Investigating Human T cells Using Multicolor Flow Cytometry Insoo Kang Section of Rheumatology
advertisement

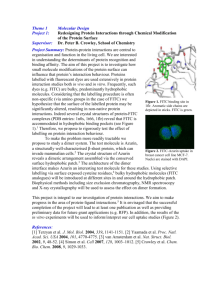
Investigating Human T cells Using Multicolor Flow Cytometry Insoo Kang Section of Rheumatology Aging and IL-7-mediated CD8+ T cell survival • IL-7, largely produced from thymus, is critical for the development and survival (homeostasis) of CD8+ T cells. • Decreased naïve CD8+ T cells and impaired memory CD8+ T cell responses occur with aging. • Decreased plasma levels of IL-7 have been observed with aging. • IL-7 has been used to rejuvenate T cell immunity in aged mice and non-human primates, without significant success. • It is critical to determine whether aging also affects other steps involved in IL-7-mediated CD8+ T cell survival pathway. • Objective: Investigating IL-7 receptor expression on CD8+ T cell subsets and their signaling and survival responses to IL-7 in healthy young (≤ 40 years) and elderly (≥ 65 years) people. Exclusion criteria: Individuals who were taking immunosuppressive drugs or who had a disease potentially affecting the immune system including infection, cancer, asthma, autoimmunity and diabetes were excluded from the study IL-7 receptor (R) signal transduction pathways IL-7 gc a P Jak1 + STAT5 X ? P ? Jak3 Box1 region @Y449, STAT5 docking sites PI3 kinase P PTEN BCL-2 Retention of Bax Akt Survival (major) P P BAD inactivation FOXO inactivation GSK inhibition Survival Proliferation Glucose use MCB 2004, 24(14):6501-6513 J Exp Med. 2004,200(5):659-69 Immunol Rev. 2003,192:7-20 Flow cytometry is the key step for experiments Measure IL-7Rα and γC expression by different CD8+ T cell subsets as well as their cellular characteristics using flow cytometry (FACSCalibur® and LSRII®). Peripheral blood mononuclear cells (PBMCs) from human subjects Stain cells with Abs Stimulate cells with IL-7 or PBS. Using FACSAria® Sort cells into different cell subsets Measure cell signaling using flow cytometry (FACSCalibur®) Conduct functional studies IL-7Ra expression by subsets of CD8+ T cells PBMCs 600 400 58 200 Elderly 600 400 10 2 10 1 0 200 400 600 800 FSC-H: FSC-Height CM 1000 10 10 0 EM 284 10 1 2 10 10 FL4-H: APC CD8 3 10 4 EMCD45RA+ 286 47.1 CM 3 Naive EM 24.5 0 252* 10 200 0 Naive 4 5.23 CD8+ T cells CCR7 800 10 FL2-H: PE CCR7 800 FSC-H: FSC-Height 1000 SSC-H: SSC-Height Stain with Abs to CD8, CD45RA, CCR7 and IL-7Rα, γC or isotype 1000 EM CD45RA+ 8.99 38.6 0 10 0 10 1 2 10 10 FL3-H: Cyc CD45RA 3 10 CD45RA 55 Isotype anti-IL-7Ra Ab 281 Young 271 306 low 267 high *median fluorescen t intensity IL-7Ra 4 More than 4 markers can be simultaneously measured using LSRII® Young 0 Late 0.212 0 10 3 4 103 102 0 4.722 10 10 10 <FITC-A>: CD28 0 10 2.61 5 105 47.7 104 103 105 102 102 0 0 73 0 10 105 39.7 104 103 103 105 102 102 0 0 0 10 103 10 104 <FITC-A>: CD28 44.72 0 10 0.27 5 103 104 10 <FITC-A>: CD28 CD28 5.49 103 104 105 <FITC-A>: CD28 IL-7Ralow 105 3.44 1.47 103 102 0 28 3.69 5 94.22 103 104 10 <FITC-A>: CD28 9.93 0 10 105 12.3 0.86 103 104 105 <FITC-A>: CD28 1.16 104 103 0 1.4 5 30.3 104 102 5.622 0 10 104 0 102 15.2 104 12 54 103 104 10 <FITC-A>: CD28 <APC-A>: CD27 19.7 <APC-A>: CD27 105 103 104 10 <FITC-A>: CD28 0.81 5 <APC-A>: CD27 0 10 22.52 10 10 10 <FITC-A>: CD28 103 0 32.62 0.4 5 4 104 102 5.03 5 0 3 IL-7Rahigh 104 7.712 102 0 10 18.8 64.6 7.58 103 2.612 10 10 10 <FITC-A>: CD28 <APC-A>: CD27 67.7 103 EMCD45RA 4 IL-7Ralow <APC-A>: CD27 EM 19.8 3 105 104 103 0 IL-7Rahigh 105 104 102 0.043 5 89.2 7.8 <APC-A>: CD27 Early 105 <APC-A>: CD27 104 103 102 80.9 11.6 CM <APC-A>: CD27 Int 105 <APC-A>: CD27 104 96.6 3.18 <APC-A>: CD27 105 <APC-A>: CD27 Markers used CD8, CD45RA, CCR7, IL-7Ra CD27 and CD28 Elderly Naive CM <APC-A>: CD27 Naive 103 102 0 61 0 102 1.09 5 103 104 10 <FITC-A>: CD28 85.92 0 10 0.6 103 104 105 <FITC-A>: CD28 P-STAT5 in subsets of CD8+ T cells in response to IL-7* can be measured using flow cytometry Stain cells with CD8,CD45RA and CCR7 Stimulate cells with IL7 or PBS. Naive CM 40 20 100 80 80 80 60 60 60 40 20 0 10 1 10 3 10 4 0 10 1 % of Max 60 40 20 2 10 FL1-H: FITC 10 3 10 4 0 10 1 2 10 FL1-H: FITC 10 3 10 4 0 10 1 2 10 FL1-H: FITC 10 3 10 4 80 60 60 60 40 20 0 10 1 2 10 FL1-H: FITC 10 3 10 4 10 1 2 10 FL1-H: FITC 10 3 10 4 Phospho-STAT5 40 *10 min at 10 ng/ml 20 0 10 IL-7Ra expression 0 0 10 100 80 0 10 EMCD45RA+ 40 80 20 0 10 100 40 Measure expression of P-STAT5 by CD8+ T cell subsets using flow cytometry (FACSCalibur®) 20 0 10 100 IL-7 PBS 80 2 10 FL1-H: FITC % of Max 0 40 20 0 10 100 % of Max 100 % of Max 60 % of Max % of Max Elderly a-IL-7Ra IgG EM 100 % of Max 100 80 Permeabilize and stain cells with Abs to P-STAT5 or isotype. % of Max PBMCs 0 10 0 10 1 2 10 FL1-H: FITC 10 3 10 4 10 0 10 1 2 10 FL1-H: FITC 10 3 10 4 Advantage of flow cytometry: Cell signaling can be measured in a small number of cells (<100,000) and quantitative analysis is possible. IL-7Ralow cells have decreased survival in response to IL-7 Sorted into subsets Using FACSAria® Naive EM 104 0.05 19.4 26.6 16.6 101 102 103 FL1-H: FITC annexin 104 104 63.8 9.54 101 102 103 FL1-H: FITC annexin 104 104 0.14 4.48 3 0.01 FL3-H: 7 AAD 89.8 100 100 5.59 101 102 103 FL1-H: FITC annexin 104 0.32 10.6 77.2 12.6 101 102 103 FL1-H: FITC annexin 104 104 0.51 55.5 19.5 24.5 10 102 101 100 100 17.6 101 102 103 FL1-H: FITC annexin 3 102 101 13.7 100 100 104 10 102 101 104 FL3-H: 7 AAD 10 102 13.5 101 102 103 FL1-H: FITC annexin 3 FL3-H: 7 AAD 10 58.4 100 100 10.1 68.4 101 104 3 0.29 102 101 100 100 EMCD45RA+ IL-7Ralow 103 102 101 63.9 104 28 FL3-H: 7 AAD 102 100 100 0.11 103 FL3-H: 7 AAD 102 101 IL-7 104 0.06 103 FL3-H: 7 AAD 103 EMCD45RA+ IL-7Rahigh Measure live cells using flow cytometry FL3-H: 7 AAD 104 PBS Incubate cells with IL-7 FL3-H: 7 AAD PBMCs 101 79.8 100 100 9.37 101 102 103 FL1-H: FITC annexin 104 100 100 101 102 103 FL1-H: FITC annexin 104 FACS sorting can be used to measure gene expression in a small number (100,000) of specific cell subsets P < 0.05 50 40 30 P > 0.05 20 Naive EMCD45RA+ IL-7Rahigh 10 EMCD45RA+ IL-7Ralow 0 IL-7Ra GABPa GFI-1 Elderly (n=5) Conclusions • Our findings suggest that aging affects IL-7Ra expression by CD8+ T cells leading to impaired signaling and survival responses to IL-7. • Flow cytometry is valuable in investing the phenotypes and functions of human immune cells. Kim et al, Blood 06 Acknowledgements • Hang-Rae Kim • Myung Sun Hong • Kyung-A Hwang • Ping Zhu • Chris Bailey • Barbara Foster • Lynne Iannone • Yale Flow Cytometry Facility • • • • • • Hartford Foundation Arthritis Foundation NIH/NIAMS Lupus Foundation AFAR Yale Pepper Center

![Anti-CD45RA antibody [B-C15] (FITC) ab27290 Product datasheet 1 References 1 Image](http://s2.studylib.net/store/data/012441203_1-afe088a0d54485dec40ccf05d944a1ff-300x300.png)

![Anti-CD1c antibody [L161] (FITC) ab95757 Product datasheet 1 Image Overview](http://s2.studylib.net/store/data/012447966_1-9fc496582347445642446baa74f4c68d-300x300.png)