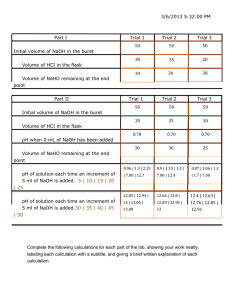
Ka , Dissociation Constant of a Weak Acid Objective : Prelab Questions:
advertisement

Ka , Dissociation Constant of a Weak Acid Objective : Determine Ka for an unknown weak acid using pH measurements. Prelab Questions: 10.00mL of 1.000M HA (a weak acid) was mixed with 5.00mL of 0.500M NaOH. Then the mixture was diluted to 50.0.mL with distilled water. The pH of the solution read 5.00. (PL1) What is the chemical equation between HA and NaOH. (PL2) What is the limiting reactant? (PL3) What is [HA] and [OH-] left at the end of this reaction? (PL4) Write a balanced equation for dissociation of HA in water. (PL5) Write an expression for Ka for HA. (PL6) Draw the appropriate ICE table. Fill in all the information. Hint : Calculate [H3O+] at equilibrium from pH. (PL7) Calculate Ka. Procedure : (1) Fill the first buret with ~0.200M unknown acid. (2) Fill the second buret with ~0.100M NaOH solution. (3) Prepare 4 solutions in large test tubes by mixing Solution # Volume of ~0.200M Volume of ~0.100M Unknown acid (mL) NaOH (mL) 1 5.00 2.50 2 5.00 5.00 3 5.00 7.50 4 5.00 10.00 (4) Rinse out one of the buret and fill it with water. Add various mL of water to all 4 solutions to make the total volume to be 15.00mL in each test tube. Mix well. (5) Measure the pH of solutions 1 to 4. Calculations : (C1) Write the chemical equation (one that would lead to the Ka) and Ka expression. (C2) Draw 4 ICE tables with all the information filled in. (C3) Calculate Ka for each solution. (C4) Calculate average Ka. Postlab Questions : (Q1) What would happen to the final answer if you forgot to add water to test tube #1 but used 15.00mL as your total volume in your calculation? (Q2) The calculations are done assuming that OH- added is reacting completely. What would happen to the final result if not all OH- was used up? Hint: WHAT is the final result? What would happen to the VALUE? Why? Summary : Unknown number Re-draw I.C.E. table for solution #1 Average Ka