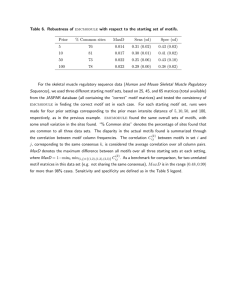
CS 6293 Advanced Topics: Current Bioinformatics Motif finding
advertisement

CS 6293 Advanced Topics:
Current Bioinformatics
Motif finding
HW2
•
•
•
•
Available on course website
First part: theoretical questions. Due on Mon, Oct 25
Second part: Presentation on Mon, Nov 1
Bonus: email me who are in your group and the papers
you choose, by Wed, Oct 20.
• 7 minutes presentation + 2 minutes Q&A
– You can choose to go to some really cool details or
give the main ideas of the papers
• Group size 1 or 2 acceptable, with the same
expectation
• Last slide of presentation
– Contributions from group members
What is a (biological) motif?
• A motif is a recurring fragment, theme or pattern
• Sequence motif: a sequence pattern of nucleotides in a
DNA sequence or amino acids in a protein
• Structural motif: a pattern in a protein structure formed
by the spatial arrangement of amino acids.
• Network motif: patterns that occur in different parts of a
network at frequencies much higher than those found in
randomized network
• Commonality:
– higher frequency than would be expected by chance
– Has, or is conjectured to have, a biological significance
Sequence motif finding
• Given: a set of sequences
• Goal: find sequence motifs that appear in
all or the majority of the sequences, and
are likely associated with some functions
– In DNA: regulatory sequences
• Other names: transcription factor binding sites,
transcription factor binding motifs, cis-regulatory
elements, cis-regulatory motifs, DNA motifs, etc.
– In protein: functional/structural domains
Roadmap
•
•
•
•
Biological background
Representation of motifs
Algorithms for finding motifs
Other issues
– Search for instances of given motifs
– Distinguish functional vs non-functional motifs
Biological background for motif finding
Genome is fixed – Cells are
dynamic
• A genome is static
– (almost) Every cell in our body has a copy of
the same genome
• A cell is dynamic
– Responds to internal/external conditions
– Most cells follow a cell cycle of division
– Cells differentiate during development
Gene regulation
• … is responsible for the dynamic cell
• Gene expression (production of protein) varies
according to:
–
–
–
–
–
Cell type
Cell cycle
External conditions
Location
Etc.
Where gene regulation takes place
• Opening of chromatin
• Transcription
• Translation
• Protein stability
• Protein modifications
Transcriptional Regulation of genes
Transcription Factor (TF)
(Protein)
RNA polymerase
(Protein)
DNA
Promoter
Gene
Transcriptional Regulation of genes
Transcription Factor (TF)
(Protein)
RNA polymerase
(Protein)
DNA
TF binding site, cis-regulatory element
Gene
Transcriptional Regulation of genes
Transcription Factor
(Protein)
RNA polymerase
DNA
TF binding site, cis-regulatory element
Gene
Transcriptional Regulation of genes
New protein
RNA
polymerase
Transcription Factor
DNA
TF binding site, cis-regulatory element
Gene
The Cell as a Regulatory Network
If C then D
gene D
A
B
C
Make D
If B then NOT D
If A and B then D
D
gene B
D
C
Make B
If D then B
Transcription Factors Binding to DNA
Transcriptional regulation:
• Transcription factors
bind to DNA
Binding recognizes
specific DNA
substrings:
• Regulatory motifs
Experimental methods
• DNase footprinting
– Tedious
– Time-consuming
• High-throughput techniques: ChIP-chip, ChIPseq
– Expensive
– Other limitations
Computational methods for finding
cis-regulatory motifs
.
.
.
Given a collection of genes that are believed to be
regulated by the same/similar protein
– Co-expressed genes
– Evolutionarily conserved genes
Find the common TF-binding motif from promoters
Essentially a Multiple Local
Alignment
instance
.
.
.
• Find “best” multiple local alignment
• Multidimensional Dynamic Programming?
– Heuristics must be used
Characteristics of cis-Regulatory
Motifs
• Tiny (6-12bp)
• Intergenic regions are
very long
• Highly Variable
• ~Constant Size
– Because a constant-size
transcription factor binds
• Often repeated
• Often conserved
Motif representation
• Collection of exact words
– {ACGTTAC, ACGCTAC, AGGTGAC, …}
• Consensus sequence (with wild cards)
– {AcGTgTtAC}
– {ASGTKTKAC} S=C/G, K=G/T (IUPAC code)
• Position-specific weight matrices (PWM)
Position-Specific Weight Matrix
1
2
3
4
5
6
7
8
9
A
.97
.10
.02
.03
.10
.01
.05
.85
.03
C
.01
.40
.01
.04
.05
.01
.05
.05
.03
G
.01
.40
.95
.03
.40
.01
.3
.05
.03
T
.01
.10
.02
.90
.45
.97
.6
.05
.91
A
S
G
T
K
T
K
A
C
frequency
Sequence Logo
1
2
3
4
5
6
7
8
9
A
.97
.10
.02
.03
.10
.01
.05
.85
.03
C
.01
.40
.01
.04
.05
.01
.05
.05
.03
G
.01
.40
.95
.03
.40
.01
.3
.05
.03
T
.01
.10
.02
.90
.45
.97
.6
.05
.91
http://weblogo.berkeley.edu/
http://biodev.hgen.pitt.edu/cgi-bin/enologos/enologos.cgi
Sequence Logo
1
2
3
4
5
6
7
8
9
A
.97
.10
.02
.03
.10
.01
.05
.85
.03
C
.01
.40
.01
.04
.05
.01
.05
.05
.03
G
.01
.40
.95
.03
.40
.01
.3
.05
.03
T
.01
.10
.02
.90
.45
.97
.6
.05
.91
http://weblogo.berkeley.edu/
http://biodev.hgen.pitt.edu/cgi-bin/enologos/enologos.cgi
Entropy and information content
• Entropy: a measure of uncertainty
• The entropy of a random variable X that
can assume the n different values x1, x2, . .
. , xn with the respective probabilities p1,
p2, . . . , pn is defined as
Entropy and information content
• Example: A,C,G,T with equal probability
H = 4 * (-0.25 log2 0.25) = log2 4 = 2 bits
Need 2 bits to encode (e.g. 00 = A, 01 = C, 10 = G, 11 = T)
Maximum uncertainty
• 50% A and 50% C:
H = 2 * (-0. 5 log2 0.5) = log2 2 = 1 bit
• 100% A
H = 1 * (-1 log2 1) = 0 bit
Minimum uncertainty
• Information: the opposite of uncertainty
I = maximum uncertainty – entropy
The above examples provide 0, 1, and 2 bits of information,
respectively
Entropy and information content
A
C
G
T
1
2
3
4
5
6
7
8
9
.97
.01
.01
.01
.10
.40
.40
.10
.02
.01
.95
.02
.03
.04
.03
.90
.10
.05
.40
.45
.01
.01
.01
.97
.05
.05
.3
.6
.85
.05
.05
.05
.03
.03
.03
.91
H
I
.24 1.72 .36 .63 1.60 0.24 1.40 0.85 0.58
1.76 0.28 1.64 1.37 0.40 1.76 0.60 1.15 1.42
Mean
1.15
Total
10.4
Expected occurrence in random DNA: 1 / 210.4 = 1 / 1340
Expected occurrence of an exact 5-mer: 1 / 210 = 1 /
Sequence Logo
1
2
3
4
5
6
7
8
9
A
C
.97
.10
.02
.03
.10
.01
.05
.85
.03
.01
.40
.01
.04
.05
.01
.05
.05
.03
G
T
I
.01
.40
.95
.03
.40
.01
.3
.05
.03
.01
.10
.02
.90
.45
.97
.6
.05
.91
1.76 0.28 1.64 1.37 0.40 1.76 0.60 1.15 1.42
Real example
• E. coli. Promoter
• “TATA-Box” ~ 10bp upstream of transcription
start
• TACGAT
• TAAAAT
• TATACT
Consensus: TATAAT
• GATAAT
• TATGAT
Note: none of the instances
• TATGTT
matches the consensus perfectly
Finding Motifs
Motif finding schemes
Conservation
Yes
No
Whole Yes Genome 1 & 2 & 3
Genome
1
Dictionary
building
genome
Phylogenetic footprinting
No Gene 1A & 1B & 1C or
“Motif finding”
Gene
Set 1
Gene Set 1 & 2 & 3
1A
1B
1C
Gene set 1
Gene set 2
Gene set 3
Genome 2
Genome 3
Genome 1
Ideally, all information should be used, at some stage.
i.e., inside algorithm vs pre- or post-processing.
Classification of approaches
• Combinatorial algorithms
– Based on enumeration of words and
computing word similarities
• Probabilistic algorithms
– Construct probabilistic models to distinguish
motifs vs non-motifs
Combinatorial motif finding
• Idea 1: find all k-mers that appeared at least m times
– m may be chosen such that # occurrence is statistically
significant
– Problem: most motifs have divergence. Each variation may only
appear once.
• Idea 2: find all k-mers, considering IUPAC nucleic acid
codes
– e.g. ASGTKTKAC, S = C/G, K = G/T
– Still inflexible
• Idea 3: find k-mers that approximately appeared at least
m times
– i.e. allow some mismatches
Combinatorial motif finding
Given a set of sequences S = {x1, …, xn}
• A motif W is a consensus string w1…wK
• Find motif W* with “best” match to x1, …, xn
Definition of “best”:
d(W, xi) = min hamming dist. between W and a word in xi
d(W, S) = i d(W, xi)
W* = argmin( d(W, S) )
Exhaustive searches
1. Pattern-driven algorithm:
For W = AA…A to TT…T
(4K possibilities)
Find d( W, S )
Report W* = argmin( d(W, S) )
Running time: O( K N 4K )
(where N = i |xi|)
Guaranteed to find the optimal solution.
Exhaustive searches
2. Sample-driven algorithm:
For W = a K-char word in some xi
Find d( W, S )
Report W* = argmin( d( W, S ) )
OR Report a local improvement of W*
Running time: O( K N2 )
Exhaustive searches
• Problem with sample-driven approach:
• If:
– True motif does not occur in data, and
– True motif is “weak”
• Then,
– random strings may score better than any
instance of true motif
Example
• E. coli. Promoter
• “TATA-Box” ~ 10bp upstream of transcription
start
• TACGAT
• TAAAAT
• TATACT
Consensus: TATAAT
• GATAAT
Each instance differs at most 2
• TATGAT
bases from the consensus
• TATGTT
None of the instances matches the
consensus perfectly
Heuristic methods
• Cannot afford exhaustive search on all
patterns
• Sample-driven approaches may miss real
patterns
• However, a real pattern should not differ
too much from its instances in S
• Start from the space of all words in S,
extend to the space with real patterns
Some of the popular tools
• Consensus (Hertz & Stormo, 1999)
• WINNOWER (Pevzner & Sze, 2000)
• MULTIPROFILER (Keich & Pevzner,
2002)
• PROJECTION (Buhler & Tompa, 2001)
• WEEDER (Pavesi et. al. 2001)
• And dozens of others
Consensus
Algorithm:
Cycle 1:
For each word W in S
For each word W’ in S
Create alignment (gap free) of W, W’
Keep the C1 best alignments, A1, …, AC1
ACGGTTG ,
ACGCCTG ,
CGAACTT ,
AGAACTA ,
GGGCTCT …
GGGGTGT …
Algorithm (cont’d):
Cycle i:
For each word W in S
For each alignment Aj from cycle i-1
Create alignment (gap free) of W, Aj
Keep the Ci best alignments A1, …, ACi
• C1, …, Cn are user-defined heuristic constants
Running time:
O(kN2) + O(kN C1) + O(kN C2) + … + O(kN Cn)
= O(kN2 + kNCtotal)
Where Ctotal = i Ci, typically O(nC), where C is a
big constant
Extended sample-driven (ESD)
approaches
• Hybrid between pattern-driven and sample-driven
• Assume each instance does not differ by more than α
bases to the motif ( usually depends on k)
motif
instance
α-neighborhood
The real motif will reside in the neighborhood of some words in S.
Instead of searching all 4K patterns,
we can search the -neighborhood
of every word in S.
Extended sample-driven (ESD)
approaches
• Naïve: N Kα 3α NK
# of patterns to test
# of words in sequences
Better idea
• Using a joint suffix tree, find all patterns
that:
– Have length K
– Appeared in at least m sequences with at
most α mismatches
• Post-processing
WEEDER: algorithm sketch
Current pattern P, |P| < K
# mismatches
(e, B)
Seq occ
A
C
G
T
T
• A list containing all eligible
nodes: with at most α
mismatches to P
• For each node, remember
#mismatches accumulated
(e α ), and a bit vector (B) for
seq occ, e.g. [011100010]
• Bit OR all B’s to get seq
occurrence for P
• Suppose #occ >= m
– Pattern still valid
• Now add a letter
WEEDER: algorithm sketch
Current pattern P
(e, B)
A
C
G
T
T
A
• Simple extension: no branches.
– No change to B
– e may increase by 1 or no
change
– Drop node if e > α
• Branches: replace a node with
its child nodes
– Drop if e > α
– B may change
• Re-do Bit OR using all B’s
• Try a different char if #occ < m
• Report P when |P| = K
WEEDER: complexity
• Can get all patterns in time
O(Nn(K choose α) 3α) ~ O(N nKα 3α).
n: # sequences. Needed for Bit OR.
• Better than O(KN 4K) and O(N Kα 3α NK)
since usually α << K
• Kα 3α may still be expensive for large K
– E.g. K = 20, α = 6
WEEDER: More tricks
Current pattern P
A
C
G
T
T
A
• Eligible nodes: with at most α
mismatches to P
• Eligible nodes: with at most
min(L, α) mismatches to P
– L: current pattern length
– : error ratio
– Require that mismatches to be
somewhat evenly distributed
among positions
• Prune tree at length K
Probabilistic modeling approaches
for motif finding
Probabilistic modeling approaches
• A motif model
– Usually a PWM
– M = (Pij), i = 1..4, j = 1..k, k: motif length
• A background model
– Usually the distribution of base frequencies in
the genome (or other selected subsets of
sequences)
– B = (bi), i = 1..4
• A word can be generated by M or B
Expectation-Maximization
• For any word W,
P(W | M) = PW[1] 1 PW[2] 2…PW[K] K
P(W | B) = bW[1] bW[2] …bW[K]
• Let = P(M), i.e., the probability for any word to
be generated by M.
• Then P(B) = 1 -
• Can compute the posterior probability P(M|W)
and P(B|W)
P(M|W) ~ P(W|M) *
P(B|W) ~ P(W|B) * (1-)
Expectation-Maximization
Initialize:
Randomly assign each word to M or B
• Let Zxy = 1 if position y in sequence x is a motif, and 0
otherwise
• Estimate parameters M, , B
Iterate until converge:
• E-step: Zxy = P(M | X[y..y+k-1]) for all x and y
• M-step: re-estimate M, given Z (B usually fixed)
Expectation-Maximization
position
5
1
Initialize
E-step
probability
1
5
9
9
M-step
• E-step: Zxy = P(M | X[y..y+k-1]) for all x and y
• M-step: re-estimate M, given Z
MEME
•
•
•
•
•
Multiple EM for Motif Elicitation
Bailey and Elkan, UCSD
http://meme.sdsc.edu/
Multiple starting points
Multiple modes: ZOOPS, OOPS, TCM
Gibbs Sampling
• Another very useful technique for
estimating missing parameters
• EM is deterministic
– Often trapped by local optima
• Gibbs sampling: stochastic behavior to
avoid local optima
Gibbs Sampling
Initialize:
Randomly assign each word to M or B
• Let Zxy = 1 if position y in sequence x is a motif, and 0
otherwise
• Estimate parameters M, B,
Iterate:
•
•
•
•
•
Randomly remove a sequence X* from S
Recalculate model parameters using S \ X*
Compute Zx*y for X*
Sample a y* from Zx*y.
Let Zx*y = 1 for y = y* and 0 otherwise
Gibbs Sampling
probability
position
0.2
probability
0.15
0.1
0.05
0
0
2
4
6
8
10
position
12
14
16
18
Sampling
• Gibbs sampling: sample one position according to probability
•
•
– Update prediction of one training sequence at a time
Viterbi: always take the highest
Simultaneously update
EM: take weighted average
predictions of all sequences
20
Better background model
• Repeat DNA can be confused as motif
– Especially low-complexity CACACA… AAAAA, etc.
• Solution: more elaborate background model
– Higher-order Markov model
0th order: B = { pA, pC, pG, pT }
1st order: B = { P(A|A), P(A|C), …, P(T|T) }
…
Kth order: B = { P(X | b1…bK); X, bi{A,C,G,T} }
Has been applied to EM and Gibbs (up to 3rd order)
Gibbs sampling motif finders
• Gibbs Sampler
– First appeared as: Larence et.al. Science 262(5131):208-214.
– Continually developed and updated. webpage
– The newest version: Thompson et. al. Nucleic Acids Res. 35 (s2):W232W237
• AlignACE
– Hughes et al., J. of Mol Bio, 2000 10;296(5):1205-14.
– Allow don’t care positions
– Additional tools to scan motifs on new seqs, and to compare and group
motifs
• BioProspector, X. Liu et. al. PSB 2001 , an improvement of
AlignACE
– Liu, Brutlag and Liu. Pac Symp Biocomput. 2001;:127-38.
– Allow two-block motifs
– Consider higher-order markov models
Limits of Motif Finders
0
???
gene
• Given upstream regions of coregulated genes:
– Increasing length makes motif finding harder –
random motifs clutter the true ones
– Decreasing length makes motif finding harder – true
motif missing in some sequences
Challenging problem
d mutations
n = 20
k
L = 600
• (k, d)-motif challenge problem
• Many algorithms fail at (15, 4)-motif for n = 20 and L = 600
• Combinatorial algorithms usually work better on challenge problem
– However, they are usually designed to find (k, d)-motifs
– Performance in real data varies
(15, 4)-motif
• Information content: 11.7 bits
• ~ 6mers. Expected occurrence 1 per 3k bp
Actual
Results
by MEME
llr = 163
E-value = 3.2e+005
llr = 177
E-value = 1.5e+006
llr = 88
E-value = 2.5e+005
Motif finding in practice
• Where the input come from?
• Possibility 1: microarray studies (later)
• Possibility 2: phylogenetic analysis (not
covered)
• Possibility 3: ChIP-chip
• Possibility 4: ChIP-seq
Chromatin Immunoprecipitation
(ChIP)
• ChIP is a method to
investigate protein-DNA
interaction in vivo.
• The output of ChIP is
enriched fragments of
DNA that were bound by a
particular protein.
• The identity of DNA
fragments need to be
further determined by a
second method.
ChIPSeq Workflow
ChIP
Size Selection
Sequencing
Mapping onto Genome
ChIP-chip
ChIP
Array of intergenic
sequences from the whole
genome
How to make sense of the motifs?
• Each program usually reports a number of motifs
(tens to hundreds)
– Many motifs are variations of each other
– Each program also report some different ones
• Each program has its own way of scoring motifs
–
–
–
–
Best scored motifs often not interesting
AAAAAAAA
ACACACAC
TATATATAT
How to make sense of the motifs?
• Now we’ve found some pretty-looking
motifs
– This is probably the easiest step
• What to do next?
– Are they real?
– How do we find more instances in the rest of
the genome?
– What are their functional meaning?
• Motifs => regulatory networks
How to make sense of the motifs?
• Combine results from different algorithms
usually helpful
– Ones that appeared multiple times are probably more
interesting
• Except simple repeats like AAAAA or ATATATATA
– Cluster motifs into groups.
• Compare with known motifs in database
– TRANSFAC
– JASPAR
– YPD (yeast promoter database)
Strategies to improve results
• How to tell real motifs (functional) from
noises? Statistical test of significance.
– Enrichment in target sequences vs
background sequences
Target set
T
Assumed to contain a
common motif, P
Background set
B
Assumed to not contain P,
or with very low frequency
Ideal case: every sequence in T has P, no sequence in B has P
Statistical test for significance
P
Target set
T
Background set + target set
B+T
N
P appeared in
n sequences
P appeared in
m sequences
• If n / N >> m / M
– P is enriched (over-represented) in T
– Statistical significance?
• If we randomly draw N sequences from (B+T), how
likely we will see at least n sequences having P?
M
Hypergeometric distribution
• A box with M balls (seqs), of which m
are red (with motifs), and the rest are
blue (without motifs).
– Red ball: sequences with motifs
– Blue ball: sequences without motifs
• We randomly draw N balls (seqs) from
the box
• What’s the probability we’ll see n red
balls?
m M m
n N n
hypegeom(n; M , N , m)
M
N
# of choices to have n red balls
Total # of choices to draw N balls
Cumulative hypergeometric test for
motif significance
• We are interested in: if we
randomly pick m balls, how likely
that we’ll see at least n red balls?
cHypegeom (n; M , N , m)
min( m , N )
hypogeom(i; M , N , m)
i n
n 1
1 hypogeom(i; M , N , m)
Null hypothesis: our selection is random.
Alternative hypothesis: our selection favored red balls.
When prob is small, we reject the null hypothesis.
Equivalent: we accept the alternative hypothesis
(The number of red balls is larger than expected).
i 0
m M m
n 1
i
N i
1
M
i 0
N
Example
•
•
•
•
•
•
•
Yeast genome has 6000 genes
Select 50 genes believed to be co-regulated by a common TF
Found a motif from the promoter seqs of these 50 genes
The motif appears in 20 of these 50 genes
In the rest of the genome, 100 genes have this motif
M = 6000, N = 50, m = 100+20 = 120, n = 20
Intuitively:
– m/M = 120/6000=1/50. (1 out 50 genes has the motif)
– N = 50, would expect only 1 gene in the target set to have the motif
– 20-fold enrichment
• P-value = cHyperGeom(20; 6000, 50, 120) = 6 x 10-22
• This motif is significantly enriched in the set of genes
ROC curve for motif significance
• Motif is usually a PWM
• Any word will have a score
–
–
–
–
Typical scoring function: Log (P(W | M) / P(W | B))
W: a word.
M: a PWM.
B: background model
• To determine whether motif M occurred in a sequence, a
cutoff has to be decided
–
–
–
–
Different cutoffs give different # of occurrences
Stringent cutoff: low occurrence in both + and - sequences
Loose cutoff: high occurrence in both + and - sequences
It may be better to look at a range of cutoffs
ROC curve for motif significance
P
Target set
T
N
Given a score cutoff
Appeared in
n sequences
•
•
•
•
•
•
Background set + target set
B+T
Appeared in m
sequences
With different score cutoff, will have different m and n
Assume you want to use P to classify T and B
Sensitivity: n / N
Specificity: (M-N-m+n) / (M-N)
False Positive Rate = 1 – specificity: (m – n) / (M-N)
With decreasing cutoff, sensitivity , FPR
M
ROC curve for motif significance
A good cutoff
Lowest cutoff. Every sequence
has the motif. Sensitivity = 1.
specificity = 0.
sensitivity
1
ROC-AUC: area under curve.
1: the best. 0.5: random.
Motif 1
Motif 2
Random
0
0
1-specificity
Motif 1 is more enriched in motif 2.
1
Highest cutoff. No motif can pass the cutoff. Sensitivity = 0. specificity = 1.
Other strategies
• Cross-validation
– Randomly divide sequences into 10 sets, hold 1 set
for test.
– Do motif finding on 9 sets. Does the motif also appear
in the testing set?
• Phylogenetic conservation information
– Does a motif also appears in the homologous genes
of another species?
– Strongest evidence
– However, will not be able to find species-specific ones
Other strategies
• Finding motif modules
– Will two motifs always appear in the same gene?
• Location preference
– Some motifs appear to be in certain location
• E.g., within 50-150bp upstream to transcription start
– If a detected motif has strong positional bias, may be a sign of its
function
• Evidence from other types of data sources
– Do the genes having the motif always have similar activities
(gene expression levels) across different conditions?
– Interact with the same set of proteins?
– Similar functions?
– etc.
To search for new instances
• Usually many false positives
• Score cutoff is critical
• Can estimate a score cutoff from the “true”
binding sites
Motif finding
Scoring function
Log (P(W | M) / P(W | B))
A set of scores for the “true” sites. Take mean - std as a cutoff.
(or a cutoff such that the majority of “true” sites can be predicted).
To search for new instances
• Use other information, such as positional biases
of motifs to restrict the regions that a motif may
appear
• Use gene expression data to help: the genes
having the true motif should have similar
activities
– Risk of circular reasoning: most likely this is how you
get the initial sequences to do motif finding
• Phylogenetic conservation is the key
References
• D’haeseleer P (2006) What are DNA sequence motifs?
NATURE BIOTECHNOLOGY, 24 (4):423-425
• D’haeseleer P (2006) How does DNA sequence motif
discovery work? NATURE BIOTECHNOLOGY, 24
(8):959-961
• MacIsaac KD, Fraenkel E (2006) Practical strategies for
discovering regulatory DNA sequence motifs. PLoS
Comput Biol 2(4): e36
• Lawrence CE et. al. (1993) Detecting Subtle Sequence
Signals: A Gibbs Sampling Strategy for Multiple
Alignment, Science, 262(5131):208-214