Refining Views of Co-morbidities and Vascular Responsiveness in Pulmonary Arterial Hypertension
advertisement

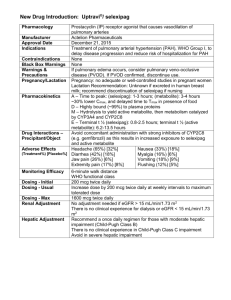
Refining Views of Co-morbidities and Vascular Responsiveness in Pulmonary Arterial Hypertension Roham T. Zamanian, MD, FCCP Assistant Professor of Medicine Director, Adult Pulmonary Hypertension Clinical Service Division of Pulmonary & Critical Care Medicine Stanford University School of Medicine Disclosures: Personal Financial Relationships with Commercial interests relevant to medicine, within the past 3 years: Consultant: Gilead, United Therapeutics, Ikaria, Bayer, Actelion Industry-Sponsor Research: United Therapeutics, Gilead, Actelion Personal Financial Relationships with Non-Commercial interests relevant to medicine, within the past 3 years: Research Grants: - Vera Moulton Wall Center for Pulmonary Vascular Disease - NIH/NHLBI - NIH/NIAID Personal relationships with tobacco industry entities within the past 3 years: No relationships to disclose No “off-label” discussions in this presentation. Pulmonary Hypertension - Diagnostic Definition: Diagnostic gold standard = hemodynamics Rest: • Mean PAP >25 mmHg (normal 10-15 mmHg) Exercise: • Mean PAP > 30 mmHg PAH = above + PCWP or LVEDP <15 mmHg (normal 8-10 mmHg) Associated with adverse changes • In the pulmonary vasculature (arteriopathy) • At the level of the right ventricle (hypertrophy) Courtesy Marlene Rabinovitch Dana Point Classification of Pulmonary Hypertension 1. Pulmonary Arterial Hypertension 3. Pulmonary hypertension due to lung diseases and/or 1.1 Idiopathic PAH hypoxia 1.2 Heritable 3.1 Chronic obstructive pulmonary disease 1.2.1. BMPR2 3.2 Interstitial lung disease 1.2.2. ALK1, endoglin (with or without hereditary hemorrhagic 3.3 Other pulmonary diseases with mixed restrictive and telangiectasia ) obstructive pattern 1.2.3 Unknown. 1.3 Drug- and toxin-induced 3.4 Sleep-disordered breathing 1.4 Associated with 3.5 Alveolar hypoventilation disorders 1.4.1. Connective tissue diseases 3.6 Chronic exposure to high altitude 1.4.2 HIV infection 3.7 Developmental abnormalities 1.4.3 Portal hypertension 1.4.4 Congenital heart diseases 1.4.5 Schistosomiasis 4. Chronic thromboembolic pulmonary hypertension 1.4.6 Chronic hemolytic anemia (CTEPH) 1.5 Persistent pulmonary hypertension of the newborn 1’. Pulmonary veno-occlusive disease (PVO) and/or pulmonary 5. PH with unclear multifactorial mechanisms capillary hemangiomatosis (PCH) 2. Pulmonary hypertension due to left heart disease 2.1 Systolic dysfunction 2.2 Diastolic dysfunction 2.3 Valvular disease 5.1 Hematologic disorders: myeloproliferative disorders splenectomy. 5.2 Systemic disorders, sarcoidosis, pulmonary Langerhans cell histiocytosis, lymphangioleiomyomatosis, neurofibromatosis, vasculitis 5.3 Metabolic disorders: glycogen storage disease, Gaucher disease, thyroid disorders 5.4 Others: tumoral obstruction, fibrosing mediastinitis, chronic renal failure on dialysis. Simonneau et al, JACC 2009 Outcomes in the Current Era: Stanford Adult & Pediatric Experience Zamanian et al 2011 REVEAL: Parameters Independently Associated With Survival (Cox Proportional Hazard Estimates) HR p-value APAH-CTD APAH-PoPH FPAH 1.59 <0.001 3.60 <0.001 2.17 0.012 Renal insufficiency Males age ≥60 yrs 1.90 <0.001 2.18 <0.001 I III IV 0.42 0.039 1.41 0.008 3.13 <0.001 Heart rate >92 bpm Systolic BP <110 mm Hg 1.39 0.005 1.67 <0.001 ≥440 m <165 m 0.58 0.008 1.68 <0.001 <50 pg/mL >180 pg/mL 0.50 0.003 1.97 <0.001 WHO Group I PAH Subgroups Demographics and Comorbidities NYHA/WHO Functional Class Vitals 6MWD BNP ECHO Pericardial effusion: any 1.35 0.014 DLCO % predicted DLCO ≥80 % predicted DLCO ≤32 0.59 1.46 0.031 0.018 RHC mRAP >20 mm Hg PVR >32 Wood Units 1.79 0.043 4.08 <0.001 1/8 1/4 1/2 1 2 4 Reduced risk Increased risk Hazard ratios and 95% CI Benza RL et al. Circulation. 2010; 122:164-172. 8 REVEAL: Observed 1-year Survival From Time of Enrollment According to Predicted Risk Strata 100 80 Survival (%) Risk strata Low Average Moderately high High Very high 60 40 0 No. at risk: Low Average Mod. high High Very high 0 1 2 3 1374 665 280 295 102 1368 659 277 293 100 1364 657 274 291 96 1359 653 269 284 89 4 5 6 7 8 Months from enrollment 1356 648 264 277 81 Benza RL et al. Circulation. 2010;122:164-172. 1352 647 263 270 74 1351 640 260 263 72 1346 628 259 255 69 1341 625 255 247 61 9 10 11 12 1336 618 254 241 59 1311 604 249 238 55 1304 602 244 233 52 1303 596 243 225 49 What Is the Optimal Treatment Strategy? Anticoagulate ± Diuretics ± Oxygen ± Digoxin Acute Vasoreactivity Testing Positive Negative Oral CCB No Sustained Response Yes Continue CCB LOWER RISK DETERMINANTS OF RISK HIGHER RISK No Clinical evidence of RV failure Yes Gradual Progression of symptoms Rapid II, III WHO class IV Longer (>400 m) 6MWD Shorter (<300 m) Peak VO2 >10.4 mL/kg/min CPET Peak VO2 <10.4 mL/kg/min Minimal RV dysfunction Echocardiography Pericardial effusion, significant RV enlargement/dysfunction; RA enlargement RAP <10 mm Hg; CI >2.5 L/min/m2 Hemodynamics RAP >20 mm Hg; CI <2.0 L/min/m2 Minimally elevated BNP Significantly elevated McLaughlin VV et al. J Am Coll Cardiol. 2009;53:1573-1619. Therapeutic Options in USA for PAH – September 2012 Traditional Tx • • • Supplemental O2 Diuretics Oral vasodilators • • • • warfarin • • • Digitalis • • Epoprostenol Treprostinil (IV/SQ/Inhaled) Inhaled Iloprost ERA’s • Inotropic agents • Prostanoids • (CCB) Anticoagulants • FDA Approved for PAH Bosentan Ambrisentan Investigational Tx • • • • • • • Sildenafil Tadalafil • • • • • • Oral Treprostinil ERA’s • PDE-5 Inhibitors • Prostanoids Actelion-1 Tyrosine Kinase Inhib’s Rho-Kinase Inhib’s Elastase Inhib’s Dicholoroacetate (DCA) TetrahydroBiopterin S. Guanylate Cyclase Act’s FK-506 LTA4 Hydrolase Inhib’s Stem Cell Tx A Paradigm Shift in PAH Clinical Research • Although we realize that WHO Group I PAH has numerous etiologies, we have assumed to be “homogeneous” in its biology/physiology. • Over the last decade there has been an explosion of clinical research in PAH / PH focused on therapeutics. • A call for clinical-translational research • It is time for a Paradigm Shift: New Tools and Topics • Identification of Novel (modifiable?) Risk Factors / Co-morbidities • Refining our understanding of acute and chronic vascular responsiveness • Recognition of potentially novel therapeutics Overview Insulin Resistance in PAH: • Pulmonary Arterial Hypertension (PAH) is a vascular disease characterized by inflammation, proliferation, and vasoconstriction. • Multi-factorial & Complex • Unlikely that a single factor, pathway, mutation in etiology • Obesity, hyperlipidemia, and insulin resistance (IR) are known risk factors for systemic cardiovascular diseases (CVD). • Impact of obesity or IR in PAH instigation or progression have not been validated. • Several lines of evidence though, suggest a link between insulin resistance and PAH….. IR in PAH: Suggestive Links • Obesity is associated with Insulin resistance • Obesity + Inactivity IR • Obesity is common in PAH and maybe an overlooked risk factor. • Exercise intolerance is a feature of severe PAH • Insulin resistance has been linked to congestive heart failure and idiopathic cardiomyopathies • Elevation of “factors” and inflammatory cytokines which have been implicated in PAH are also involved in pathogenesis of IR • IL-6, ET-1, ADMA, MCP-1 Humbert el al. AJRCCM 1995 Ikeda et al. Am J Physiol Heart Circ Physiol 2002 Stuhlinger et al. JAMA 2002 Yudkin el al. Lancet 2005 Abenhaim et al. NEJM 1996 Rich et al. Chest 2000 Taraseviciute et al. Eur J Med Res 2006 PPARg Activation Reverses PAH in Insulin Resistant ApoE Deficient Mice IR 5 4 6 6 Hansmann et al Circulation 2007 Hypothesis • Insulin resistance: • May be more prevalent in the PAH population • May be associated with disease severity • Modulation of insulin resistance may lead to improved outcomes in PAH Design & Methods • Prospective screening of patients with pulmonary arterial hypertension at SUMC PH Clinic between 2004-2006. • The National Health and Nutrition Examination Survey (NHANES) as control population. • Identification of TG/HDL-C ratio as a surrogate marker for insulin resistance. • Excluded: Overt Diabetics, Secondary PH forms including those with pulmonary parenchymal disease and left heart failure • Data Collection (PAH): detailed demographic, functional, hemodynamics, and event-free survival. Receiver-operating Characteristic Curves for Metabolic Markers of Insulin Resistance McLaughlin & Reaven et al. Ann Int Med. 2003 Higher Prevalence of IR in PAH Zamanian et al, ERJ 2009 Unlike Healthy Controls Insulin Resistance in PAH is NOT Associated with Age or BMI Zamanian et al, ERJ 2009 Zamanian et al, ERJ 2009 IR is a Predictor of Short-term Worse Outcomes 79% 58% Log-rank p = 0.043 Zamanian et al, ERJ 2009 Age & BMI Adjusted Univariate Analysis Zamanian et al, ERJ 2009 Right Ventricular Glucose Metabolism is Altered in IR PAH RV LV Healthy RV LV IR PAH RV LV IS PAH FIGURE 1: Representative FDG PET/CT images in (A) a healthy control and (B and C) 2 patients with PAH. (A) Fasting images in a healthy subject reveal nearly absent uptake in the right ventricle (RV) and minimal patchy FDG activity in the lateral wall of the left ventricle (LV) which is considered normal. (B) By comparison, abnormally intense FDG activity can be identified throughout the right ventricular myocardium in an insulin resistant (IR) patient with PAH. The activity appears significantly greater throughout the RV than in the LV. (C) Interestingly, even non-fasting, glucose challenged images in an insulin sensitive (IS) PAH patient demonstrate relatively normal RV FDG uptake highlighting the potential differences in RV myocardial metabolism between IS and IR PAH. Impact of IR on RV Structure & Function: MESA-RV Study • The Multi-Ethnic Study of Atherosclerosis (MESA) performed interpretable cardiac MRIs on 5,004 participants without clinical cardiovascular disease at six field centers. • 4168 non-diabetic healthy controls were evaluated categorized as IR or IS and Cardiac MRI data analyzed. • IR in healthy cohort is associated with higher BMI, systolic and diastolic blood pressure, and higher CRP. Parameter RVEDM, g RVEDV, mL RVESV, mL RVEF, % RVSV, mL/min Insulin Sensitive 21.48±0.11 125.01±0.55 37.69±0.33 70.66±0.19 90.02±0.55 Insulin Resistant 20.86±0.11 123.19±0.55 36.74±0.33 70.61±0.19 85.67±0.55 p <0.0001 0.0077 0.0269 0.9728 <0.0001 Zamanian et al ATS 2012 A3453 Impact of IR on RV Function in PAH Hemodynamic Data mRAP (mmHg) mPAP (mmHg) RVEDP (mmHg) PCWP (mmHg) CO (L/min) SV (mL) PVR (WU) IS (n=59) 8+/-3.9 51+/-13.5 12.5+/-4.5 10.8+/-3.5 3.9+/-1.2 53+/- 20.5 11.7+/-6 IR (n=25) 8.2+/-4.3 51.5+/-15 12.5+/-6 10.5+/-3.3 4.1+/-1.6 55.5 +/- 25 12.7+/-7.9 p >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 Echocardiography MV E (cm/sec) MV A (cm/sec) Lat E‘ (cm/sec) E/A E/E' TAPSE (cm) IS n=19 81.5+/-17 72.5+/-23.5 13.9+/-3.5 1.18+/-0.4 6.1+/-1.6 2+/-0.4 IR n=11 64.5+/-19 78.5+/-18 10.4+/-2.2 0.8+/-0.2 6.3+/-1.9 2.1+/-0.3 p <0.05 >0.05 <0.01 <0.01 >0.05 >0.05 LA maximum volume index (cm2/m2) 19.5+/-7 17+/-4.5 >0.05 RA maximum volume index (cm2/m2) 45+/-25 40+/-18 >0.05 RV-MPI 0.6+/-0.18 0.65+/-0.18 >0.05 TR max PG (mmHg) RV FAC (%) IVC (cm) TR max PG (mmHg) RV FAC (%) IVC (cm) 82.4+/-16 26+/-10 1.7+/-0.5 82.4+/-16 26+/-10 1.7+/-0.5 75.8+/-15 24+/-11 1.6+/-0.6 75.8+/-15 24+/-11 1.6+/-0.6 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 Skhiri et al ATS 2012 A3463 Early Experience with Pioglitazone in PAH Bosentan Pioglitazone Interim Summary • Insulin resistance is more prevalent in women with PAH than the general female population. • Although obesity may be a link between IR and PAH, our results do not support the idea that obesity alone is the cause of insulin resistance in pulmonary arterial hypertension. • Though IR confers a poorer prognosis, Insulin resistance in PAH does not appear to correlate with functional class or disease severity. • IR may be linked to subtle changes in diastolic ventricular function and right ventricular metabolism. Clinical Case Scenario • 35 yo French female (mother of 2) • Historically very active 3-5 sets of tennis daily • Over last year with profound dyspnea • Can’t garden • 1 episode of LOC picking up child RPAP RPCWP RPAP after 5 minutes NO @ 20 ppm Vasoreactivity Traditionally Defined • Right Heart Catheterization: • Reduction in mPAP by at least 10 mmHg • Must reduce to a mPAP of <40 mmHg. • Cardiac output must be maintained or improved as a result. • Agents used: • Nitric Oxide • Adenosine • Epoprostenol • Standard of Care in 1st time RHC for iPAH. French Registry: Response to Acute Vasodilator Challenge Response (%) 10.3% 6.8% 2.6% 3.3% 1.6% 0% Idiopathic Portal Anorexigens Familial Connective Congenital Tissue Heart Hypertension 0% 0% HIV >2 Factors N=649. Challenge with vasodilator at time of right heart catheterization. Humbert M, et al. Am J Respir Crit Care Med. 2006;173(9):1023-1030. Survival in IPAH on Oral Calcium Channel Blocker Therapy (Long-term Calcium Channel Blocker Therapy) Cumulative Survival 1.0 Responders 0.8 0.6 Failures 0.4 0.2 0 Subjects at Risk (n) 0 2 4 6 8 38 19 33 12 30 7 22 4 13 0 10 Years 8 12 3 14 3 16 2 18 1 Responders Failures Survival endpoint included those who received transplants or were lost to follow-up. Acute response defined as defined by a fall in both mean pulmonary artery pressure (PAP) and pulmonary vascular resistance (PVR) >20%. Sitbon O, et al. Circulation. 2005;111(23):3105-3111. Thinking of the Right Ventricle – PA Compliance • Pulmonary arterial compliance (SV/Ps-Pd) is being recognized as an important contributor to right ventricular afterload and has been shown to be a strong predictor of survival. • Vasoreactivity or milder degrees of vascular responsiveness have not yet been correlated with pulmonary vascular compliance. Lankhaar J-W et al. European Heart Journal (2008)29,1688-1695 Hypotheses 1. Vasoreactivity is found not only in IPAH but also other forms of PAH and can change over time. 2. Even a mild degree of vasoresponsiveness is of prognostic value. 3. Correlating changes in PVR, mPAP and PAC during vasoreactivity testing could help identify additional patients with a reactive vascular bed. Methods • Retrospective study (220 patients PAH Group 1) presented to Stanford Medical Center between 2000 - 2010. • Diagnosed at the time of RHC with vasodilator testing with 20ppm NO • Demographics, functional status, medication as well as hemodynamic parameters were evaluated. • A positive vasoreactivity was defined by a reduction in pulmonary artery mean pressure (PAPm) 10 mmHg to reach an absolute value of PAPm ≤ 40mmHg with an increased or unchanged cardiac output after challenge with NO (20 PPM) • Previous definitions of actue vasoreactivity (as defined by changes in pulmonary vascular resistance (PVR) or PAPm by > 20%) was also evaluated Spiekerkoetter et al, ATS 2011 Overall Demographics and Clinical Characteristics Female:Male 168:59 (74%,26%) 44.8 ± 0.9 Age: Therapies None 96 (42%) Prostanoids PAH etiology: Epoprostenol 20 (9%) Iloprost 9 (4%) Treprostinil 7 (3%) IPAH 38 (17 %) Drugs and toxins 54 (24 %) CTD 45 (20 %) ERA 38 (17%) Portal hypertension 12 (5 %) PDE-I 54 (24%) Congenital heart disease 41 (18 %) multifactorial 37 (16 %) NYHA Baseline hemodynamics NO challenge p mRA 9.3 ± 0.3 - N/A mPAP 56.1 ± 1.0 51.8 ± 1.1 < 0.001 9.7 ± 0.3 - N/A I 4% PCWP II 29% CI 2.13 ± 0.04 2.18 ± 0.05 < 0.005 III 45% SV 48.5 ± 1.3 52.8 ± 1.5 < 0.001 IV 22% PVRI 23.8 ± 0.8 20.0 ± 0.8 < 0.0001 HR 81.6 ± 1.2 78.2 ± 1.2 < 0.0001 MAP 83.3 ± 1.4 83.8 ± 1.1 NS PAC 0.96 ± 0.04 1.16 ± 0.05 < 0.001 6-min walk (m) (n=204) NT-pro BNP pg/mL (n=110) 381 ± 10 1276 ± 168 * 27% on CCB held for VR testing, 28 (12%) on multiple therapies Spiekerkoetter et al, ATS 2011 Clinical Characteristics Based on VR Status Non-Vasoreactive n=203 New Definition Vasoreactivity n=17 Old definition Vasoreactivity n=54 IPAH 31 (15%) 7 (39%) 16 (30%) D&T 49 (23%) 5 (28%) 5 (9%) CTD 43 (21%) 2 (11%) 10 (19%) Portal hypertension 12 (6%) 0 1 (2%) Congenital 40 (19%) 1 (6%) 7 (13%) Multifactorial 34 (16%) 3 (16%) 15 (27%) I 8 (4%) 0 1 (2%) II 55 (24%) 9 (50%) 21 (39%) III 91 (44%) 7 (39%) 25 (46%) IV 46 (22%) 2 (11%) 7 (13%) 49 (22%) 1 (5%) 7 (13%) 9 (4%) 0 2 (4%) PAH etiology NYHA class events death transplant Spiekerkoetter et al, ATS 2011 Spiekerkoetter et al, ATS 2011 Acute Vasoreactivity isn’t “the” determinant to outcomes in PAH Spiekerkoetter et al, ATS 2011 2007 2009 Arterial Phase CHRONIC VASCULAR REACTIVITY VASCULAR REMODELING? Capillary Blush Diffusing Capacity for Carbon Monoxide as a Surrogate of Chronic Vaso-reactivity (remodeling?) • Diffusing capacity of the lung for carbon monoxide (DLCO) is a relatively simple, standardized, inexpensive, and widely available pulmonary function test. • DLCO is recognized as a measure of pulmonary gas exchange efficiency across the alveolar capillary interface. Decreased DLCO is associated with conditions such as parenchymal (e.g. interstitial lung diseases, emphysema) and pulmonary vascular diseases. • The decrease in DLCO in pulmonary hypertension has been thought to be due to pulmonary arterial remodeling and subsequent reduction in perfused pulmonary capillary bed. • Furthermore, reduction in DLCO has been related to the degree of functional capillary surface area loss in scleroderma associated pulmonary arterial hypertension (PAH), suggesting that DLCO is also a marker of endothelial cell function. DLCO Cont • Numerous recent studies have demonstrated the clinical utility of DLCO in PAH. Baseline DLCO can predict long-term survival in PAH. • Decreasing DLCO over time can also predict the development of pulmonary hypertension in patients at risk, such as ones with limited scleroderma. • While changes in DLCO over time have been demonstrated to be more powerful in predicting prognosis in idiopathic pulmonary fibrosis than single-point DLCO measurement, there are currently no studies evaluating the extent and utility of delta DLCO in pulmonary hypertension. Aims 1) to validate previous findings that decreased baseline DLCO is a predictor of poor prognosis 2) to describe changes in DLCO over time (DLCOdelta) 3) to investigate the prognostic utility of DLCOdelta in patients with PH. We hypothesized that decrease in DLCO over time is a predictor of poor prognosis. Saito et al, ATS 2011 PAH or CTEPH (n=313) Figure 1 No DLCO (n=18) Subjects with DLCO (n=295) TLC > = 60% (n=250) TLC TLC < 60% (n=16) TLC unavailable (n=29) FVC > = 60% (n=250) DLCO available (n=268) FVC FVC < 60% (n=11) DLCOadj DLCOadj unavailable (n=22) “Total Cohort” DLCOadj available (n=246) Legend: Included Excluded “Follow-up Cohort” DLCOadj available @ 1yr (n=34) DLCOadj @ follow up unavailable (n=22) Figure 2 100 Survival (%) 75 50 DLCOadj >80% DLCOadj 40-80% 25 DLCOadj <40% Log-rank p = 0.0035 0 0 365 730 1095 1460 Days 1825 2190 2555 2920 Figure 3 Figure 4 100 Survival (%) 75 50 25 Absolute increase in DLCO>=10% Absolute increase in DLCO<10% or loss Log-rank p=0.029 0 0 365 730 1095 1460 Days 1825 2190 2555 2920 External Validation Cohort DLCOdec * DLCOstab DLCOinc ** Figure 1: Change in DLCO & Survival – Kaplan Meier analysis demonstrates best survival for patients who had improvement of DLCO over time. * DLCOdec vs DLCOstab Log rank p=0.041. ** DLCOinc vs DLCOstab Log rank p=0.007. Conclusions • Advancement in understanding of co-morbidities in a rare disorder such as PAH may have profound clinical and therapeutic implications. • Insulin resistance is modifiable (pharmacologic versus non-pharm) • Improvement in clinical phenotyping of the acute vasoreactive pulmonary hypertension patient is needed. • Gain of Vaso-responsiveness maybe suggestive of “reverse • remodelling” Potentially informative of novel clinical endpoints and mechanisms of action. Acknowledgements: Juliana Liu Sherrie Jones Angela Herrera Darlene Frie Yuwen Liao Val Scott Andrew Hsi Patricia Del Rosario Allyson Rupp Nathan Brunner Krithika Ramachandran Mark Nicolls Mark Krasnow Marlene Rabinovitch Jeffery Feinstein Francois Haddad Vinicio De Jesus Perez Edda Spiekerkoetter Kristina Kudelko Lorinda Chung Ramona Doyle Steven Kawut Work Supported by: NIH/NHLBI NHLBI-HV-10-05, 1U01HL107393-01, PAR-09-185, N01-HV-00242 Vera Moulton Wall Center for Pulmonary Vascular Disease