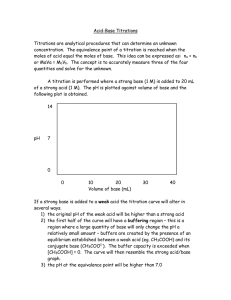
DIPROTIC ACID TITRAION CURVE NAME__________ __________ DATE _____PERIOD___ A.P. CHEMISTRY
advertisement

NAME__________ __________ DATE _____PERIOD___ A.P. CHEMISTRY MARCH 20. 2009 DIPROTIC ACID TITRAION CURVE ABSTRACT: PART ONE: 1) Present a researched titration curve for a weak diprotic acid, a curve for citric acid would be best. 2) Label each part of the curve and explain the theory of the following: a. The weak acid before titration. b. pKa 1 point c. Ka 1 equivalence d. pKa 2 point e. Ka 2 equivalence f. Base overshoot. g. How to calculate the pH of any point on the curve ( buffer zones for Ka 1 and Ka2 etc.) h. Show how to calculate the Ka values if you know the pH OR the volume of base added. i. Show how the Henderson-Hasslebauch equation is adapted for Ka2. 3) Plot your own titration curve with your lab data. CALCULATE the pH for the points mentioned above and compare your answers to the accepted values you researched. 4) Show how to get Ka 1 and 2 from the pH at the important inflection points of the curve.