticides)– subsurface flows to aquifers and
surface return flows to streams
• Fertilizer management: take into
account the N and S content in the
irrigation water.
– 5 ppm N in irrigation water times 2.7
equals 13 lbs of N per acre-foot of water.
– 5.3 ppm SO4 equals 4.7 lbs of S per acre
foot. See the footnotes for how this was
calculated.
• Liming potential: The waters of SE Mo
have a ‘hardness’ (Ca and Mg
carbonates) that range from 50 to about
400 mg/L.
– 200 mg/L of ‘hardness’ has a calcium
carbonate equivalent to about 500 lbs per
acre foot of water.
• Impacts of liming potential on zinc
nutrition.
• Ion Toxicity concerns: Na, Cl, B
• Operation concerns: prevent clogging
(Fe problem)
Electrical conductivity, EC
A quick method to measure the salinity of
water.
EC is approximately one-tenth of the total
dissolved cation, or anion concentration.
EC units for salt tolerance: dS/m or
mmho/cm
not micromho/cm (μmho/cm)
5 mmho/cm = 5 dS/m = 5000 μmho/cm
Sodium Adsorption Ratio, SAR
• Based on the sodium, calcium and
magnesium concentration in solution
– SAR = Na/{sqrt(Ca + Mg)/2}
• A rapid method to estimate the
exchangeable sodium percentage.
– ESP SAR
Remember water quality indices
• EC (electrical conductivity) is the salinity
index. ECiw for irrigation water and ECe
for water extracted from a saturated
soil paste.
• SAR (sodium adsorption index) is the
sodicity index. SAR is about equal to
the exchangeable sodium percentage.
Excess salinity drought
Upon sudden exposure to salinity
1.
Plants wilt
but in a few hours
2.
Plants recover
Excess salinity increases the energy plants
must expend to grow: when levels are
excessive, plant growth rates and yields are
reduced.
The salinity of the soil water reduces crop
growth if the average rootzone salinity,
(ECe), exceeds the threshold level for the
crop.
Saturation extract electrical conductivity, ECe
Soil
Surface
ECiw
Average
Rootzone
Salinity,
Bottom
of the
rootzone
ECe
YIELD RESPONSE TO SALINITY
Average Rootzone Salinity, ECe
100 %
Threshold
salinity
Crop Yield
0%
Slope
Crop salt tolerance
Crop
Corn
Alfalfa
Rice
Soybean
wheat
Cotton
Threshold
(t)
1.7
2.0
3.0
5.0
6.0
7.7
Slope
(s)
12.0
7.3
12.0
20.0
7.1
5.2
Water Quality, SE Mo, EC
7
6
EC, dS/m
5
median
4
Lo
3
2
Grape
Corn
Alfalfa
1
0
surface
ditch
Alluvial
Wilcox
Paleozoic
McNairy
Water Quality, SE Mo, EC
8
Cotton
7
Sorghum
Wheat
EC, dS/m
6
5
Hi
median
4
Lo
Rice
3
2
1
0
surface
ditch
Alluvial
Wilcox
Paleozoic
McNairy
Chloride and sodium -- can damage
some plants, but this depends on the
irrigation method
Surface or drip irrigation: potential problem for
trees and shrubs, usually not for grass, grain,
fiber, and forage crops.
Sprinkler irrigation:which wets the leaves
intermittently during the day can damage
leaves for many plants, if the sodium or
chloride concentration in the irrigation water
is greater than 5 meq/l.
Na/Cl foliar damage from sprinkler irrigation,
concentrations in meq/L.
5 – 10 meq/L
% of grnd water: 4
10 – 20
% of grnd
water: 3 - 4
>20
% of grnd
water: ~1 %
Grape
Alfalfa
Cotton
Potato
Corn
Sorghum
Growth stage effects:
exceptions to
the time-depth average assumption.
• Germination: Salt stress delays germination.
• Emergence: Plants are sensitive during
emergence and early seedling development.
• Reproductive stages: Salinity stress during
spike differentiation reduces yield.
Climate
• “Climate probably influences the response of
plants to salinity as much as, if not more than,
any other factor. Most crops can tolerate
greater salt stress if the weather is cool and
humid than when it is hot and dry.” Maas,
1990.
• These effects have been observed on alfalfa,
strawberry clover, and salt grass.
Fertility-salinity interactions
• Grattan, S.R., and C.M. Grieve. 1999.
Salinity-mineral nutrient relations in
horticultural crops. Scientia
Horticulturae 78:127-157.
• Shalhevet, Y. 1994. Using water of
marginal quality for crop production:
major issues. Agr. Water Management
25:233-269.
Nitrogen
• Nitrogen applied above levels
considered optimum under non-saline
conditions will not increase plant
growth in saline soils.
• Saline conditions reduce plant growth,
thereby reducing nitrogen needed by
the crop.
Effects of salinity and sodicity on
water infiltration into soils.
Water in an unsaturated coarsetextured soil.
Possible arrangements of quartz
particles, clay domains, and organic
matter in a soil aggregate.
Dr. J. P. (Jim) Quirk
Dept. Soil Science and Plant Nutrition,
Un. of W. Australia, Nedlands, WA
At A, the permeability was reduced 15 %: Quirks
choice -- first observable impaired soil structure
(TEC).
At B, drainage water was turbid -- dispersed clay
(TUC)
Relative
Hydraulic
Conductivity
1.2
0.3
Salinity, dS/m
Mechanisms
40
TUC
TEC
30
SAR
Swelling - blocking
conducting pores
Aggregate failure -unequal swelling
throughout the soil
Deflocculation -- clay
particles separated to
distance where
repulsive forces
dominate -clays
disperse and move
20
10
0
0
0.5
1
1.5
Salinity, dS/m
2
2.5
McNeal and Coleman, 1966
California Soils
SAR
50
40
30
Pachappa sl
Grangeville sl
Oasis cl
Waukena cl
20
10
0
0
1
2
3
4
Salinity, dS/m
The data obtained by McNeal is in reasonable
agreement with the TEC function of Quirk and
Schofield
From hydraulic conductivity to
infiltration -- from within the soil to the
soil surface.
Impact of water drops,
rapid soil wetting,
overland water flow
cause physical disintegration of soil
aggregates, clay dispersion and
compaction at the soil surface.
Infiltration rates are
particularly sensitive to salinity
and SAR
At the soil surface both are closely linked
to the SAR and salinity of the irrigation
water.
At the soil surface, the EC and SAR of the
soil quickly approaches that of the
irrigation water.
RNa is the same as SAR.
Photo taken
through a
microscope
(60x)
showing
three clay
layers.
TUC
TEC
Water Quality, SE Mo, SAR-EC
40
Water Quality, SE Mo, SAR-EC
W ater Q u ality, S E Mo , S AR -E C
35
40
40
S EV .
35
30
M O D . R ES T R IC T IO N
W ater Q u ality, S E Mo , S AR -E C
40
30
30
S EV .
SAR
25
25
S A R a llu v
25
SAR
SAR
SAR
15
20
N O R ES T R IC T IO N
30
T UC
20
M O D . R ES T R IC T IO N
RS TR.
35
25
N O R ES T R IC T IO N
35
RS TR.
20
T EC
S A R d itc h
T UC
T EC
S A R P a le o
15
15
S A R W ilc o x
10
15
SAR Wilcox
2
3
4
5
6
ditch
S A R P a le o
McNairy
S A R W ilc o x
Paleo
E C , d S /m
surface
0
0
10
1
5
0
alluv
0
0
5
S A R d itc h
SAR surface S A R s u r fa c e
5
10
SAR ditch
SAR Paleo
S A R s u r fa c e
10
S A R a llu v
SAR McNairy S A R M c Na ir y
S A R M c Na ir y
20
SAR alluv
0
1
2
1
2
33
E C , d S /m
EC, dS/m
4
4
5
5
6
6
Wilcox
5
0
0
1
2
3
4
5
6
Water Quality, SE Mo, SAR-EC
40
Water Quality, SE Mo, SAR-EC
W ater Q u ality, S E Mo , S AR -E C
Severe Reduction
35
40
S EV .
M O D . R ES T R IC T IO N
W ater Q u ality, S E Mo , S AR -E C
40
30
30
S EV .
15
20
S A R a llu v
25
SAR
20
N O R ES T R IC T IO N
30
T UC
SAR
SAR
25
SAR
25
M O D . R ES T R IC T IO N
RS TR.
35
25
reduction
N O R ES T R IC T IO N
35
RS TR.
35
30
slight to moderate
40
20
T EC
S A R d itc h
T UC
T EC
S A R P a le o
15
15
S A R W ilc o x
10
15
SAR Wilcox
2
3
4
5
6
ditch
S A R P a le o
McNairy
S A R W ilc o x
Paleo
E C , d S /m
surface
No reduction
0
0
10
1
5
0
alluv
0
0
5
S A R d itc h
SAR surface S A R s u r fa c e
5
10
SAR ditch
SAR Paleo
S A R s u r fa c e
10
S A R a llu v
SAR McNairy S A R M c Na ir y
S A R M c Na ir y
20
SAR alluv
0
1
2
1
2
33
E C , d S /m
EC, dS/m
4
4
5
5
6
6
Wilcox
5
0
0
1
2
3
4
5
6
Rainfall -- major hazard
• Rainfall coupled with irrigation with low
sodicity waters (5 < SAR < 10) may cause
enhance runoff and erosion.
• Fresh organic matter content and
crop/stubble cover can be expected to
compensate (somewhat) the effect of rainfall
on soils irrigated with low sodicity waters.
• Available data indicate these effects can not
be predicted – be a good observer/manager.
Iron problem
• Most of the groundwaters in SE Missouri
have moderate clogging potentials due to
their iron contents.
• The red numbers in the following table are
the percentages of the groundwaters that
have iron concentrations in the range shown
by the white numbers.
Iron clogging potential
Parameter
Clogging Potential
Low
Iron, mg/L
< 0.1
Moderate
38%
0.1 – 1.5
High
60% > 1.5
2%
Ferrous iron is much more soluble than ferric iron.
Oxidation of ferrous iron to ferric iron, by bacteria or by
air, results in a formation of ochre and/or bacterial
slimes.
These can clug intake screens of wells, and the filters,
laterals and emitters of a drip irrigation system
Iron – ochre, red oxide films, and
bacterial slimes
1. Exposure of well waters to air will cause
iron to be oxidized: red oxide films on
exposed surfaces is one result, another is
large deposits of iron oxide in pipes known
as ochre,
2. In anaerobic well waters, bacteria grow just
below the static level of a well where
pumping agitates and aerates the water.
Iron bacteria obtain their energy from
oxidizing iron. Once started iron bacteria
develop slimes throughout the interior of the
well casing, mainlines, and laterals.
Iron removal
•
•
•
•
Aeration
Clorination (also kills bacteria)
Settling basins
Filtration
Well treatment for iron clogging
• Chlorination
• Acid treatment
• Brushing
More details are given by Juhdorff – see
footnote for citation.
Iron and manganese
• Manganese is usually also present in
waters that contain iron.
Water quality criteria for drip irrigation
Parameter
Clogging Potential
Low
Moderate
High
Mn mg/L
< 0.1
12%
0.1 – 1.5
33% > 1.5
55%
Iron, mg/L
< 0.1
38%
0.1 – 1.5
60% > 1.5
2%
The clogging potential due to oxidation of
manganese ranges from moderate to high for
most of the groundwaters.
Manganese chemistry is similar to iron
chemistry.
Water quality criteria for drip irrigation
Parameter
Clogging Potential
Low
Dis. Solids, mg/L < 500
Moderate
87%
High
500 – 2000 12% > 2000
1%
Acidification of water to a pH of about 6.5 will
reduce clogging due to precipitation of Calcium
as calcium carbonate, also referred to as soil
lime.
• Know the chemical composition of the water.
• Consultants and Specialist are available who
can help interpret the chemical composition.
Thanks for the opportunity.
J. D. (Jim) Oster
Emeritus Specialist
University of Ca.
Oster@mail.ucr.edu