What is TDF and is it nephrotoxic?
advertisement

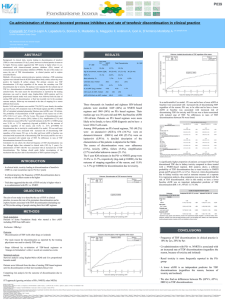
Tuesday Case Conference Introduction • What is Tenofovir disoproxil fumarte (TDF)? • How Nucleotide RTI work • Nephrotoxicity of NtRTI • Is TDF Nephrotoxic? – In vitro evidence – Epidemiologic evidence What is tenofovir disoproxil fumarate (TDF)? • Orally administered pro-drug of tenofovir • Tenofovir is a nucleotide analogue inhibitor of reverse transcriptase (NtRTI) – Others in the family are Adefovir and Cidofovir, well described nephrotoxins – Tenofovir similar to Adefovir – The only NtRTI used for treatment of HIV TDF • Single Agent – Viread • marketed for the treatment of HIV since 2001 • Combination – Truvada • Fixed-dose combination – TDF and emtricitabine (NRTI), 2004 – Atripla • Fixed-dose triple combination of – TDF, emtricitabine (NRTI) and efavirenz (NNRTI), 2006 Reverse Transcriptase Inhibitors • Competitive substrate inhibitors – Nucleoside RTI (NRTI) – Nucleotide RTI (NtRTI) • Non-competitive substrate inhibitor – Non-nucleoside RTI (NNRTI) www.web-books.com/.../Images/HIV_Cycle.jpg How does NtRI work? • Nucleoside+Phosphate – Prolonged action – Low resistance profile • TDF – Compete with normal substrate for viral DNA polymerase (HIV-1 RT) – Minimally interfere with nuclear DNA synthesis Clercq-Holy_Acyclic nuc phosphonates_Nat Rev Pharm_2005 TDF in treatment of HIV infection Clercq-Holy_Acyclic nuc phosphonates_Nat Rev Pharm_2005 Regimen with TDF • >80% of patients with HIV RNA (<50 copies per ml) at 48 weeks Is TDF nephrotoxic? • Potential for nephrotoxicity – Similar structure to Adefovir, known nephrotoxin – Accumulation in renal proximal tubule • Vd of 0.8 L/kg • Minimally protein bound (<8%) • Mainly excreted in urine, unchanged pro-drug from – The Mitochondrial Cytopathy Hypothesis Blood TDF Proximal Tubule OAT1 MRP Lumen TDF is eliminated through the Kidney ATP MRP 2 / 4 Tenofovir Tenofovir Potential for accumulation of high concentration of TDF in proximal tubule cells Mechanisms of Tubular Toxicity The Mitochondrial Cytopathy Hypothesis http://www.retroconference.org/2002/Posters/13560.pdf In vitro study In vitro assessment of mitochondrial toxicity The effect of TDF and other NRTIs on mtDNA synthesis Tenofovir has little mitochondrial toxicity Birkus-Cihlar_Assmt of Mitochondrial Toxicity in Human CellsTreated with Tenofovir_AAC_2002 In vitro study In vitro study Effects of tenofovir and cidofovir on the human renal proximal tubule epithelial cells Inhibition of cell proliferation Effects on viability TDF with low cytotoxicity in proximal tubule epithelial cells Cihlar-Hitchcock_TFV exhibits low cytotoxicity in various human cell types comariosn with other NRTI_Antiviral Res_2002 Does TDF have nephrotoxic effect? • in vitro study – TDF is a weak inhibitor of mamalian DNA polymerases • Has not decreased mtDNA levels • Shows low cytotoxicity • Epidemiologic study… Epidemiology • Phase I/II – – – – – Barditch-Crovo P et al, Antimicrob Agents Chemother. 2001 The Johns Hopkins University School of Medicine N = 49 Tenofovir: 75mg, 150 mg, 300 mg, or 600 mg No renal abnormalities at 28 days • Phase II – – – – – – Schooley, et al, AIDS. 2002 University of Colorado RCT N = 181 Tenofovir: 75mg, 150 mg, or 300 mg No renal abnormalities after 48 weeks Clinical Trials Gilead-Sponsored Clinical Studies of TDF Study Tx- Comparison N Results Available GS903 Naive TDF vs. d4T 600 Week 144 GS902 Exp. TDF vs. Placebo 186 Week 48* GS907 Exp. TDF vs. Placebo 550 Week 48 GS934 Naive TDF/FTC vs. CBV 517 Week 24 TOTAL 1853 In all of these studies the rates of renal abnormalities were similar between TDF and control arms. Clinical Trial • Multicenter, RCT • FU of 144 weeks TDF + 3TC + EFV vs. d4T + 3TC + EFV GS903 Izzedine-Deray_Long-term renal safety of TFV_NephDialTranp_2005 GS903 Incidences of elevated serum creatinine and hypophosphatemia (-0.2) Izzedine-Deray_Long-term renal safety of TFV_NephDialTranp_2005 (-0.1) GS903 Calculated Creatinine Clearance Through Week 144 Conclusion: -Renal safety profile between 2 groups was similar -Incidence of renal failure and hypophosphatemia were reversible -No patient developed Fanconi syndrome Izzedine-Deray_Long-term renal safety of TFV_NephDialTranp_2005 Conclusion from Clinical Trial • Double-blind, placebo-controlled studies – No difference in incidence of renal events between TDF and placebo groups • No TDF-related toxic side effects were noted in the recommended drug combination regimes of TDF Case Report first report of TDF associated fanconi syndrome • Renal failure – Verhelst et al, AJKD, 2002 • First report of TDF related renal failure, nephrogenic DI and Fanconi syndrome • Reversible renal failure with withdrawal of tenofovir • Nephrogenic DI • histology demonstrated mainly proximal tubular cell abnormalities Fanconi Syndrome Proximal Tubule Cell Glucose Phosphate Bicarbonate Sodium Amino Acids X X X Phosphate Hypophosphatemia, acidosis, glycosuria, aminoaciduria, hypokalemia = FANCONI SYNDROME • Literature search for published case reports of TDF related ARF • 1990 - 2005 Tenofovir- associated Renal Dysfunction literature review Characteristic N=27 Age; years 45.5 (31 – 65) Concurrent ARV; n(%) Ritonavir Didanosine 21 (77) 9 (33) Time to diagnosis; months 11.3 (1 – 29) Diabetes insipidus; n (%) 5 (18) Zimmermann-Braden_TFD associated acute and ckd, HIV / AIDS, 2006 Tenofovir- associated Renal Dysfunction literature review Characteristic (N=27) Baseline creatinine; mg/dL 0.9 (0.5 – 2.1) Peak creatinine; mg/dL 3.9 (0.89 – 20) P < .05 Post creatinine; mg/dL 1.2 (0.67 – 2.6) P < .05 Fanconi Syndrome; n (%) 16 (59) Return to baseline creatinine; n (%) 22 (81) Urine protein; n (%) 6/17 (35) Hemodialysis; n (%) 2 (7) Mean (range) unless otherwise specified Zimmermann-Braden_TFD associated acute and ckd, HIV / AIDS, 2006 Tenofovir-induced Fanconi syndrome literature review • Other features – Acidosis, Hypokalemia – When checked TDF levels were elevated • In all cases acidosis, hypokalemia, hypophosphatemia and glycosuria resolved after discontinuation of TDF • Biopsy findings (8) – Proximal acute tubular necrosis (ATN) Tenofovir-induced Fanconi syndrome • Retrospective review of the FDA Adverse Event Reporting System • 2001-2006 Gupta_Tenofovir associated fanconi_AIDS pt care_2008 Conclusion from case reports • Potential role of drug interactions – Ritonavir • has been shown to increase serum TDF by >30% • Inhibitor of MRP-2 -> increase proximal tubular concentration of TDF by decreasing secretion – Didanosine • Coadministration with TDF may increase serum concentration of didanosine -> proximal tubular dysunfction • Polymorphism in the renal tublar drug transporter – variant MRP 2 or 4 The End