EXPERIMENT 3.3 WATER HARDNESS TITRATION
advertisement

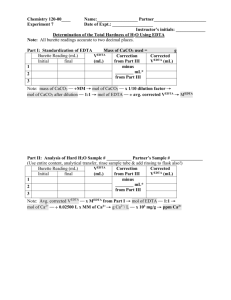
EXPERIMENT 3.3 WATER HARDNESS TITRATION INTRODUCTION One of the important contrasts between the alkali metals and alkaline earth metals is the difference in the degree of solubility of the metal ions with respect to a variety of anions. Given a metal from each group on the same row of the periodic table, the higher charge density on the alkaline earth metal cation compared to the alkali metal cation tends to allow the former to make stronger bonds with anions. This is illustrated by the observation that nearly all ionic compounds containing alkali metals are highly soluble in water whereas many alkaline earth salts are insoluble or only slightly soluble. Most sources of “fresh” water contain some concentration of alkaline earth cations, mostly Ca2+ and Mg2+, that come from the slight solubility of these ions in the rocks, soils, and minerals that are in contact with the water. It is a particular nuisance that with variations in concentrations or in pH these ions may form precipitates with anions already present in the water (such as hydroxide and carbonate) or ions added during use of the water (such as phosphates or the fatty acid anions in soaps). Bathtub ring and scale in water system pipes are examples of this type of precipitation. Water with a high concentration of Ca2+ and /or Mg2+ is called “hard” due to its potential to produce solid precipitates. Water with low concentrations is called “soft” water. A common measure of water hardness is obtained by determining the combined concentration of these metal ions and imagining that it is all Ca2+ from dissolved CaCO3. The concentration is expressed in milligrams of dissolved CaCO3 per liter of water sample. Since one liter of water has a mass of about one kilogram and a milligram is one millionth of a kilogram, concentrations in milligram per liter of water are often referred to as parts-per-million or ppm. Water hardness can vary considerably from one water system to another. Values from tens to hundreds of ppm are common in the continental United States. In this experiment, you will measure the hardness of a sample of water. In order to make this measurement by a titration, we need a reagent that will ignore alkali metal ions but react with Ca2+ and Mg2+. One such reagent is EDTA (Ethylene Diamine Tetraacetic Acid). Its structure will be described in a text chapter on transition metals. For now, consider that it is a tetraprotic acid and that in a basic solution, its anions tend to bond to many metal cations (but not alkali metal ions) in a one-to-one ratio. Metal ions bound to EDTA are less available to form precipitates with other anions so EDTA is used as a water softener in many shampoos and in other applications. We will use EDTA to measure the concentration of Ca2+ and Mg2+ (and any other strong bonding cations) by adding it to water samples from a buret until the metal ions are all bound. As an indicator, we will use a dye known as Eriochrome Black T. Like EDTA it tends to bond to metals, however it bonds less strongly and to fewer of them (e.g., Mg2+ but not Ca2+). Eriochrome Black T is a deep blue green by itself. If it is added to a sample containing at least some Mg2+, it turns wine red as they become bound together. This will be the initial sample color during a titration. As EDTA is slowly added to a sample, it will begin tying up the metal ions. As long as there are excess metal ions, the indicator can find them and assume the wine red color. As the EDTA amount becomes equal to the metal ion amount, it will displace the indicator from the last of the Mg2+. The indicator will then return to its blue green color. This marks the endpoint. As mentioned earlier, the water hardness measure treats the bonding metal ion content as if it were all Ca2+. So we can treat the fundamental relationship in the titration as moles EDTA = moles Ca2+ Calculation of the moles of EDTA requires knowledge of its volume and molar concentra-tion. Volumes will be measured with a buret. The concentration will be found by titration against a standard solution of known Ca2+ equivalent concentration that will be provided. MATERIALS AND SAFETY The standard and unknown solutions used do not contain corrosive substances. However, the pH 10 buffer is considerably basic and constitutes an eye hazard. Wear safety goggles at all times as usual. The Eriochrome Black T indicator is not particularly toxic, but it will tend to stain. Handle it with care and clean up spills promptly. All solutions used and produced in the experiment can be disposed of in the sinks when finished. PROCEDURE PART A: STANDARDIZATION Obtain a buret and a buret clamp from the equipment cart. Treat the burets with great care, especially the fragile tips. Rinse the buret with distilled water, then secure it to a vertical stand with the clamp. Assign one of your beakers to collect rinsings. Use distilled water to verify that the stopcock functions properly with no leaks or air bubbles. Use a small beaker to transfer EDTA solution from the reservoir to the buret. Take EDTA only as you need it to avoid wasting excess. Rinse the buret with very small portions of EDTA before filling. To find the molarity of the EDTA it will be used to titrate a known concentration of CaCO3. Use a 50 mL beaker to obtain about 40 mL of the CaCO3 standard solution from the cart. Use a transfer pipet to deliver 10.00 mL of the standard into a clean 125 mL or 250 mL Erlenmeyer flask. Use a graduated pipet to add 5 mL of pH 10 buffer (The instructor may have set out a beaker and pipet for the entire class to use with the buffer). Next add 3-5 drops of the Eriochrome Black T indicator. The standard solution has been laced with Mg2+ so the solution should appear wine red. Record the initial liquid level in the buret. All readings should be recorded to the nearest 0.01 mL. Add EDTA to the flask until the indicator changes from wine red to blue green. Some may find this endpoint difficult to see at first. On the verge of the endpoint the color is a somewhat complex mixture. Focus on the elimination of traces of redness in it. It may help to compare the color to an untitrated (next) sample and to saved, previously titrated samples. Record the final buret volume. Repeat the process for a second and third trial. Refill the buret between trials. Each partner should do at least one standard titration. Try to perform operations consistently to enhance precision but do not be overly concerned if it is not high. Several factors including the low concentrations and interference from the buffer work against obtaining a sharp endpoint. PART B: UNKNOWN ANALYSIS In this experiment, you are usually given the option to use your own untreated water sample or to draw from a reservoir of lab tap water. Indicate the source of your sample (e.g., what city or water district or “lab water”) in the report section. Due to the range of possible metal ion concentrations in the samples, choosing a good sample size may require a trial-and-error approach. Ideally, a sample would require 20-40 mL of EDTA to titrate. Start with a 200 or 250 mL sample for the first trial, then adjust the sample size if necessary for the remaining trials. Be sure to record the sample volume you use in each case. Before taking a sample, be sure that the source is well-mixed by shaking or swirling. Your instructor will indicate whether to use repeated transfers with a 50 mL pipet or with your 50 mL graduated cylinder to deliver the sample volume into your 500 mL Erlenmeyer flask. Add buffer and indicator as with the standard. If at this stage, the sample solution does not give the wine red color, consult your instructor. You will either be directed to choose an alternative sample source or be instructed in a method to add an Mg2+ spike and perform a spike-plus-blank titration to correct for it. Assuming your sample did become wine red, titrate with EDTA as with the standard recording the buret levels. If your group has one sample source, perform at least three titrations with it. If two partners wish to analyze samples from two different sources, perform two titrations on each. Then each partner should determine the average Ca2+ molarity for their source only and record it under item (o) of the calculations section of the report. When finished, dispose of all solutions in the sink and rinse all glassware. Carefully return the burets and pipets to the cart along with the buret clamps. PRELAB QUESTIONS 1. A 10.00 mL aliquot of 0.010 M CaCO3 is titrated with 18.22 mL of EDTA solution. What is the EDTA molarity? 2. A 250.0 mL water sample requires 30.85 mL of the EDTA solution from question 1 to reach the Eriochrome Back T endpoint. What was the molarity of the hard metal ions in the water sample? 3. If the metal ions in the water sample of question 2 are assumed to be Ca2+ from CaCO3, express the concentration in ppm CaCO3. This is the same as the concentration in units of mg CaCO3 per liter of sample. PART A (Create the following blank Tables in your lab notebook as part of prelab) DATA AND POSTLAB CALCULATIONS a) Molarity of standard Ca2+ solution______________ TRIAL b) Initial EDTA level 1 2 3 ___________ ___________ ___________ c) Final EDTA level ___________ ___________ ___________ d) EDTA volume delivered ___________ ___________ ___________ e) Qualitative comments TRIAL f) EDTA molarity 1 2 3 ___________ ___________ ___________ g) Average EDTA molarity ± average deviation ___________ ± ___________ PART B (Create the following blank Tables in your lab notebook as part of prelab) DATA AND POSTLAB CALCULATIONS h) Source of water sample____________________________________ i) TRIAL Water sample volume 1 2 3 4 ___________ ___________ ___________ ___________ j) Initial EDTA level ___________ ___________ ___________ ___________ k) Final EDTA level ___________ ___________ ___________ ___________ l) EDTA volume delivered ___________ ___________ ___________ ___________ (Corrected EDTA volume) ___________ ___________ ___________ ___________ m) Qualitative comments TRIAL n) Sample molarity as Ca2+ 1 2 3 4 ___________ ___________ ___________ ___________ o) Average Ca2+ molarity ± average deviation ___________ ± ___________ CONCLUSION Assume sample water “hardness” was all Ca2+ from CaCO3. p) Average mass, in grams, of CaCO3 per liter ___________ g/L. q) Parts per million CaCO3 (i. e., mg CaCO3/L) ___________ ppm.