IMPROVING PERFUSION OF SYNTHETIC SKIN Jordan S. Pober and Jeffrey S. Schechner
advertisement

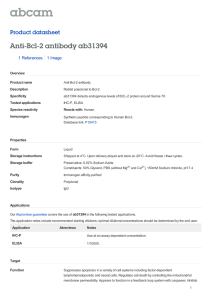
IMPROVING PERFUSION OF SYNTHETIC SKIN Jordan S. Pober and Jeffrey S. Schechner CLINICAL PROBLEM • Patients with impaired angiogenesis (e.g., the elderly, diabetics) develop non-healing wounds • Avascular skin substitutes (Apligraf) may promote healing but fail as long term grafts • Graft failure is ischemic, not immunological HYPOTHESIS • Incorporating endothelial cells into synthetic skin will promote perfusion • The effect will be most obvious in a setting of impaired angiogenesis EXPERIMENTAL SYSTEM • Synthetic skin prepared from decellularized human dermis seeded with human keratinocytes +/- human endothelial cells • Orthotopic skin graft on immunodeficient (C.B-17 SCID/bg) mice +/- rapamycin • Assess vascularity by histology Vascularized Synthetic Skin (orthotopic implant) Human Neonatal Foreskin Human Umbilical Vein Human Adult Cadaver Skin Human Umbilical Cord Blood Human Adult Peripheral Blood EPC Cultured Keratinocytes Decellularized Dermis Simple Synthetic Skin Control SCID Mouse Recipient Moderately Vascularized Skin Graft (Mouse EC-Lined Vessels Only) Cultured EC +/- Bcl-2 Transduction EC-Seeded Synthetic Skin Rapamycin-Treated SCID Mouse Recipient Control SCID Mouse Recipient Rapamycin-Treated SCID Mouse Recipient Poorly Vascularized Skin Graft (Necrosis) Well Vascularized Skin Graft (Mouse and Human EC-Lined Vessels) Well Vascularized Skin Graft (Human EC-Lined Vessels Only) A B C D E F Figure 1 Culture of human EC derived from cord blood or adult peripheral blood (a). Colonies of differentiated cells were noted as early as 7-10 days after isolation from CB and 21-35 days after isolation from AB using the conditions of isolation and culture described in the Materials and Methods(b). Colonies of differentiated EC differentiated from CB-EPC continued to proliferate, establishing a cellular monolayer capable of serial passage and expansion. AB-EC exhibited similar behavior (c,d). Immunofluorescence microscopy of monolayers of cells derived from AB-EPC revealed the presence of VE-cadherin at the cell junctions, and cytoplasmic granules of vWF (e,f), indicative of differentiated EC. Similar cells were derived from CB-EPC. Each cell type was cultured on at least five separate isolations with similar results. A B C D E F Figure 5 Histologic analysis of microvessels in human skin equivalents harvested 21 days after transplantation. Note the paucity of vessels in grafts constructed without EC-seeding (a), compared with well vascularized grafts seeded with AB-EC (b) or CB-EC (c). In human skin equivalents seeded with CB-EC both mouse EC-lined (d) and human EC-lined (e) vessels were present. The observed microvessels were coated by cells expressing smooth muscle cell-specific alpha actin, indicative of vessel maturation. All images are representative of the results seen in AB-EC or CB-EC-seeded human skin equivalents from at least four separate experiments with each cell type (except actin staining, which was only examined in two experiments). 3500 3500 3000 3000 3000 2500 2500 2500 * * 2000 1500 1000 500 Vessels/mm2 3500 Vessels/mm2 Vessels/mm2 HUMAN EC-LINED VESSELS 2000 1500 1000 500 0 HUVEC * HUVEC Rapamycin # HUVEC Bcl-2 Rapamycin 1500 1000 500 0 HUVEC Bcl-2 2000 0 CB CB Bcl-2 # CB Rapamycin # CB Bcl-2 Rapamycin AB EPC AB EPC Bcl-2 # AB EPC Rapamycin # AB EPC Bcl-2 Rapamycin # 2000 2000 1500 1500 1500 1000 500 Vessels/mm2 2000 Vessels/mm2 Vessels/mm2 MOUSE EC-LINED VESSELS 1000 500 0 0 HUVEC HUVEC Bcl-2 HUVEC Rapamycin HUVEC Bcl-2 Rapamycin 1000 500 0 CB CB Bcl-2 CB Rapamycin CB Bcl-2 Rapamycin AB EPC AB EPC Bcl-2 AB EPC Rapamycin AB EPC Bcl-2 Rapamycin New Questions • Will the inclusion of EC accelerate and/or increase perfusion of synthetic skin? • What is the best source of EC? • Will Bcl-2 or other transduced genes improve EC performance in synthetic skin? • Will endothelialized skin provoke rejection? • CAN PERFUSION BE QUANTIFIED?