The Chemistry of Life
advertisement

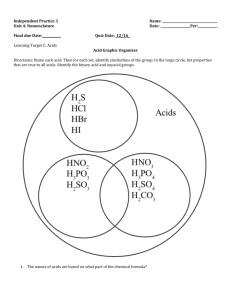
The Chemistry of Life v Elements and the Periodic Table v Atomic Structure v Shells, Orbitals, and the Octet Rule v Bonding between atoms • Covalent: non-polar and polar • Ionic bonds • Hydrogen bonds v Chemical Reactions v Acids, Bases, and pH v Functional Groups v Carbohydrates: Monosaccharides and Polysaccharides v Lipids: Fatty Acids and Complex Lipids v Proteins: Amino Acids and Polypeptides v Nucleic Acids: Nucleotides and RNA/DNA Periodic Table of the Elements C - Carbon Fe - Iron (from ferrum) He - Helium Al - Aluminum Au - Gold (from aurum) Cu - copper (from cuprum) Table 2.1 The Chemistry of Life v Elements and the Periodic Table v Atomic Structure v Shells, Orbitals, and the Octet Rule v Bonding between atoms • Covalent: non-polar and polar • Ionic bonds • Hydrogen bonds v Chemical Reactions v Acids, Bases, and pH v Functional Groups v Carbohydrates: Monosaccharides and Polysaccharides v Lipids: Fatty Acids and Complex Lipids v Proteins: Amino Acids and Polypeptides v Nucleic Acids: Nucleotides and RNA/DNA Atoms Are the Apples in the Bins Called Elements • Atoms (apples) are the individual items belonging to a specific element (variety) in the Periodic Table. • Atoms belonging to an element are very similar but not identical (they have slightly different masses) Three atoms (apples) from the variety (element) Golden Delicious 24.2 g Periodic Table of Apple Varieties 29.6 g 25.9 g The Study of Atoms Electrically neutral atoms have equal numbers of protons and electrons Figure 2.1 Different Shells (Orbits) Have Different Capacities For Electrons Atoms whose outer shells are not full are more reactive: they seek to fill their outermost shells (Octet Rule) The Chemistry of Life v Elements and the Periodic Table v Atomic Structure v Shells, Orbitals, and the Octet Rule v Bonding between atoms • Covalent: non-polar and polar • Ionic bonds • Hydrogen bonds v Chemical Reactions v Acids, Bases, and pH v Functional Groups v Carbohydrates: Monosaccharides and Polysaccharides v Lipids: Fatty Acids and Complex Lipids v Proteins: Amino Acids and Polypeptides v Nucleic Acids: Nucleotides and RNA/DNA Three Types of Chemical Bonds Ionic Covalent Hydrogen Chemical Reactions 530 kcal Categories of Reactions Synthesis Reactions A + B AB Decomposition Reactions AB A + B Exchange Reactions NaOH + HCl NaCl Reversible Reactions A + B AB + H2O Dissociation of Salts and Solubility hydrophilic hydrophobic Figure 2.5 The Chemistry of Life v Elements and the Periodic Table v Atomic Structure v Shells, Orbitals, and the Octet Rule v Bonding between atoms • Covalent: non-polar and polar • Ionic bonds • Hydrogen bonds v Chemical Reactions v Acids, Bases, and pH v Functional Groups v Carbohydrates: Monosaccharides and Polysaccharides v Lipids: Fatty Acids and Complex Lipids v Proteins: Amino Acids and Polypeptides v Nucleic Acids: Nucleotides and RNA/DNA Acids, Bases, and Salts Figure 2.6a Acid-Base Balance: The pH Scale H2O water H+ + OHhydrogen ion hydroxide ion The Chemistry of Life v Elements and the Periodic Table v Atomic Structure v Shells, Orbitals, and the Octet Rule v Bonding between atoms • Covalent: non-polar and polar • Ionic bonds • Hydrogen bonds v Chemical Reactions v Acids, Bases, and pH v Functional Groups v Carbohydrates: Monosaccharides and Polysaccharides v Lipids: Fatty Acids and Complex Lipids v Proteins: Amino Acids and Polypeptides v Nucleic Acids: Nucleotides and RNA/DNA Table 2.3.1 Large Biological Molecules Are Polymers of Monomers HO HO polymer OH OH + HOH Dehydration/condensation monomers HO + HOH HO OH Hydrolysis O O + HOH OH + HOH The Four Classes of Biological Molecules –Carbohydrates –Lipids –Proteins –Nucleic acids