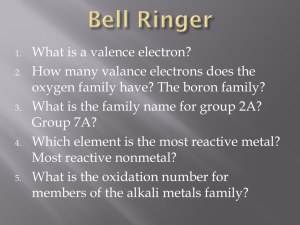graded assignment 6- chapters 7 8- fall2012.doc
advertisement

Graded Assignment 6-Chapters 7, 8; Due Tuesday November 27, 2012 1.For a principle quantum number, n=4 ,what is the total number of orbitals permitted? 2. How many electrons can be placed in n=1 shell? 3. How many electrons can be placed in the n=3 shell? 4.How many orbitals are there in a 5d subshell? 5.How many orbitals are there in a 7s subshell? 6. How many electrons can be placed in the 5f subshell? 7. How many electrons can be placed in a 3p subshell? 8.What is the maximum number of orbitals with n=3, l=2 9. What is the maximum number of electrons with n=4, l=3 10. What is the maximum number of orbitals with n=5, l=1 11. What is the maximum number of electrons with n=2, l=0 12. Which orbitals cannot exist? a. 2p b. 3p c. 4d d. 3f e. 6s f. 2d 13.Circle the set of quantum numbers that is allowed? a. n = 6, l=0, ml=0, ms=1 b. n= 5, l=3, ml = -3, ms = -1/2 c. n=2, l=2, ml = 0, ms = ½ d. n = 3, l=1, ml = -2, ms = 1/2 14. Which of the following quantum numbers represents a 6p electron? a. n = 6, l=0, ml=0, ms=-1/2 b. n= 6, l=5, ml = -3, ms = -1/2 c. n=6, l=1, ml = 0, ms = ½ d. n = 6, l=2, ml = -2, ms = 1/2 15. How many electrons can have the quantum numbers n=5, l=3, ml = 0? Graded Assignment 6-Chapters 7, 8; Due Tuesday November 27, 2012 16. How many electrons can have the quantum numbers n=5, l=3, ml = 0, ms = 1/2? 17. Pick the smallest atom of the set: Te-2, Ba+2, I-, Cs+, Sr+2, Se-2? 18. Which of the following atoms would you expect to have the highest first ionization energy: Te, Br, Sb, Se, Cl? 19. (12 points)Name the orbitals defined by the following quantum numbers: a. n =6, l= 2, ml = 0 b. n = 2, l=1, ml = 1 c. n = 4, l = 3, ml = -1 d. n = 5, l = 0, ml = 0 20. ( 24 points) Write the condensed electron configurations for the following: b. Cr+3 a. Cr c. Cs+ d. Ga e. Se-2 h. Te 21. (10 points) Write all the quantum numbers for a 5d electron? f. Ga+3 g. Sr