Electromagnetic Radiation and Energy
advertisement

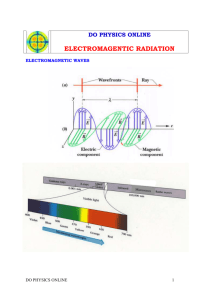
Electromagnetic Radiation and Energy Wave Description of Light • Electromagnetic radiation (ER): form of energy that exhibits wavelike behavior as it travels through space • All forms of ER together make the electromagnetic spectrum • All forms of ER move at a constant speed of about 3.0 x108 m/s (speed of light, c) • Wavelength (λ): distance between corresponding points on adjacent waves. • Frequency (ν): number of waves that pass a specific point in a given time, usually one second. • Unit: Hertz (Hz), aka (1/s) or (s-1) • For electromagnetic radiation, frequency and wavelength are related C=λν • If λ increases, what must happen to ν? Does c change? Practice • Calculate the wavelength of a wave with a frequency of 7.0 x 1016 Hz. • Pp 20 #3,4 Photoelectric Effect • Photoelectric effect: emission of electrons from a metal when light shines on that metal • Experiment: no e- were emitted if light’s frequency was below a certain threshold, regardless of time shone. • According to wave theory, shining any light long enough should supply enough energy to eject an e- Particle description of light • Max Planck, 1900s, suggested that object emit energy in small, specific amounts called quanta • Quantum: minimum amount of energy that can be gained or lost by an atom • Planck proposed a relationship between a quantum of energy and the frequency of radiation E=hν • E is energy, in Joules, of a quantum of radiation • h is Planck’s constant (fundamental physical constant)= 6.626 x 10-34 J*s • ν is freqency of radiation • 1905 Einstein expands on this idea. ER have dual wave/particle nature. • While light emits many wavelike particles, it can also be thought of as a stream of those particles • Einstein named the particles photons • Photon: particle of ER having zero mass and carrying a quantum of energy • Energy of a particular photon depends on the frequency of the radiation Ephoton= hν • Einstein’s Explanation: ER is absorbed by matter only in whole numbers of photons – For e- to be ejected, must be struck by single photon possessing at least minumum energy – According to equation, this energy corresponds to frequency – If photon’s frequency is below minimum, no e- ejected Practice • PP 20 Work problems 1-2 and 5-7 The Hydrogen Atom • Ground state: lowest energy state of an atom • Excited state: atom has higher potential energy than ground state • When an excited atom returns to ground state, it gives off energy it gained in the form of ER. • Production of colored signs (neon) is example Experiment • Pass electrical current through H gas in vacuum tube at low pressure • Emits characteristic pink glow • When the light was passed through prism, separated into series of specific frequencies and therefore wavelength (… equation?) of visible light • These bands are Hydrogen’s line emission spectrum Problem? • Classical theory predicted that H atoms would be excited by whatever amount of energy was added to them. • Expected to observe continuous range of frequencies of ER, or continuous spectrum. • Why had H only given off specific frequencies of light? • Quantum Theory • Excited H atom falls back from excited state to ground state, and emits photon • This energy is equal to difference between initial and final state Implications • Since H atoms emit only specific frequencies, difference between energy states must be fixed. • Therefore, e- of H atom exists only in very specific energy states • In 1913, Bohr proposed a model that linked the atom’s electron with photon emission • Energy is higher in orbits farther from nucleus (like a ladder) • Based on the wavelengths of hydrogen’s line emission spectrum, Bohr calculated energies the e- would have in the allowed energy level for H atom. • Bohr’s calculated values agreed with experimentally observed values for lines in each series • Scientists tried to apply this model to other element’s atoms