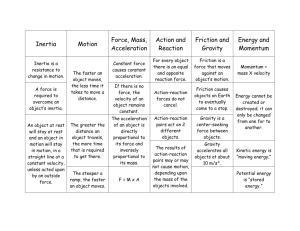
Final Exam Practice
advertisement

Final Exam Practice Multiple Choice Identify the choice that best completes the statement or answers the question. ____ 1. Which of the following is NOT a unit of measurement in the metric system? a. Meter b. Second c. Centimeter d. Foot ____ 2. How many centimeters are in a stick 3.0 meters long? a. 0.3 cm b. 30 cm c. 300 cm d. 3,000 cm Figure 1-1A ____ 3. What is the volume of liquid in the graduated cylinder shown in Figure 1-1A? a. 8 mL b. 18 mL c. 18.5 mL d. 19 mL ____ 4. How many significant digits are in the measurement 14,433 feet? a. 3 b. 4 c. 5 d. 6 ____ 5. A meniscus: a. is formed when a liquid is in a tube. b. makes volume measurements impossible. c. always curves downward. d. All of the above ____ 6. A kilogram represents a unit of: a. volume. b. mass. c. time. d. weight. ____ 7. How does the density of a pure silver teapot compare to a pure silver bracelet? a. b. c. d. ____ It is higher. It is lower It is the same. You can’t tell without the numbers. 8. Material Wood Glass Aluminum Iron Density 0.9 g/cm3 2.3 g/cm3 2.7 g/cm3 7.8 g/cm3 A block has a mass of 23 grams and a volume of 10 cm3. Using the table above, what material is the block made of? a. Wood b. Glass c. Aluminum d. Iron ____ 9. A graph is: a. a picture that shows how two variables are related. b. drawn with the independent variable on the horizontal axis. c. a mathematical model that helps you interpret the data you collect. d. All of the above ____ 10. In a direct relationship: a. as one variable increases, so does the other. b. when one variable increases, the other decreases. c. there is no change in data. d. variables change randomly. ____ 11. Which of the following hypotheses could be tested by an experiment? a. A chocolate bar tastes better than potato chips. b. The harder you push a small wagon, the faster it goes. c. Aliens control everything that happens on Earth. d. All of the above ____ 12. Speed that does not change is called: a. average speed. b. constant speed. c. instant speed. d. minimum speed. ____ 13. In 1/2 hour, a bicyclist traveled 20 kilometers. What was the bicyclist’s average speed? a. 60 km b. 10 km/h c. 40 km d. 40 km/h ____ 14. On this graph, the dependent variable is: a. b. c. d. distance. speed. acceleration. the slope of the speed vs. distance curve. ____ 15. When constructing a graph, the x-axis is most closely related to: a. the dependent variable. b. the vertical axis. c. the independent variable. d. the slope. ____ 16. Use the graph to predict the speed of the car when the car is at 60 cm. a. b. c. d. 220 cm/s 230 cm/s 240 cm/s 250 cm/s ____ 17. Calculate the speed of the object from the position vs. time graph shown below. a. 0.5 m/s b. 0.75 m/s c. 2 m/s d. 3 m/s ____ 18. Which point on this position vs. time graph has the fastest speed? a. b. c. d. Point 1 Point 2 Point 3 Point 4 ____ 19. In which situation would a bicycle rider NOT be accelerating? a. If her direction and speed were unchanged. b. If her direction changed and speed was constant. c. If her speed increased and direction was unchanged. d. If her speed decreased. Selina drops a golf ball down a deep well. The ball falls with only the force of gravity acting on it. Figure 4-2A ____ 20. Without any air resistance, as the golf ball in Figure 4-2A falls into the well, its speed is: a. constant. b. increasing. c. decreasing. d. directional. ____ 21. Which of the following statements about friction is TRUE? a. Friction is a force that resists motion. b. Friction should always be removed. c. Friction only occurs between solids. d. Oils and other lubricants totally eliminate friction. ____ 22. The diagram shows a box being pushed from left to right. Which shows the force of sliding friction on the box? a. A b. B c. C d. D ____ 23. How can the force of friction between two surfaces be reduced? a. By changing rolling friction to sliding friction. b. By making smooth surfaces rougher. c. By separating surfaces with a lubricant. d. By increasing the force between the surfaces. ____ 24. Luka pushes a box across the floor in the direction shown by the arrow in the diagram below: The direction of the friction force acting on the box is: a. up. b. down. c. right. d. left. ____ 25. A net force must be applied to an object to do all of the following EXCEPT: a. turn. b. maintain constant speed. c. slow down. d. stop moving. ____ 26. The picture below shows 4 forces applied to an airplane. Due to the forces acting on the airplane, the airplane: a. speeds up. b. slows down. c. goes higher. d. goes lower. ____ 27. The diagram below is a free-body diagram picturing a cyclist moving along a highway. 4 forces, represented by arrows A, B, C, and D are acting on the cyclist as she pedals. The arrow that would best represent the normal force is: a. A b. B c. C d. D ____ 28. The property of matter that resists changes in motion is: a. acceleration. b. inertia. c. force. d. speed. ____ 29. Newton’s first law of motion is also known as: a. the law of universal gravitation. b. the law of force pairs. c. the law of unbalanced forces. d. the law of inertia. ____ 30. A 0.15-kilogram baseball and a 7.25-kilogram bowling ball are both rolling along at 3 m/s. Which object is easier to stop and why? a. The bowling ball because it has less inertia b. The baseball because it has more inertia c. The bowling ball because heavy objects are naturally prone to stop on their own d. The baseball because it has less inertia ____ 31. An object at rest has: a. both inertia and momentum. b. neither inertia nor momentum. c. momentum but no inertia. d. inertia but no momentum. ____ 32. Compare the inertia and momentum of a 0.15-kilogram baseball moving at 50 m/s with the inertia and momentum of a 7.25-kilogram bowling ball moving at 1 m/s. a. The baseball has more inertia, but the bowling ball has more momentum. b. The baseball has both more inertia and more momentum. c. The bowling ball has more inertia, but the baseball has more momentum. d. The bowling ball has both more inertia and more momentum. ____ 33. Tessa uses a toy slingshot to launch a tennis ball across the park for her dog to fetch. For her first launch, she uses 50 N of force. Her second launch uses 100 N of force, and her third launch uses 150 N. Which launch had the greatest acceleration of the tennis ball? a. The 50-N launch b. The 100-N launch c. The 150-N launch d. They all had the same acceleration. ____ 34. A rocket can fly into space because: a. when it is launched, the hot exhaust gases hit the ground and push the rocket forward. b. the rocket pushes the exhaust gases backward, and there is an equal and opposite reaction pushing the rocket forward. c. when the gases are burning up, the mass of the rocket decreases, changing the amount of gravity on the rocket. d. the launch pad pushes the rocket forward like a slingshot. ____ 35. “Forces occur in pairs” is another way of stating Newton’s: a. first law of motion. b. second law of motion. c. third law of motion. d. universal law of motion. ____ 36. According to Newton’s third law, an action reaction pair of forces have: a. equal magnitudes in the same direction. b. different magnitudes in the same direction. c. equal magnitudes in opposite directions. d. different magnitudes in opposite directions. ____ 37. A 100-kg astronaut is floating in outer space. If the astronaut throws a 2-kilogram wrench at a speed of 10 meters per second, which best describes her velocity?: a. Less than 10 m/s in the same direction as the wrench b. Greater than 10 m/s in the same direction as the wrench c. Less than 10 m/s in a direction opposite to the wrench d. Greater than 10 m/s in a direction opposite to the wrench ____ 38. You stand on a skateboard and push off the ground with one of your feet. The reason that the skateboard moves forward is that the force you apply to the skateboard: a. equals the force applied to you by the skateboard. b. is larger than the force applied by you to the skateboard c. equals the force applied by the ground to the skateboard. d. is larger than the force applied by the ground to the skateboard. ____ 39. Batteries are devices that change chemical energy to ____ energy. a. mechanical b. radiant c. electrical d. nuclear ____ 40. Energy can be thought of as stored ____. a. Force b. Mass c. Weight d. Work ____ 41. What happens to the kinetic energy of a moving car if you double the mass of the car? a. It increases by 4 times b. It increases by 2 times c. It decreases by 4 times d. It decreases by 2 times. ____ 42. What happens to the potential energy of a car parked at the top of a hill if you double the mass of the car? a. It increases by 4 times b. It increases by 2 times c. It decreases by 4 times d. It decreases by 2 times. ____ 43. On what does kinetic energy depend? a. Mass and velocity b. Gravity and mass c. Gravity and height d. Velocity and gravity ____ 44. Which of the following use the same units of measurement? a. Power and work b. Force and work c. Energy and work d. Power and energy ____ 45. What is the most work that can be done with 100 joules of energy? a. 10 J b. 100 J c. 1,000 J d. 10,000 J ____ 46. The action that would require NO work to be done is: a. holding a 100-pound object over your head. b. pushing a 25-kilogram box of books across the floor. c. pedaling a 100-newton bicycle up a small hill. d. lifting a balloon filled with air from the floor to a desktop. ____ 47. What is required to get work out of a machine? a. b. c. d. Friction Efficiency Input work Gears ____ 48. There are many different ways to classify matter, but one way is to use only two categories for ALL matter. All matter can be classified as either: a. solids or liquids. b. mixtures or substances. c. atoms or molecules. d. elements or compounds. ____ 49. A substance made of two or more elements that cannot be separated by physical means is called a: a. compound. b. homogeneous mixture. c. heterogeneous mixture. d. multi-element. ____ 50. Which of the following is true of a heterogeneous mixture? a. It is not possible to separate the various parts of the mixture. b. Taking one sample will enable you to determine what it is made of. c. Different samples may not be exactly alike. d. The density is the same throughout the entire mixture. ____ 51. The smallest particle of an element that retains the identity of the element is a(n): a. compound. b. atom. c. molecule. d. ion. ____ 52. The term used to describe anything that has mass and takes up space is: a. weight b. capacity c. volume d. matter ____ 53. The number of degrees between the freezing point of water and the boiling point of water on the Celsius scale is: a. 0 b. 100 c. 180 d. 273 ____ 54. The temperature at which water molecules have the LEAST amount of thermal energy is: a. 0ºC b. 32ºF c. -273 K d. 0 K ____ 55. Solids retain their shape and size, if kept at constant temperature and pressure. At a constant temperature and pressure, liquids retain their: a. volume. b. shape. c. size. d. shape and volume. ____ 56. All of the following phases of matter may flow EXCEPT: a. plasma. b. solid. c. liquid. d. gas. ____ 57. The phase of matter most likely to be present at absolute zero would be: a. plasma. b. gas. c. liquid. d. solid. ____ 58. Evaporation occurs when: a. a solid changes to a gas. b. a gas changes to a liquid. c. a liquid changes to a solid. d. a liquid changes to a gas. The figure below represents a graph drawn using data taken as a substance is cooled. Answer the questions below based upon this graph and your knowledge of phase change. Figure 10-1A ____ 59. The minimum time required for the substance in Figure 10-1A to change from all liquid to all solid is: a. 30 minutes. b. 11 minutes. c. 8 minutes. d. 5 minutes. ____ 60. The boiling point of the substance Figure 10-1A is: a. 40ºC b. 130ºC c. 230ºC d. 300ºC ____ 61. The change which is NOT a physical change is: a. bending of a copper wire. b. melting of an ice cube. c. formation of water from oxygen and hydrogen. d. ripping a piece of paper in half. Graph 12-2 was prepared from data taken by measuring the mass and volume of varying amounts of 4 different substances, A, B, C, and D. Graph 12-2 ____ 62. According to Graph 12-2, the density of substance C is: a. 0.67 g/cm3 b. 1.0 g/cm3 c. 1.5 g/cm3 d. 3.0 g/cm3 ____ 63. When poured into water, glycerol (also a liquid) falls to the bottom of the beaker. Which of the following is a true statement? a. The water is more dense than the glycerol. b. An object that floats in water will always float in glycerol. c. An object that floats in glycerol will always float in water. d. The glycerol is less dense than the water. ____ 64. According to Boyle’s law, the volume and pressure of a fixed quantity of gas are: a. show no relationship. b. are inversely related. c. directly related. d. independently related ____ 65. The scientific law relating the pressure and temperature of a gas maintained at constant mass and volume is called: a. Pascal’s law. b. Boyle’s law. c. Charles’ law. d. Gay-Lussac’s law. ____ 66. A hot air balloon will rise in air because: a. the volume of air inside the balloon is less than the volume of air outside the balloon. b. the mass of the air inside the balloon is less than the mass of the air outside the balloon. c. the density of air inside the balloon is less than the density of air outside the balloon. d. the temperature of the air rises as the balloon gets closer to the Sun. ____ 67. The same amount of air is pumped into two containers. Container A has a volume of 10 liters. Container B has a volume of 5 liters. Both containers are held at the same temperature. Which statement below is true? a. The pressure in container A is equal to half of the pressure in container B. b. The pressure in container A is equal to twice the pressure in container B. c. The pressure in container B is equal to half of the pressure in container A. d. The pressure in both containers is the same. ____ 68. The "building blocks" of matter are: a. electrons. b. atoms and molecules. c. found only in the plasma state. d. as of yet undiscovered. ____ 69. Protons and neutrons are found grouped together in the: a. electron cloud. b. charge. c. periodic table. d. nucleus. ____ 70. Protons and neutrons in the nucleus are held together by: a. gravity. b. electromagnetic force. c. the strong nuclear force. d. the weak force ____ 71. Compared to protons, electrons have: a. much smaller mass and opposite charge. b. about the same mass and opposite charge. c. much larger mass and the same charge. d. much larger mass and opposite charge. ____ 72. A particle with zero charge found in the nucleus of an atom is called a(n): a. electron. b. proton. c. neutron. d. positron ____ 73. A common isotope of Carbon has an atomic number of 6 and a mass number of 13. How many neutrons are in an atom of this isotope? a. 6 b. 7 c. 12 d. 13 ____ 74. Do any of the atom diagrams below represent atoms of the same element? a. b. c. d. No, they are all different elements. Yes, atom A and atom B are the same element. Yes, all of the atoms are the same element. Yes, atom A and atom C are the same element. ____ 75. Electrons with higher energy are located: a. farther from the nucleus of the atom. b. closer to the nucleus of the atom. c. in the nucleus of the atom. d. at lower levels of the atom. ____ 76. An atom contains ____ in various energy levels. a. neutrons b. protons c. electrons d. ions ____ 77. An atom emits light when one of its electrons: a. moves from a low energy level to a high energy level. b. remains at a high energy level. c. moves from a high energy level to a low energy level. d. remains at a low energy level. ____ 78. A way of organizing the elements based on their chemical properties is the: a. energy level. b. periodic table. c. nucleus. d. isotope. ____ 79. Characteristics of non-metals include all of the following EXCEPT: a. b. c. d. ductile. brittle. poor conductors. dull. ____ 80. The location of metals on the periodic table is on the: a. left. b. top. c. right. d. bottom. ____ 81. An atomic mass unit is the approximate mass of a single: a. electron. b. proton. c. carbon atom. d. carbon molecule. ____ 82. Which of the following is evidence that a chemical change has occurred? a. A material changes from solid to liquid. b. The temperature of a material changes from 10ºC to 20ºC. c. A material is hammered from a round shape to a flat shape. d. A material changes color from blue to red. ____ 83. Which of the following is an example of a chemical property of a substance? a. Melting point of 150ºC b. Shiny, silver color c. Forms molecules with hydrogen d. Density of 28.7 g/mL ____ 84. The periodic table notation for nitrogen (N ) is shown below. An atom of nitrogen has how many electrons? a. b. c. d. 7 7 or 8 14 or 15 Cannot be determined with the information given. ____ 85. According to the periodic table, the group known as the halogens include the element: a. sodium. b. calcium. c. chlorine. d. phosphorus. ____ 86. Alkali metals, highly reactive with oxygen, are found on the periodic table in group: a. 1 b. 2 c. 17 d. 18 ____ 87. The atomic mass of boron is 10.811. What is the mass number of the most abundant isotope of boron? a. 9 b. 10 c. 11 d. 12 ____ 88. The noble gases, such as helium and xenon, are non-reactive because: a. they have completely filled outer electron shells. b. they are chemically unstable. c. they are unusually large atoms. d. they have been around longest on Earth. ____ 89. Of the following, the best insulator is the element: a. zinc. b. chromium. c. sulfur. d. magnesium. ____ 90. The element that is shiny in appearance, quite dense and liquid at room temperature is: a. silver. b. aluminum. c. mercury. d. zinc. ____ 91. When you eat an apple, the process of digestion involves: a. physical change only. b. chemical change only. c. physical and chemical change. d. phase change. ____ 92. When a chemical change occurs: a. atoms are rearranged. b. the law of conservation of mass is always obeyed. c. the chemical properties of new substances are different from the ones you started with. d. All of the above ____ 93. Of the following, the one which most likely involves a chemical reaction is: a. Slicing a piece of bread produces two pieces. b. Placing a solid and a liquid together produces heat and a gas. c. Hitting a board with a hammer forms a dent in the surface of the wood. d. Heating an ice cube produces liquid water. ____ 94. In the periodic table notation below, what information does the number 40.08 tell you? a. b. c. d. It is the number of protons. It is the number of neutrons. It is the atomic number. It is the atomic mass. ____ 95. The numerical value of the Avogadro number is: a. 6.02 b. 6.02 2310 c. 6.02 10-23 d. 6.02 1023 ____ 96. What is the whole number placed in front of a molecular formula in a chemical equation called? a. Subscript b. Superscript c. Coefficient d. Mole ____ 97. The number of oxygen atoms in a molecule of Mg(OH)2 is a. 1 b. 2 c. 3 d. 5 ____ 98. Of the following equations, the one which is NOT balanced is: CaCl2 + 2H2O a. 2HCl + Ca(OH)2 Mg(OH)2 b. MgO + H2O 2NaOH c. Na2O + H2O CaO d. Ca + O2 ____ 99. The balanced equation for the reaction between iron sulfide and water is best represented by: 2Fe(OH)3 + 3H2S a. Fe2S3 + H2O FeAl S + 3H O 2Fe(OH)3 + 3H2S b. 2 3 2 2Fe(OH)3 + 3H2S c. Fe2S3 + 6H2O Fe(OH)3 + 2H2S d. Fe2S3 + 3H2O ____100. Classify the following reaction: 3CuSO4 + 2Al a. Addition b. Single-displacement c. Double-displacement d. Decomposition Al2(SO4)3 + 3Cu