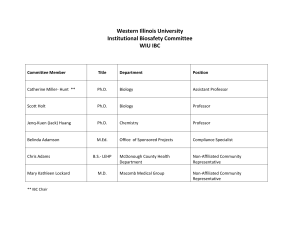
IBC ) TITLE AND PERSONNEL
advertisement

For IBC Use Only Protocol Number: Received: Approved: IBC (INSTITUTIONAL BIOSAFETY COMMITTEE) PROTOCOL SUBMISSION FORM IBC Office, 1430 Tulane Ave. TW-5, New Orleans, LA 70112 504.988.0300 (ph) 504.988.0370 (fax) Submit the completed application by email to ibc@tulane.edu with “IBC-Application” in the subject line. TITLE AND PERSONNEL Title of the study: Enhancing hormone therapy and chemo-therapy of human prostate cancer and breast cancer Submission Date: Is this a new submission? YES Principal investigator: Yan Dong Department: Structural and Cellular Biology Position/title: Associate Professor School: Shool of Medicine Email: ydong@tulane.edu Phone: 504-988-4761 Co-investigator’s name: Title: Emergency phone: Email: Phone: TYPE OF RESEARCH The research described in this application involves: (select all that apply) 1. Recombinant DNA/RNA YES If “YES” complete Sections A, B, E and F of this application NO 2. Infectious/Pathogenic microorganisms or toxins YES If “YES” complete Sections A , C, E and F of this application NO 3. Select Agents (see appendix 1) YES If “YES” complete Sections A, C, D, E and F of this application 4. Animals YES NO NO If “YES” provide IACUC approval number:and 4154 5. Humans YES NO If “YES” provide IRB approval number: IBC Protocol Form- Jan/2010 Page 1 of 14 SECTION A: GENERAL INFORMATION PERFORMANCE SITES Research location(s)--check all that apply: Tulane University St. Charles Campus Tulane University School of Medicine Tulane University School of Public Health and Tropical Medicine Tulane National Primate Research Center Other location(s). Specify : Building and Room Number(s) where research will be conducted (provide buildings/room numbers for all experiments detailed in this application), and biocontainment level of that room. Biocontainment Building: Room No: level for this Experiment (ie. in vitro work, animal infection) laboratory* JBJ 472 JBJ JBJ 462 Vivarium BSL2 BSL2 BSL2-N (animals) in vitro work in vitro work in vivo animal xenograft Does this project involve work to be done by collaborators at other institutions? Name of the institution(s): If “Yes”, this institution granted approvals for: IBC IACUC IRB Don’t know Yes No Yes No SECTION A PROJECT OVERVIEW Provide a description of the work to be conducted in this project. Briefly explain in language understandable to a layperson the aim of this research and its importance to human or animal health, the advancement of knowledge, or the benefit to society. Succinctly describe in chronological order the experiments that will be conducted. Our aim is to develop agents with low toxicity that can enhance the efficacy of hormone therapy and/or chemo-therapy in human prostate and breast cancer. We plan to investigate the ability and mechanism of these agants in enhancing the efficacy of current treatment methods. The study could lead to the development of an effective, mechanism-driven strategy to significantly improve the therapeutic outcome and ultimately to reduce prostate/breast cancer mortality. IBC Protocol Form- Jan/2010 Page 2 of 14 SECTION B Recombinant DNA Research Questionnaire. (Check 'Yes' or 'No' to the following questions (refer to the NIH Guidelines* for more information). The relevant section of the NIH Guidelines is referenced for each question. For any items checked 'YES', include a thorough description of the work in the following page (question 1). 1. Does your project include deliberate transfer of a drug resistance trait to microorganisms that are not known to acquire the trait naturally (Section IIIA*)? 1a. If “YES”, could such a transfer compromise the use of the drug to control disease agents in humans, veterinary medicine, or agriculture? 2. Does your project include cloning toxin molecules with an LD50 of less than 100 nanograms per kilogram body weight (Section III-B*)? 3. Does your project include experiments involving the deliberate transfer of recombinant DNA, or DNA or RNA derived from recombinant DNA, into one or more human research participants (Section III-C*)? 4. Does your project include experiments using Risk Group 2, Risk Group 3, Risk Group 4, or Select Agents as host-vector systems (Section III-D-1*)? 5. Does your project include experiments in which DNA from Risk Group 2, Risk Group 3, Risk Group 4 , or Restricted Agents is cloned into nonpathogenic prokaryotic or lower eukaryotic host-vector systems (Section III-D-2*)? 6. Does your project include experiments involving the use of infectious DNA or RNA viruses or defective DNA or RNA viruses in the presence of helper virus in tissue culture systems (Section III-D-3*)? 7. Does your project include experiments involving whole animals in which the animal’s genome has been altered by introduction of DNA into the germ line (i.e. transgenic animals) (Section III-D-4, III-E-3*) 8. Does your project include experiments involving viable rDNA-modified microorganisms tested on animals (Section III-D-4, III-E-3*)? 9. Does your project include experiments involving genetically engineered whole plants (Section III-D-5, III-E-2*)? 10. Does your project include experiments involving more than 10 liters of culture (Section III-D-6*)? 11. Does your project include experiments involving the formation of recombinant DNA molecules containing two-thirds or more of the genome of any eukaryotic virus (Section III-E-1*)? Yes No Yes No Yes No Yes No Yes No Yes No Yes No Yes No Yes No Yes No Yes No Yes No * The NIH Guidelines are found in this link: http://oba.od.nih.gov/rdna/nih_guidelines_oba.html Updated information about required levels of Biosafety are found in the CDC/NIH Biosafety in Microbiological and Biomedical Laboratories (BMBL) http://www.cdc.gov/OD/ohs/biosfty/bmbl5/bmbl5toc.htm IBC Protocol Form- Jan/2010 Page 3 of 14 IBC Protocol Form- Jan/2010 Page 4 of 14 SECTION B Recombinant DNA Research Questions 1. Address any items checked ‘Yes’ in the Recombinant DNA Research Questionnaire (include the question number). Question 1: To develop stable transfectants of aromatase expressing MCF-7 cells, we need to transfect a plasmid containing zeocin resistant cassette to be used as a selective marker. Qusetion 6: We need to use retroviral and lentiviral vectors for our eukaryotic infection to obtain the high efficiency of transient transfection. 2. Describe any use of animals or human subjects. The approval of your protocol will not be effective until required IACUC and/or IRB approvals have been received. Groups of Balb/c male or female mice will be inoculated with ~5x10^6 human prostate cancer cells or breast cancer cells to setup xenograft model. At the end point, the mice will be euthanized and blood and tissue samples will be harvested for further analysis. 3. Identify the host(s) to be used (e.g., the target of gene transfer). Examples: E. coli, S. cerevisiae, human/animal cells, whole animals, humans. Provide species designations for all organisms where possible. The host used for prokaryotic transformation is E. coli. The hosts used for eukaryotic transfection are human prostate cancer cell lines and breast cancer cell lines. 4. Identify the vector(s) to be used. Examples: Bacterial plasmids, yeast vectors, mammalian cell vectors, baculoviruses, transforming viruses, etc. Provide name and description of the vector, including antibiotic resistance genes. The vectors to be used are: 1) pCI-Neo from Promega with ampicilin resistance; 2) pGL4.19 from Promega with ampicilin resistance; 5. Identify the nature of the insert DNA sequence, including the species of origin (i.e., specific gene, promoter, expressed product and function (if known). If available attach a vector and/or insert map. The insert DNA sequences include: 1) full length of human hTERT gene; 2) hTERT promoter (4kb) region; 6. If foreign gene product(s) will be purified, indicate which foreign gene product will be purified and briefly describe the procedures for purification, including volumes of culture N/A IBC Protocol Form- Jan/2010 Page 5 of 14 7. If replication-incompetent vectors will be used, include information about how incompetent vectors are tested for reversion mutations (e.g., endpoint dilution analysis, plaque assay). N/A IBC Protocol Form- Jan/2010 Page 6 of 14 SECTION C Hazardous Biological Research Questionnaire Check 'YES' or 'NO' to the following questions. For any items checked 'YES', include a thorough description of the work in the following page (question 1). 1. Is agent/material a potential human, animal or plant pathogen? Yes Human Animal Plant N/A 2. Is agent/material a toxin? If “YES” name of the toxin (s): Toxic to: LD50 : 3. Do you work with quantities (i.e. cultures) larger than 1 liter? If so, what is the largest volume (liters)? 4. Do you inactivate the agent/material prior to laboratory manipulation? If “yes”, by what method? Heat Chemical Radiation Other Yes No No Yes No Yes No Yes No Yes No Yes No Yes No 9. Does your work involve a SELECT AGENT? (see appendix H) If “YES” name the organism (s): Yes No 10. Do you work with human blood, tissues, or body fluids? If “yes” contact Tulane OEHS for guidelines on working with blood-borne pathogens Yes No 5. Do you concentrate the agent/material? If Yes, specify method: Centrifugation Precipitation Filtration Other 6. Do you expose live animals or humans to the agent/material? If Yes, specify the Species: IACUC Approval # IRB Approval:# 7. Does your project include experiments with Risk Group 2, Risk Group 3 or, Risk Group 4 agents? * If yes, which Risk Group? RG-2 RG-3 RG-4 8. Does your experiment involve work with arthropods (i.e. insects, spiders. others)? If yes, provide specific details in the next page (question 1) Work with certain arthropods must conform to the guidelines set forth in the BMBL and those established by the American Committee of Medical Entomology 11. What is the recommended Biosafety Level for this agent/material? * BSL-1 BSL-2 BSL-3 BSL-4 *To identify the risk group (RG) classification of the recombinant or etiologic agent(s) and the proposed biosafety level (See the NIH Guidelines at http://oba.od.nih.gov/rdna/nih_guidelines_oba.html And the CDC/NIH Biosafety in Microbiological and Biomedical Laboratories at: http://www.cdc.gov/OD/ohs/biosfty/bmbl5/bmbl5toc.htm IBC Protocol Form- Jan/2010 Page 7 of 14 SECTION C Hazardous Biological Research Questions 1. Address any item checked ‘YES’ in the Hazardous Biological Research Questionnaire (include the question number). 2. Describe the key features of the agent/microorganism/toxin or material that you will use in this project, particularly as it refers to biosafety considerations. Briefly describe the pathogenesis of the disease caused by this agent. 3. What is the source or your biohazardous agent? (Be specific: i.e. ATCC, CDC, clinical isolate, etc). 4. Do you work with microorganisms of unknown identity (such as those obtained from soil or other environmental samples). 5. Does import of this agent into Tulane facilities require USDA/CDC import/transport permit? 6. Will the agent be cultured/propagated at Tulane? If yes provide a synopsis of the procedures. 7. Will your work be performed in a BSL3 laboratory? If yes detail the procedures to be done in the BSL3. 8. Describe any specific biosafety considerations for this agent that have not been addressed in the previous questions. IBC Protocol Form- Jan/2010 Page 8 of 14 Section D Select Agent Questionnaire No work with Select Agents can be initiated until IBC approval is obtained IBC protocols involving Select Agents will be subjected to yearly review. 1. Is there a vaccine available and recommended for persons handling this Select Agent? If “yes” will personnel working in this project be offered the vaccine? Expand the answer to this question under question 1 in the next page Yes No Yes No 2. Do you plan on shipping, or transporting the select agent? If “Yes” be aware that a Select Agent must be transported under the conditions described in specific Code of Federal Regulations (CFRs). Consult the Office of Biosafety for guidelines. Yes No 3. If you are working with a toxin, indicate the largest amount that you will have in your possession at any given time. mgs 4. What is the largest volume of culture (bacterial/viral) and approximate concentration that you will have at any given time? (i.e. 1 liter at 10x106 CFU/ml). 5. Will access to the Select Agent be meticulously controlled at all times? Yes No 6. Have you submitted an agent-specific SOP with this application? (This is a requirement for approval of your protocol). Yes No 7. Does your SOP include specific instructions to handle accidental spills and accidental personnel exposures? Yes No Yes No 9. Will a copy of the training certification and assessment evaluation for each person working with the agent be filed with the Biosafety Office? Yes No 10. Are you and all the personnel involved in this project proficient in the general BSL3 SOPs written by the Biosafety Office? Yes No 8. Will personnel involved in this project receive agent-specific training, to make them aware of the risk and hazards associated with this agent, and the SOPs for accidental spills or exposures? Who will provide the agent-specific training? IBC Protocol Form- Jan/2010 Page 9 of 14 Section D: Select Agent Questions 1. Provide relevant and detailed information to expand the answers to the above questionnaire (provide item number) 2. What General SOPs (as provided by the Tulane Biosafety Office) will be used in the course of these experiments? See Appendix 2 and check all that apply 3. If working with animals indicate the final fate of all animals exposed to the select agent IBC Protocol Form- Jan/2010 Page 10 of 14 SECTION E Biohazards, Decontamination and Training 1. Identify all known and potential hazards or risks associated with the specific biohazardous agent or with the use of recombinant DNA materials described in this application (i.e. generation of aerosols, sharps punctures, animal bites, and scratches, etc.) Biosafety risks in this project (although minimal) are as follows: - Risk of wounds/punctures with contaminated sharps, - Contact with human cancer cells, - Contact with animals tissues. blood or animal waste. - 2. Describe the necessary facilities/equipment/practices that will be used for all aspects of the work (for the indicated safety/containment level). (i.e. use of biosafety cabinets , aerosol proof centrifuge, aerosolization chambers, etc) Cell cultures will be done in biosafty cabinets; Animal works will be done in the vivarium in biosafety cabinets; BSL2 guidelines will be strictly followed 3. Indicate the personal protective equipment or PPE (gloves, mask, lab coat, PAPR, etc) required for personnel working in this project. Personnel working with this project will wear lab coats, gloves for routine work. 4. Specifically describe the decontamination practices to be used. (Lysol, bleach, autoclave, etc.). Provide detailed description of decontamination for materials/samples that will be transported out of the laboratory. All surfaces will be decontaminated with 70% alcohol before and after all procedures Glassware will be autoclaved Disposable consumables will be placed in properly labelled bioharzard box Sharps will be placed in a "sharp containers" 5. Describe the disposal practices of contaminated material, contaminated tissue or animal carcasses. Include practices for disposal of liquid and solid waste, including sharps. Disposable waste will be placed in properly labelled bioharzard box Sharps will be placed in a "sharp containers" 6. For Risk Group 3 agents and for agents that require BSL3 containment, briefly describe your prior training or experience in this type of work. N/A 7. Describe how personnel have been (or will be) trained in the handling of the biological agents described in this application. Who is in charge of training? (The PI is responsible for keeping accurate records of personnel training) IBC Protocol Form- Jan/2010 Page 11 of 14 The PI (Dr. Yan Dong) will train all personnel working in this project. Personel will be instructed on the risks of working with animals, human cancer cells and with recombinant DNA materials. Personnel will be trained to follow BSL2 guidelines and wear the required PPE at all times while doing experiments. 8. If your experiments involve animals, do animal handlers need to be aware of specific precautions or need to implement changes to the routine animal care? No changes to routine veterinary care are necessary. IBC Protocol Form- Jan/2010 Page 12 of 14 SECTION F: Investigator’s Assurance 1. I confirm that all persons conducting this work at Tulane University (including students, fellows, technicians, and collaborators) have been adequately trained in good microbiological techniques; have received instruction on the specific hazards associated with the work and are aware of the specific safety equipment, practices, and behaviors required during the course of the work and use of these facilities. 2. I will report to the Biological Safety Officer immediately any spill of biohazardous material, any equipment or facility failure (e.g., ventilation failure), and/or any breakdown in procedure that could result in potential exposure of laboratory personnel and/or the public to biohazardous material. 3. I confirm that any proposed changes to my work that would result in an increased level of biohazard will be reported to the IBC before the change is implemented. 4. I confirm that no work requiring IBC approval will be initiated or modified until approval is received. 5. I have read and understand my responsibilities as Principal Investigator outlined in Section IV-B-7 of the NIH Guidelines and the Tulane University Institutional Biosafety Committee (IBC) Policy Manual for the Use of Recombinant DNA, and agree to comply with these responsibilities. 6. I certify that the information provided within this application is accurate to the best of my knowledge. I also understand that, should I use the project described in this application as a basis for a funding proposal (either intramural or extramural), it is my responsibility to ensure that the description of the work in the funding proposal is identical in principle to that contained in this application. Yan Dong Typed Name of Principal Investigator Signature of Principal Investigator Date UPON APPROVAL YOU MUST SEND A SIGNED COPY OF THIS LAST PAGE TO THE IBC OFFICE (by Campus mail Box TW-5, Fax 8-0370, or email (scanned copy) to ibc@tulane.edu) For IBC Use Only Protocol Number: Protocol Title: Experiment Class Determination (check one) III-A Requires IBC approval, RAC review, and NIH Director approval before initiation III-B Requires NIH/OBA and IBC approval before initiation III-C Requires IBC and IRB approvals and RAC review before research participant enrollment III-D Requires IBC approval before initiation III-E Requires IBC notice simultaneous with initiation III-F Exempt IBC Protocol Form- Jan/2010 Page 13 of 14 IBC Signature: Date: IBC Protocol Form- Jan/2010 Page 14 of 14