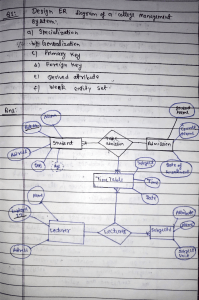
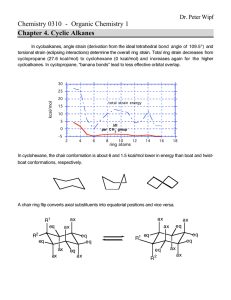
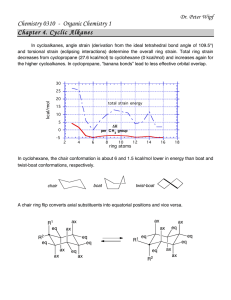
download full powerpoint file
advertisement

Yun Hee Jang, Mario Blanco, Siddharth Dasgupta, William A. Goddard, III MSC, Beckman Institute, Caltech David A. Keire, John E. Shively The Beckman Research Institute of the City of Hope Chelating ligand (DOTA) COO- -OOC N N 90 3+ Y N N -OOC • Therapy: 90Y3+(64h) • Diagnosis: 111In3+(2.8d) 64Cu2+(12.8h) • MRI contrast agent: Gd3+ H C N Antibody O b-emitting targeting (2.25 MeV, t1/2=64h) Tumor cell D. Parker, Chem. Soc. Rev. 19, 271 (1990) -O O O NH+ COO- -OOC N N OO- COO- -OOC +HN N N N O COO- O- DOTA DTPA (log K=24.8) (log K=22.1) Thermodynamic stability O Kinetic inertness at pH 2~8 w.r.t. acidpromoted dissociation <0.5% dissociated over 18 days in serum (pH 7.4, 37oC): inert Rapid complexation x1600 slower than Y-DTPA formation Not inert leading to bone-marrow toxicity Lewis, Raubitschek and Shively, Bioconjugate Chem. 5, 565 (1994) O -O Y3+ + -O O O NH+ O- O NH+ E.T. Clark and A.E. Martell, Inorg.Chim.Acta 190, 27 (1991) X.Y. Wang, et al. Inorg.Chem. 31, 1095 (1992) N Y3+ N O- O- +HN N O- +HN N O OO fast O or O- O -O O slow O -O N O- N Y3+ O O ON N O NH+ N O- O- Y3+ O- O N N O OO Y3+ + H2(DOTA)2- Type I: labile Type II: stable/inert • Calculate structure/energy change occurring during complex-formation • Identify the rate-determining step: Deprotonation or conformation change? • Design new chelating agent and predict its energetics/kinetics • B3LYP/LACVP* // HF/LACVP* (6-31g* for C/H/O/N; Hay-Wadt ECP for Y) • Vibration analysis ZPE / thermodynamic quantity Gibb’s free energy • Continuum solvation calculation by solving Poisson-Boltzmann equation • Jaguar 3.5 (Schrodinger Inc.) -H+ YH2(DOTA)+ Y3+ outside the cage -H+ YH(DOTA) Y(DOTA)the same as x-ray structure of final complex • Y3+ moves into the cage spontaneously with deprotonation. • RMS deviation between ring conformations < 0.5 Å. • Deprotonation is the rate-determining step. Direct attack of outside base on the ring proton? No room for it. top view side view bottom view Conformation change to the one favorable to attack? Too high cost, especially, for YH(DOTA) 4-coordinate YH2(DOTA)+ YH(DOTA) 3-coordinate 2-coordinate 16.6* (12.1)** kcal/mol 42.7* (34.5)** kcal/mol 21.6* (24.6)** kcal/mol * 1.807 Å for r(Y) ** 1.673 Å for r(Y) in solvation calculation Proton transfer from ring NH to COO (more accessible to outside base)? reactant (NH...COO) Relative energy (kcal/mol) Gas-phase E Aqueous-phase DGactivation(aq) TS (N..H..COO) reactant 0 0 TS 2.0 12.2 *,** 8.4 *,** product (N...COOH) Product -25.2 -8.8* (-6.0)** expt'l *** 8.1~9.3 ***experimental DG for Eu,Gd,Ce,Ca-complexes (Inorg.Chem. 32, 4193 (1993)) • Proton transfer is easier than conformation change. • Calculated activation free energy is in agreement with experimental value. Structural change leading to more stable TS: 6-membered ring of DO3A1Pr rather than 5-membered ring of DOTA -O O O NH+ N O- -O OO O +HN N one more CH2 DO3A1Pr (Pr=propionate) TS (DO3A1Pr) TS (DOTA) B3LYP//HF in aqueous phase (kcal/mol) DO3A1Pr DOTA Energy barrier 4.5 12.2 Activation free energy 3.9 8.4 -O O O one more CH2 -O O N N O- -O O O N N +HN O- +HN N N OO OO one more CH2 Hpr(DO3A1Pr): 0.0 kcal/mol O O- Hac(DO3A1Pr): 7.8 kcal/mol Protonation at propionate site is more stable. 6-membered ring TS • Deprotonation from ring nitrogen is the rate-determining step. • Deprotonation occurs by proton transfer from ring nitrogen to carboxylate. • Adding CH2 to one carboxylate arm can improve the incorporation rate. • Explicitly-coordinated water molecules How many water molecules? Effect on structure/energetics • Introduction of amide linkage -O O O NH+ O- N ? O +HN N N OO H Caltech City of Hope William A. Goddard, III John E. Shively Siddharth Dasgupta David Keire Mario Blanco Daniel Mainz Sungu Hwang Supported by NSF