AEM Data
advertisement

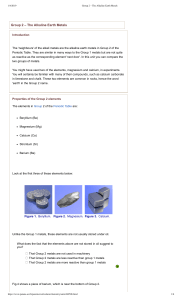
Group 2: The Alkaline Earth Metals Name Part A – Solid and Water Ca + HOH: Result of the test for H2 gas Result of the test of adding phenolphthalein Mg + HOH: Result of test for H2 gas (before heating) Mg + HOH: Result of test for H2 gas (after heating) Result of the test of adding phenolphthalein Part B - pH Mg(OH)2 Ca(OH)2 Ba(OH)2 Part C - Solubility MgSO4 CaSO4 BaSO4 Honors Chemistry Unit 5 – Periodic Law Part D Precipitate - addition of Na2CO3 Observations MgCl2 CaCl2 BaCl2 Questions: 1. Below is a list of compounds that were formed or used in this experiment. Write the correct formulas for each: a) b) c) d) e) f) g) h) i) j) calcium hydroxide magnesium hydroxide barium hydroxide calcium sulfate magnesium sulfate barium sulfate sodium carbonate magnesium chloride calcium chloride barium chloride 2. Describe the reactivity of the metals in Group 2 in terms of their location in the group. 3. How does the reactivity of an alkaline earth metal compare with that of an alkali metal (Group 1) in the same period? 4. What oxidation states can the alkaline earth metals exhibit? Honors Chemistry Unit 5 – Periodic Law 5. Why does the metallic character of the alkaline earth metals increase as you go down the group? Conclusion: Honors Chemistry Unit 5 – Periodic Law