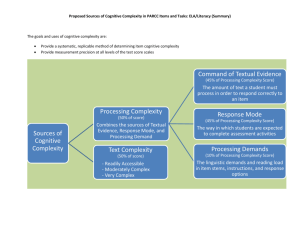
Cognitive stimulation to improve cognitive functioning in people with dementia Authors
advertisement

Cognitive stimulation to improve cognitive functioning in people with dementia Cognitive stimulation to improve cognitive functioning in people with dementia Review information Authors Bob Woods1, Elisa Aguirre 2, Aimee E Spector3, Martin Orrell4 1Dementia Services Development Centre Wales, Bangor University, Bangor, UK College London, London, UK 3Research Department of Clinical, Educational and Health Psychology, University College, London, London, UK 4Research Department of Mental Health Sciences, University College London, London, UK 2University Citation example: Woods B, Aguirre E, Spector AE, Orrell M. Cognitive stimulation to improve cognitive functioning in people with dementia. Cochrane Database of Systematic Reviews 2005 , Issue 4 . Art. No.: CD005562. DOI: 10.1002/14651858.CD005562 . Contact person Bob Woods Professor of Clinical Psychology of Older People Dementia Services Development Centre Wales Bangor University 45 College Road Bangor Gwynedd LL57 2DG UK E-mail: b.woods@bangor.ac.uk Dates Assessed as Up-to-date:6 December 2011 Date of Search: 6 December 2011 Next Stage Expected: 15 January 2013 Protocol First Published: Issue 4 , 2005 Review First Published: Not specified Last Citation Issue: Issue 4 , 2005 What's new Date Event Description Event Description History Date Abstract Background Cognitive stimulation is an intervention for people with dementia which offers a range of enjoyable activities providing general stimulation for thinking, concentration and memory usually in a social setting, such as a small group. Its roots can be traced back to Reality Orientation (RO), which was developed in the late 1950s as a response to confusion and disorientation in older patients in hospital units in the USA. RO emphasised the engagement of nursing assistants in a hopeful, therapeutic process but became associated with a rigid, confrontational approach to people with dementia, leading to its use becoming less and less common. Cognitive stimulation is often discussed in normal ageing as well as in dementia. This reflects a general view that lack of cognitive activity hastens cognitive decline. With people with dementia, cognitive stimulation attempts to make use of the positive aspects of RO whilst ensuring that the stimulation is implemented in a sensitive, respectful and person-centred manner. There is often little consistency in the application and availability of psychological therapies in dementia services, so a systematic review of the available evidence regarding cognitive stimulation is important in order to identify its effectiveness and to place practice recommendations on a sound evidence base. Objectives To evaluate the effectiveness and impact of cognitive stimulation interventions aimed at improving cognition for people with dementia, including any negative effects. 1 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Search methods The trials were identified from a search of the Cochrane Dementia and Cognitive Improvement Group Specialized Register, called ALOIS (updated 6 December 2011). The search termsused were: cognitive stimulation, reality orientation, memory therapy, memory groups, memory support, memory stimulation, global stimulation, cognitive psychostimulation. Supplementary searches were performed in a number of major healthcare databases and trial registers to ensure that the search was up to date and comprehensive. Selection criteria All randomised controlled trials (RCTs) of cognitive stimulation for dementia which incorporated a measure of cognitive change were included. Data collection and analysis Data were extracted independently by two review authors using a previously tested data extraction form. Study authors were contacted for data not provided in the papers. Two review authors conducted independent assessments of the risk of bias in included studies. Main results Fifteen RCTs were included in the review. Six of these had been included in the previous review of RO. The studies included participants from a variety of settings, interventions that were of varying duration and intensity, and were from several different countries. The quality of the studies was generally low by current standards but most had taken steps to ensure assessors were blind to treatment allocation. Data were entered in the meta-analyses for 718 participants (407 receiving cognitive stimulation, 311 in control groups). The primary analysis was on changes that were evident immediately at the end of the treatment period. A few studies provided data allowing evaluation of whether any effects were subsequently maintained. A clear, consistent benefit on cognitive function was associated with cognitive stimulation (standardised mean difference (SMD) 0.41, 95% CI 0.25 to 0.57). This remained evident at follow-up one to three months after the end of treatment. In secondary analyses with smaller total sample sizes, benefits were also noted on self-reported quality of life and well-being (standardised mean difference: 0.38 [95% CI: 0.11, 0.65]); and on staff ratings of communication and social interaction (SMD 0.44, 95% CI 0.17 to 0.71). No differences in relation to mood (self-report or staff-rated), activities of daily living, general behavioural function or problem behaviour were noted. In the few studies reporting family caregiver outcomes, no differences were noted. Importantly, there was no indication of increased strain on family caregivers in the one study where they were trained to deliver the intervention. Authors' conclusions There was consistent evidence from multiple trials that cognitive stimulation programmes benefit cognition in people with mild to moderate dementia over and above any medication effects. However, the trials were of variable quality with small sample sizes and only limited details of the randomisation method were apparent in a number of the trials. Other outcomes need more exploration but improvements in self-reported quality of life and well-being were promising. Further research should look into the potential benefits of longer term cognitive stimulation programmes and their clinical significance. Plain language summary Can cognitive stimulation benefit people with dementia? People with dementia and their caregivers are often advised that 'mental exercise' may be helpful in slowing down the decline in memory and thinking experienced by many people with dementia. This review examined the evidence for one form of mental exercise, described as cognitive stimulation. This involves a wide range of activities that aim to stimulate thinking and memory generally, including discussion of past and present events and topics of interest, word games, puzzles, music and practical activities such as baking or indoor gardening. Typically this is carried out by trained staff with a small group of four or five people with dementia for around 45 minutes at least twice a week. Family caregivers have also been trained to provide cognitive stimulation to their relative on a one-to-one basis. This review included 15 trials with a total of 718 participants. The findings suggested that cognitive stimulation has a beneficial effect on the memory and thinking test scores of people with dementia. Although based on a smaller number of studies, there was evidence that the people with dementia who took part reported improved quality of life. They were reported to communicate and interact better than previously. No evidence was found of improvements in the mood of participants or their ability to care for themselves or function independently, and there was no reduction in behaviour found difficult by staff or caregivers. Family caregivers, including those who were trained to deliver the intervention, did not report increased levels of strain or burden. The trials included people in the mild to moderate stages of dementia and the intervention does not appear to be appropriate for people with severe dementia. More research is needed to find out how long the effects of cognitive stimulation last and for how long it is beneficial to continue the stimulation. Involving family caregivers in the delivery of cognitive stimulation is an interesting development and merits further evaluation. Background Interventions with a cognitive focus have long been used in dementia care. They have been developed in parallel with approaches emphasising the stimulation of the senses (Woods 1977). Reality Orientation (RO) (Taulbee 1966) was developed in the late 1950s as a response to confusion and disorientation in older patients in hospital units in the USA, and 2 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia was the prototype of the cognitive stimulation approach. Classes were held for 30 minutes once or twice per day. Basic personal and current information was presented to the patient and a variety of materials used, such as individual calendars, word-letter games, building blocks and large-piece puzzles. A Reality Orientation board would be used in each session and would list the name of the unit and its location, the day, date, weather, current events etc. The approach emphasised the engagement of nursing assistants in a hopeful, therapeutic process. The first controlled evaluation of RO was reported in the UK by Brook et al (Brook 1975). They found positive benefits on ratings of intellectual and social functioning in patients attending RO group sessions for 30 minutes per day, five days per week for four months compared with a control group who visited a special RO room daily but were not given any encouragement to engage with the materials or each other. A spate of controlled studies followed (Holden 1995), with outcome measures typically including assessments of orientation, other aspects of cognitive functioning and level of independent functioning. A Cochrane review specifically examining Reality Orientation (Spector 2000a; Spector 2000b) concluded that there was some evidence that RO had benefits for people with dementia on both cognition and behaviour. However, outside of a few countries (notably Italy), RO has been little practised or researched since 1990 and has attracted some criticism (Burton 1982; Dietch 1989; Powell-Proctor 1982), especially for being applied in a mechanical, inflexible, insensitive and confrontational manner. Doubts were also raised about the clinical significance of any improvements; the person with dementia might now know what day of the week it was but would this have any meaningful impact on the person's life? Such was the concern regarding the insensitive use of RO and other cognitive approaches that one influential set of guidelines on the management of dementia (APA 1997) cautioned against their use with the possibility of a negative impact on the person's well-being that outweighed any small cognitive improvements. In addition to the 'classroom' element of RO, from the early days '24 hour RO' was also advocated. This involves staff taking every opportunity to provide current information to the person, outside of the formal setting of the RO group, as well as using environmental features such as sign-posting and orientation boards to assist orientation. There have been some positive evaluations of the effects of training and sign-posting on orientation around a care facility (for example Hanley 1981; McGilton 2003) but the effects of 24 hour RO per se have been more difficult to evaluate. This is not least of all because of the difficulty in monitoring its implementation, with informal interactions being much more difficult to document than a formal group meeting. Williams 1987 reports using a modified form of 24 hour RO where staff were trained to respond appropriately to residents' requests for information, and found that 90% of staff responses to residents' requests complied with the treatment protocol. This study reported improvements in cognition, independent functioning and orientation, compared with a control group on a separate ward, when this form of 24 hour RO was implemented. Alongside the RO literature, in recent years there has been increasing discussion of 'cognitive stimulation'. In part this reflects a general view that lack of cognitive activity hastens cognitive decline, in normal ageing as well as in dementia (Breuil 1994; Small 2002), and in part it is an attempt to make use of the positive aspects of RO whilst ensuring that it is implemented in a properly sensitive and respectful manner (Spector 2001; Woods 2002). There has also been growing interest in the application of various forms of cognitive training and in teaching individual people with dementia to use memory aids and strategies to assist with their particular difficulties that have been identified with the person. A Cochrane review of cognitive training and cognitive rehabilitation in early stage dementia has been completed (Clare 2003) and it is important to ensure that clear definitions are used to avoid confusion between the various cognition-based approaches, as in the past 'training', 'stimulation' and 'rehabilitation' have been used almost interchangeably. Clare and Woods (Clare 2004) proposed the following definitions. Cognitive stimulation is engagement in a range of activities and discussions (usually in a group) aimed at general enhancement of cognitive and social functioning. Cognitive training is guided practice on a set of standard tasks designed to reflect particular cognitive functions; a range of difficulty levels may be available within the standard set of tasks to suit the individual's level of ability. It may be offered in individual or group sessions, with pencil and paper or computerised exercises. Cognitive rehabilitation is an individualised approach where personally relevant goals are identified and the therapist works with the person and his or her family to devise strategies to address these. The emphasis is on improving performance in everyday life rather than on cognitive tests, building on the person's strengths and developing ways of compensating for impairments Using these definitions, this review included studies on RO group sessions but not on 24 hour RO or direct training in spatial orientation. The primary outcomes examined will be in relation to the person's cognitive functioning. It is considered that this is the minimum expectation of a general approach with this focus. However, given the concerns discussed above of a possible negative effect, measures of quality of life, mood and well-being are highly pertinent secondary outcome measures. The effects on the person's general level of function in everyday life also need to be considered in evaluating the meaning of any changes observed for the individual and his or her supporters. The impact on family caregivers and careworkers is also important to consider as they are key partners in the process of care. Objectives To evaluate the effectiveness and impact of cognitive stimulation interventions aimed at improving cognition for people with dementia, including any negative effects. To indicate the nature and quality of the evidence available on this topic. To assist in establishing the appropriateness of offering cognitive stimulation interventions to people with dementia and identifying the factors associated with their efficacy. 3 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods Criteria for considering studies for this review Types of studies This review focused on randomised controlled trials (RCTs) for which adequate information was provided or could be obtained from the researchers. The studies included must have been published, written in English and presented in a peerreviewed journal article. Types of participants Participants with a diagnosis of dementia. The main diagnostic categories that were included were Alzheimer's disease, vascular dementia or mixed Alzheimer's and vascular dementia. These diagnostic categories were considered together. Older studies, included from the previous review of RO, used other terms for this population but were included where the review authors were satisfied that the included population would now be described as having a dementia. Participants with mild cognitive impairment, where the extent of cognitive impairment or its effects on day-to-day function were insufficient to justify a dementia diagnosis, were not included. Severity of dementia was indicated through group mean scores, range of scores, or individual scores on a standardised scale such as the Mini-Mental State Examination (MMSE) (Folstein 1975) or Clinical Dementia Rating (CDR) (Hughes 1982). All levels of severity were included. Qualifying participants received the intervention in a range of settings, including their own home, as outpatients and in day-care and residential settings. No specific restrictions regarding age were applied. Data from family caregivers were included where this was available and where the relationship between the caregiver and the person with dementia was specified, including whether they were co-resident. The number of participants receiving concurrent treatment with acetylcholinesterase inhibitors was documented, where possible. Types of interventions Studies were considered for this review if they described a cognitive stimulation intervention targeting cognitive and social functioning. These interventions may also have been described as RO groups, sessions or classes. The definition of cognitive stimulation as proposed by Clare 2004 was adopted. This meant that some studies which described their intervention as 'cognitive stimulation' were excluded. Interventions needed to offer exposure to generalised cognitive activities rather than training in a specific modality. Interventions were typically conducted in a group to enhance social functioning, or could involve family caregivers. Studies were included if a comparison was made to 'no treatment', 'standard treatment' or placebo. Standard treatment was understood to be the treatment that was normally provided to patients with dementia in the study setting and could include provision of medication, clinic consultations, contact with a community mental health team, day care, or support from voluntary organisations. Placebo conditions could consist, for example, of an equivalent number of sessions in which general support, but no structured intervention, was offered. The minimum duration of intervention for inclusion of a study was one month. There were no restrictions on the number of treatment sessions, although this was noted. Types of outcome measures Outcomes were considered in relation to the impact of the intervention on the person with dementia and on the primary family caregiver. Studies could present data in both these categories. Short term (immediately after the intervention) and medium term (follow-up one month to one year after the intervention finished) outcomes were considered. Outcomes for the person with dementia and the caregiver were considered where these were assessed using scores on standardised tests, rating scales and questionnaires. Rates of attrition and reasons for participants dropping out from the study were noted. Outcomes for the person with dementia Outcome measures for the person with dementia sought to identify whether changes were observed following the intervention. The following variables were considered as outcome measures for the person with dementia. Performance on at least one test of cognitive functioning (including tests of memory and orientation). Self-reported, clinically-rated or carer-reported measures for mood of the person with dementia. Self-reported or carer-reported quality of life or well-being measures for the person with dementia. Observer or carer ratings of everyday functioning (activities of daily living) of the person with dementia. Carer ratings of the participant's behaviour. Clinician or carer ratings of neuropsychiatric symptoms or behaviour problems of the person with dementia. Clinician or carer ratings of the social engagement of the person with dementia. 'Carer' in this context included care staff as well as family caregivers. Outcomes for the family caregiver The outcomes for the family caregiver that were considered included any of the following. Self-reported well-being, depression and anxiety. 4 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Self-reported burden, strain and coping. Satisfaction with the intervention. Search methods for identification of studies We searched ALOIS (www.medicine.ox.ac.uk/alois), the Cochrane Dementia and Cognitive Improvement Group Specialized Register, on 6 December 2011. The search terms used were: cognitive stimulation, reality orientation, memory therapy, memory groups, memory support, memory stimulation, global stimulation, cognitive psychostimulation. ALOIS is maintained by the Trials Search Co-ordinator of the Cochrane Dementia and Cognitive Improvement Group and contains studies in the areas of dementia prevention, dementia treatment and cognitive enhancement in healthy populations. The studies are identified from: 1. monthly searches of a number of major healthcare databases: MEDLINE, EMBASE, CINAHL, PsycINFO and LILACS; 2. monthly searches of a number of trial registers: meta Register of Controlled Trials; Umin Japan Trial Register; WHO portal (which covers ClinicalTrials.gov; ISRCTN; Chinese Clinical Trials Register; German Clinical Trials Register; Iranian Registry of Clinical Trials and the Netherlands National Trials Register, plus others); 3. quarterly search of the Cochrane Central Register of Controlled Trials (CENTRAL) (The Cochrane Library); 4. six-monthly searches of a number of grey literature sources: ISI Web of Knowledge Conference Proceedings; Index to Theses; Australasian Digital Theses. To view a list of all sources searched for ALOIS see About ALOIS on the ALOIS website. Details of the search strategies used for the retrieval of reports of trials from the healthcare databases, CENTRAL and conference proceedings can be viewed in the ‘methods used in reviews’ section within the editorial information about the Dementia and Cognitive Improvement Group (CDCIG). Additional searches in each of the sources listed above, to cover the timeframe from the last searches performed for the Specialized Register to December 2011, were run to ensure that the search for the review was as up to date as possible. The search strategies used can be seen in Appendix 1. A total of 804 references were retrieved from the December 2011 search. After de-duplication and a first assessment, authors were left with 41 references to further assess for either inclusion, exclusion or discarding. Data collection and analysis Searches were conducted as detailed above to identify all relevant published studies. The date and time of each search, together with details of the version of the database used, were recorded. Additional information was sought, as outlined above, and hard copies of articles were obtained. Quality assessment RCTs were identified and the two review authors (BW and EA) worked independently to determine which studies met the criteria for inclusion. Trials that did not meet the criteria were excluded, and reasons for exclusion were noted in the table 'Characteristics of excluded studies'. Review authors' selections of trials were compared and the final list of included studies was reached by consensus. The selected RCTs were described in tabular form, permitting an evaluation of their methodological quality. Studies were assessed against a checklist of quality requirements using the Cochrane approach (see risk of bias tables). Grade A, 'Low risk': adequate concealment (randomisation; concealed allocation). Grade B, 'Unclear risk': "randomised", but methods uncertain. Grade C, 'High risk': inadequate concealment of allocation or no randomisation, or both. Only trials with a grade A or B ranking were included in the review. Again, the review authors worked independently to ascertain which studies met the quality criteria, and consensus was reached through discussion. Attempts were made to obtain additional information from the study authors when further data were needed. Data extraction Data from the RCTs selected for inclusion were extracted, recorded and entered into RevMan. The summary statistics required for each trial and each outcome for continuous data were the mean change from baseline, the standard error of the mean change, and the number of patients for each treatment group at each assessment point. Where changes from baseline were not reported, the review authors extracted the mean, standard deviation and the number of patients for each treatment group at each time point, if available. The review authors calculated the required summary statistics from the baseline and post-treatment group means and standard deviations, assuming in this case a zero correlation between the measurements at the baseline and follow-up time points. This method overestimates the standard deviation of the change from baseline but this conservative approach was chosen as it is preferable in a meta-analysis. For binary data, the review authors sought the numbers in each treatment group and the numbers experiencing the outcome of interest. The baseline assessment was defined as the latest available assessment prior to randomisation, but no longer than two months prior. For each outcome measure, data were sought on every patient randomised. To allow an intention-to-treat analysis, the data were sought irrespective of compliance and whether or not the patient was subsequently deemed ineligible or otherwise excluded from treatment or follow-up. If intention-to-treat data were not available in the publications, 'on-treatment' data or the data of those who completed the trial were sought and were indicated as such. In studies where a cross-over design was used, only data from the first treatment phase after randomisation were eligible for inclusion. 5 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia As the outcomes measured in clinical trials of dementia and cognitive impairment often arise from ordinal rating scales, where the rating scales had a reasonably large number of categories (more than 10) the data were treated as continuous outcomes arising from a normal distribution. Data analysis The meta-analyses included the combination of data from trials that may not use the same rating scale to assess an outcome. Therefore, the measure of the treatment difference for any outcome used was the weighted mean difference when the pooled trials used the same rating scale or test and the standardised mean difference (the absolute mean difference divided by the standard deviation) when they used different rating scales or tests. The duration of the trials may vary considerably. If the review authors considered the range too great to combine all trials into one meta-analysis, they divided it into smaller time periods and conducted a separate meta-analysis for each period. Some trials may have contributed data to more than one time period if multiple assessments were made. The meta-analyses presented overall estimates of the treatment difference from a fixed-effect model and a test for heterogeneity was performed using a standard Chi2 statistic. Where there was evidence of heterogeneity of the treatment effect between trials then a random-effects model was utilised (which results in broader confidence intervals than for those of a fixed-effect model). The review authors discussed and reached consensus on the interpretation of the statistical analyses, seeking specialist statistical advice from CDCIG as required. The review authors discussed and reached consensus on the presentation of the findings in the background to the review. Results Description of studies From the initial set of references identified by the updated systematic searches (since our last review, Spector 2000a), 53 potentially relevant studies were identified. Of these, seven studies met the inclusion criteria (Baldelli 2002; Bottino 2005; Chapman 2004; Onder 2005; Requena 2006; Spector 2001; Spector 2003) and were included in this review. Our previous review (Spector 2000a) included eight studies in the meta-analysis, six of which were included in this replacement review ( Baines 1987; Baldelli 1993a; Breuil 1994; Ferrario 1991; Wallis 1983; Woods 1979). For the two studies excluded at this stage, the data needed for the current analyses were not available (Gerber 1991; Hanley 1981). Full details of included studies and reasons for exclusion of excluded studies are presented in the tables 'Characteristics of included studies' and 'Characteristics of excluded studies'. The pre-publication search in December 2011 identified 41 further studies for consideration. Two further studies (Buschert 2011; Coen 2011) met the inclusion criteria and have been added to the review, with three further studies awaiting classification (Buettner 2011; Fernandez-Calvo 2010; Niu 2010). Overall, 718 participants, 407 in the treatment groups and 311 in the control groups, were included in the analyses of the 15 included studies. The included studies varied in many aspects: (1) participant characteristics; (2) number and duration of cognitive stimulation sessions; (3) activities which defined cognitive stimulation; (4) the activity of the control group; and (5) outcome measures. These factors will be considered in turn. 1) Participant characteristics Diagnosis: eight of the nine new studies specified the diagnostic criteria used. Coen 2011 simply described their participants as having mild to moderate dementia; Spector 2001 and Spector 2003 used DSM-IV criteria, but did not break the participants down by dementia subtype; Baldelli 2002 included similar numbers of participants with “Degenerative senile dementia of the Alzheimer’s type (SDAT)” (N = 46) and “vascular multi-infarct dementia” (N = 41), although all had experienced at least one cerebrovascular accident resulting in motor deficits; Bottino 2005, Buschert 2011, Chapman 2004, Onder 2005 and Requena 2006 all specified a diagnosis of probable Alzheimer's disease (AD) according to NINCDSADRDA criteria linked with either ICD-10 or DSM-IIIR criteria. In these studies, participants were on a stable dose of an acetylcholinesterase inhibitor (ACHEI) (rivastigmine in the Bottino 2005 study; donepezil in the remaining studies apart from Buschert 2011 where a variety of medications including memantine were being taken). Amongst the earlier studies, Breuil 1994 specified DSM-III criteria for dementia and Baldelli 1993a stated that their participants were diagnosed with "Alzheimer’s (SDAT) ". The four studies from 1991 and earlier specified more general criteria using cognitive measures that indicated that dementia diagnoses were justifiable (Baines 1987; Ferrario 1991; Wallis 1983; Woods 1979). Ten of the 11 most recent studies provided mean baseline scores on the Mini-Mental State Examination (MMSE), and the 11th (Ferrario 1991) provided the MMSE score range for participants. Coen 2011, Spector 2001 and Spector 2003 were the only studies with a mean MMSE score in the moderate range (10 to 20), perhaps reflecting the upper limit of 24 for participants to be included in these studies. The mean scores (16.9, 13.1 and 14.4 respectively) were several points lower than those in the eleven studies reporting mean scores (mean average 19.7), and outside the range of 18 to 25 specified by Ferrario 1991. However, those studies where the mean score was in the mild range (> 20) may well have included participants in the moderate or even the severe range, for example the lowest score in the Breuil 1994 study was reported to be 9. In general, however, it can be said that the studies included in this review had targeted participants in the mild to moderate range of cognitive impairment. The average age of participants was over 70 years in all studies (except Wallis 1983 where it was 69.8 years); in 6 studies it was over 80 years. The average mean age across the 15 studies was 78.8 years, with the range of ages that were reported from 38 to 97 years. Over half the studies reported inclusion of participant(s) aged 90 years and above. Participants were resident in care homes, nursing homes or hospitals, apart from six studies (Bottino 2005; Breuil 1994; 6 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Buschert 2011; Chapman 2004; Onder 2005; Requena 2006) where all the participants were outpatients living in the community. The participants included in the Spector 2001 and Spector 2003 studies were recruited from both residential care homes and day centres, with the former being in the majority. 2) Length, number and duration of sessions The length of the intervention varied from four weeks (the minimum for inclusion in the review) to 24 months, with the stated length of sessions varying from 30 minutes to 90 minutes. In general, the sessions of longer duration were associated with the lowest frequency (once a week). The median session length across the studies was 45 minutes, and the median frequency was three times a week, ranging from once to five times a week. The total possible exposure to the intervention varied dramatically, from 10 to 12 hours (Baines 1987; Breuil 1994; Chapman 2004; Coen 2011; Spector 2001; Spector 2003 ) to 375 hours in the two-year (Requena 2006) study. Across the 15 studies, the median exposure time was 30 hours. Requena 2006 presented data from both the 12 month and 24 months time point in their study. As there was less attrition at the 12 month time point, and this was more comparable (although still longer) in duration to the other studies, the 12 month data were used in combination with other studies in the meta-analyses, with the 24 month data reported separately. 3) Activities during cognitive stimulation The level of detail provided in the published papers regarding the activities undertaken varied greatly. All studies used small group sessions, typically with groups of five to seven participants, with the exception of Onder 2005 where family caregivers were taught to carry out cognitive stimulation with the person with dementia on an individual basis. These individual sessions, led by family caregivers, included current information, topics of general interest, historical events and famous people, attention, memory and visuo-spatial exercises and the use of clocks, calendars and notes. Early studies described the use of an RO board and discussion of current orientating information through newspapers, photographs, calendars and clocks etc., with materials selected to stimulate all five senses (for example Baines 1987; Wallis 1983; Woods 1979). Breuil 1994 introduced a number of more specific cognitive activities including drawing, associating words, object naming and categorising. Spector 2006 provided a detailed session by session treatment manual for the approach used in their studies. Activities in their sessions were designed with four themes: (1) the senses, (2) remembering the past, (3) people and objects, and (4) everyday practical issues. Activities included naming objects and people, association of words, remembering the past, discussion of hobbies, activities and current affairs, using money, knowing the way around and orientation topics. This treatment manual was also used by Coen 2011. Chapman 2004 reported topics including current events, discussion of hobbies and activities, education regarding Alzheimer’s disease, life story work, and links with daily life with groups of six to seven participants. Bottino 2005 described temporal and spatial orientation, discussion of interesting themes, reminiscence activities, naming people, planning of daily activities and use of calendars and clocks and other external memory aids. Requena 2006 described, for groups of five people with dementia, visual images being shown on a TV screen from a computer and that reflected seven themes: orientation, bodily awareness, family and society, caring for oneself, reminiscing, household activities, animals, people and objects. These were accompanied by questions for discussion. None of the included trials adopted 24 hour RO in addition to group sessions, although Bottino 2005 described involving family caregivers in encouraging the use of external memory aids at home, Buschert 2011 described the use of exercises and tasks to be carried out at home between sessions and Onder 2005 encouraged family caregivers to informally engage in reality-based communication with the person with dementia two or three times a day. 4) Control group(s) activities In the earlier studies, alternative group activities were offered that were of a social (Woods 1979) or diversional (Wallis 1983) nature. Baines 1987 offered an alternative treatment, reminiscence groups, but for the purposes of this review it was the notreatment group that was included in the analyses. 'Treatment as usual' or no treatment was the control condition in a number of studies (Baldelli 1993a; Breuil 1994; Coen 2011; Ferrario 1991; Spector 2001; Spector 2003). In those studies where participants were also taking ACHEIs, the control group were typically monitored in relation to the medication ( Chapman 2004; Bottino 2005; Onder 2005; Requena 2006). Requena 2006 reported that their control participants watched TV whilst the cognitive stimulation groups were in session. Baldelli 2002 engaged both the control and cognitive stimulation participants in a physical therapy programme. Buschert 2011 asked control participants to complete pencil and paper tasks at home, encouraged by monthly group meetings. 5) Outcome measures As a condition of inclusion, cognitive tests were used in all the studies. Eleven studies used the MMSE (Folstein 1975) and eight of the more recent studies also used the Alzheimer's Disease Assessment Scale - Cognitive (ADAS-Cog) (Rosen 1984 ). Unfortunately, only the 10 month follow-up data on these and other measures could be utilised from Chapman 2004 as it has not proved possible to obtain their data at earlier time points in an extractable form. Four studies used a self-report quality of life measure with participants with dementia (Buschert 2011; Chapman 2004; Coen 2011; Spector 2003), but again the Chapman 2004 data was not in a useable form. Five studies used a self-report depression measure (four making use of a version of the Geriatric Depression Scale: Yesavage 1983), and four studies used a depression or anxiety scale completed from carer reports as well as from interviews with the participants (such as the Cornell Scale for Depression in Dementia: Alexopoulos 1988). A variety of scales have been used to evaluate behaviour, with activities of daily living (ADL) scales used in four studies, general behaviour ratings in seven studies and problem behaviour scales used in three studies. Family caregiver outcome measures were used in three studies (Bottino 2005; Onder 2005; Spector 2001). A full list of the outcome measures used in the included studies can be found in the table Characteristics of included studies'. 7 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Risk of bias in included studies Details for each study are provided in the 'Characteristics of included studies' table. Allocation (selection bias) For a study to be included in this review, the review authors had to be satisfied that random allocation to treatment conditions had been used. To ascertain this, in several cases it was necessary to seek further information from the study authors (for example Baldelli 1993a; Baldelli 2002; Ferrario 1991; Requena 2006). Remote or computerised randomisation was only used in four of the most recent studies (Bottino 2005; Chapman 2004; Buschert 2011). Earlier studies described drawing names from a hat or a sealed container, where it was possible to obtain details of the randomisation procedure. Blinding (performance bias and detection bias) Performance bias With psychological interventions, unlike drug trials, it is impossible to totally blind participants and staff to treatment. Participants will often be aware that they are being treated preferentially and the staff involved may have different expectations of treatment groups. There may also be 'contamination' between groups in terms of group sessions not being held in separate rooms and staff bringing ideas from one group to another, so that control participants receive elements of cognitive stimulation. The latter effect would be reduced with clear therapeutic protocols, the existence of which was not clear in most study reports. In relation to contamination, Wallis 1983 and Baines 1987 both stated that the staff were unaware of the allocation of participants to groups, as they were removed from the ward setting for treatment, and several other studies described the groups being run in a separate or specific room (for example Ferrario 1991; Spector 2001; Spector 2003; Woods 1979). Detection bias Most studies took steps to ensure that at least part of the assessment of outcomes was carried out by assessors blind to treatment allocation. Only three studies (Baldelli 1993a; Baldelli 2002; Ferrario 1991) did not report blinding of assessors. Of course, even independent assessors may be given clues from participants during the assessments, but this was not reported as an issue in the studies reviewed here. Using independent assessors works well for evaluating changes in cognition or self-reported mood, well-being and quality of life. Ratings of day-to-day behaviour and function are typically carried out by care staff, who may be more difficult to keep blind to group allocation, unless the group sessions were carried out in a separate location to which all participants were taken. Incomplete outcome data (attrition bias) Only two studies described following an intention-to-treat analysis plan (Chapman 2004; Spector 2003). In contrast, Breuil 1994 stated that "All those who for any reason did not attend all evaluation and training sessions were eliminated", with five participants excluded on this basis (three from the cognitive stimulation group). All studies reported data on attrition. Given the nature of the condition and the age of the participants, attrition in several studies was remarkably small, with zero attrition recorded in six studies (Baines 1987; Baldelli 1993a; Baldelli 2002; Bottino 2005; Buschert 2011; Coen 2011), out of 180 participants. The largest attrition rate was reported by Wallis 1983 where there was 39% attrition in the group of participants with dementia. In this study patients who attended less than 20% of the group sessions were eliminated from the study. Requena 2006 reported 32% attrition but this was over a two year period. The two largest studies had rates of 19% (Onder 2005) and 17% (Spector 2003) over periods of six months and two months respectively. Other potential sources of bias The absence of detailed treatment protocols raised queries regarding the extent to which the cognitive stimulation was delivered as intended (having noted that there may have been differences in emphases between studies in any case). Several studies noted that staff received training or supervision, or both, in running the groups and, from an early study, Woods 1979 stated in a personal communication that "A sample of sessions were tape-recorded and rated to ensure compliance with the therapeutic protocol". More recently, Chapman 2004 described weekly meetings to ensure their treatment programme was implemented as designed. Subgroups were led by a licensed speech-language pathologist and three master's level speech-language pathology students; all underwent two hour training before the groups started and weekly meetings were held to ensure that the programme was implemented as designed. Onder 2005 described how family caregivers were trained by a multi-disciplinary team and given a manual and specific schedules for each session. No records were made, however, of how often caregivers did deliver the sessions, or how closely the manual was followed. Effects of interventions For meta-analyses we used RevMan 5.1. Cognition (See Figure 1) For the overall evaluation of the effects of cognitive stimulation on cognitive function, all 14 RCTs which included useable data immediately post-treatment were included, including a total of 658 people with dementia of whom 377 received cognitive stimulation and 281 received no treatment or a placebo treatment. As most studies included more than one measure of cognitive function, this analysis was conducted on the most extensive assessment included. For seven studies this was the ADAS-Cog, and for two each it was the MMSE and CAPE Information/Orientation scales. The overall effect size, the 8 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia standardised mean difference (SMD) was 0.41 (95% CI 0.25 to 0.57). This was a statistically significant finding (Z = 5.04, P < 0.00001). For the seven studies, including 434 participants, using the ADAS-Cog as an outcome measure (Figure 2), the mean difference between the cognitive stimulation and control groups was 2.27 points (95% CI 0.99 to 3.55), a statistically significant difference (Z = 3.48, P < 0.0005). In total, 10 studies involving 600 participants used the MMSE (Figure 3). The overall mean difference was 1.74 points (95% CI 1.13 to 2.36). Again, this was a statistically significant difference (Z = 5.57, P < 0.00001). These analyses were strongly influenced by two of the more recent studies, Spector 2003 and Onder 2005, which were relatively large and had smaller confidence intervals around their reported mean differences. Five of the older studies involving 81 participants used other measures of information or orientation (Figure 4); here the SMD was no smaller (SMD 0.45, 95% CI -0.01 to 0.90) and was just statistically significant despite the much smaller numbers involved (Z = 1.93, P = 0.05). The largest effect sizes were seen at the 12 month time point in the Requena 2006 study (SMD 0.70 on ADAS-Cog) and the Baldelli 1993a study (SMD 0.99 on MMSE), both of which offered above average durations of exposure to cognitive stimulation. However, Breuil 1994, which offered only 10 hours exposure, also had an above average effect size (0.63 on global cognitive score) and other studies with longer exposure (for example Ferrario 1991) had below average effect sizes. The 24 month data from Requena 2006 indicated that effect sizes appeared to be maintained through continued exposure (ADAS-Cog SMD 0.66: MD 11.94 points, 95% CI -0.97 to 24.85; MMSE SMD 0.56: MD 5.99 points, 95% CI -1.58 to 13.56). However, these effects require replication as the confidence intervals were broad and crossed zero. Communication and social interaction (See Figure 5) Four studies, involving 223 participants, included staff ratings of the person's communication and social interaction (outside of the cognitive stimulation group), three using the Holden Communication Scale. The overall effect size (SMD) was 0.44 (95% CI 0.17 to 0.71) with participants in the cognitive stimulation groups showing a significant improvement in this area (Z = 3.15, P = 0.002). Spector 2003 was the most influential study in this analysis, although the effect was not reported as significant in the primary study report. Well-being and quality of life (See Figure 6) Four studies, involving 219 participants, included relevant self-report measures. Baines 1987 used the Life Satisfaction Index and Spector 2003, Buschert 2011 and Coen 2011 used the QoL-AD. The meta-analysis indicated that cognitive stimulation was associated with a significant benefit to well-being and quality of life compared with no treatment (SMD 0.38, 95% CI 0.11 to 0.65) (Z = 2.76, P = 0.006). The Spector 2003 findings were again a major influence. Mood (See Figure 7; Figure 8) Five studies, involving 201 participants, used a self-report measure of mood (the Geriatric Depression Scale or the MADRS). Cognitive stimulation was not associated with a clear improvement in mood across these studies. The SMD was 0.22 (95% CI -0.09 to 0.53) (Z = 1.42, P = 0.16). Staff ratings of mood and anxiety similarly did not show any benefit from cognitive stimulation. Four studies, involving 239 participants, contributed to this analysis, two using the Cornell Scale for Depression in Dementia, a third using a subscale of the MOSES scale and the fourth the Rating of Anxiety in dementia. The SMD was close to zero in this domain (SMD 0.05, 95% CI -0.21 to 0.31). Behaviour (See Figure 9; Figure 10; Figure 11) Three separate meta-analyses were conducted in this domain. One focused on activities of daily living (ADL) and basic selfcare skills; a second focused on behaviours seen as a problem, such as irritability, being demanding and difficult. The third included 'general' behaviour rating scales, which may include some of the previous two aspects, together with some higher level daily living skills. Four studies, involving 160 participants, used ADL scales. There was no benefit identified with cognitive stimulation (SMD 0.21, 95% CI -0.05 to 0.47) (Z = 1.56, P = 0.12). Three studies, including 166 participants, used scales evaluating behaviour problems. Again there was no difference related to cognitive stimulation (SMD -0.14, 95% CI 0.44 to 0.17) (Z = 0.86, P = 0.39). General behaviour rating scales showed a similar picture, with no difference emerging. Eight studies, including 416 participants, reported data on relevant scales (SMD 0.13, 95% CI -0.07 to 0.32) (Z = 1.30, P = 0.20). Caregiver outcomes (See Figure 12) Three studies reported on outcomes for family caregivers. The largest of these (Onder 2005) taught family caregivers to deliver the cognitive stimulation, so the effects on caregivers were especially pertinent for that study. The effect sizes for anxiety, depression and caregiver burden were all close to zero (SMDs 0.11, 0.04, -0.03 respectively), with confidence intervals crossing zero indicating no differences between the caregivers in the cognitive stimulation condition and those in the control conditions. Only one study (Baines 1987) reported outcomes for care staff, so no meta-analysis was possible. The results of this study indicated a significant increase in staff knowledge about residents participating in the cognitive stimulation intervention 9 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia compared with knowledge of residents in the control condition. Follow-up The analyses for assessments carried out after the intervention had been completed were reported in two groups: Baines 1987 and Wallis 1983 had a one month follow-up, and Baldelli 1993a a three month follow-up on certain measures. These three studies represent a short term follow-up; whereas Chapman 2004 reported useable data only from a 10 month followup, a much longer period in the context of the progression of dementia. For cognitive measures (Figure 13), the three older studies with short term follow-up reported data for 52 participants. The significant advantage for cognitive stimulation on cognitive measures seen immediately post-treatment remained at this point (SMD 0.57, 95% CI 0.01 to 1.14) (Z = 2.00, P = 0.05). For the 54 participants included by Chapman 2004, there was no significant effect on either the MMSE (SMD 0.18) or the ADAS-Cog (SMD 0.12) at the 10 month follow-up. For self-report well-being and quality of life measures (Figure 14), the only short term follow-up data came from Baines 1987, with only 10 participants, and showed no differences. The longer term follow-up from Chapman 2004 showed a SMD of 0.34 for the QoL-AD measure but this difference was not significant. No measures of mood were included in the studies reporting follow-up. For general behaviour rating scales (Figure 15), two studies reported short term follow-up, with 29 participants. The SMD of 0.44 was not significant. Similarly at 10 months the scale used by Chapman 2004 showed a non-significant SMD of 0.43. A similar picture appeared in relation to problem behaviour (Figure 16), where there was only one small study in the short term follow-up group (Baines 1987) and the longer term follow-up reported by Chapman 2004 did not show a significant difference on either the Neuropsychiatric Inventory (NPI) severity score (SMD 0.29) or the caregiver distress score related to the problem behaviour (SMD 0.41). Finally, there were no differences in measures of communication and interaction (Figure 17) at either short term (Baines 1987) or longer term follow-up (Chapman 2004). Discussion In total, 15 RCTs with a total of 718 participants (407 receiving cognitive stimulation, 311 in control groups) met the inclusion criteria for the meta-analyses. The most striking finding reflects the effects of cognitive stimulation on performance in tests of cognitive function. The results of the meta-analyses reported here indicate that cognitive stimulation programmes for dementia have a significant positive effect on cognition, which is evident in the post-2000 studies included in this review as well as in the older studies which were included in our previous review of Reality Orientation. This is perhaps the most consistent finding in the literature on psychological interventions with people with dementia. The studies included here came from a variety of countries and contexts, from France, the UK, Italy, Spain and Brazil; from hospital, care home, nursing home, day centre and outpatient settings; and administered in groups by staff or volunteers, or individually by family caregivers. The extent to which these changes in cognitive function are clinically important has not been generally addressed. An average benefit of 1.74 points on the MMSE or 2.27 points on the ADAS-Cog can be taken to indicate a slowing down of the rate of decline, which has been estimated, in mild to moderate dementia, to be between 2 and 4 points on the MMSE per annum (Mohs 2000). In relation to the number needed to treat (NNT) for one more participant in the treatment group than in the control group to benefit by a certain amount, Spector 2003 and Onder 2005 provide some evidence. For an improvement of 4 or more points on the ADAS-Cog, Spector 2003 calculated an NNT of 6, and Onder 2005 14, figures broadly comparable to those seen in trials of the ACHEIs. However, as has been pointed out repeatedly over the years (Woods 2006), changes in cognition are not sufficient to justify an extensive programme of intervention, unless they are accompanied by other changes, in behaviour and well-being. Here there are two positive findings. Firstly, results from four RCTs (with 223 participants) indicated that positive changes in communication and social interaction were evident in staff ratings outside the context of the cognitive stimulation group sessions. Secondly, results from four RCTs (219 participants) identified a benefit on quality of life and well-being associated with cognitive stimulation. In contrast, there was no indication that cognitive stimulation was associated with changes in mood, whether self-rated or rated by staff, or in behaviour including activities of daily living and self-care and problem behaviour. It is notable that in general there is much less evidence available regarding these domains compared with that available for changes in cognition. No benefits to family caregivers were identified in terms of mood or caregiver burden. However, it is important to note that in the study where the responsibility for delivering the cognitive stimulation fell on famiy caregivers (Onder 2005), this additional task did not appear to add to their stress and strain. One study showed an improvement in care home staff knowledge of residents following participation in cognitive stimulation. There is little evidence available on the cost-effectiveness of cognitive stimulation (CST). Knapp 2006 reported costeffectiveness acceptability curves for cognition and quality of life from the Spector 2003 trial and conclude that, for both outcomes, 'under reasonable assumptions, there is a high probability that CST is more cost-effective than treatment as usual'. These findings all relate to the assessment point immediately after the treatment period has been completed. Only four relatively small studies reported data on whether any changes were maintained after a period of follow-up without further intervention. Here, again, cognition stands out with three RCTs showing a positive effect evident at a follow-up of one to three months after the intervention; this was not evident in the one RCT reporting a 10 month follow-up. Other follow-up data were limited in their extent and scope but there were no indications of benefits in the areas of behaviour and well-being that 10 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia were evaluated. A key area of difference between studies relates to the duration and frequency of the cognitive stimulation offered, leading to a wide variation in the 'dosage' of cognitive stimulation received, as indicated by the total number of hours of stimulation offered. As pointed out previously, there does not appear to be a clear relationship between dosage and the effect size on cognitive function in the various studies; the Spearman's correlation was 0.25 (N = 14, P = 0.392). It is difficult to ascertain whether the frequency of sessions per week makes a difference as the study with the largest effect sizes (Requena 2006) has five 45-minute sessions per week and has the longest duration. As the duration increases, the anticipated decline associated with dementia should tend to reduce the effects of cognitive stimulation, and so a simple linear relationship is unlikely to hold. One study (Requena 2006) continued cognitive stimulation for two years. The decision was made to include one year data from this study in the primary meta-analyses reported here, to reduce the impact of attrition. However, the effects on cognition and self-reported mood appeared to be sustained over a further one year period, although the effects were not statistically significant in our analyses. There is a need for further research on maintenance cognitive stimulation, looking at whether lower-intensity input maintains gains from an initial period of cognitive stimulation (Aguirre 2010). In five of the included studies all of the participants were prescribed ACHEI medication. For the four of these RCTs providing post-treatment data, the additional effect of cognitive stimulation over and above the medication was 3.18 points on the ADAS-Cog, compared with the overall finding (from seven RCTs) of 2.27 points. This supports the proposition that cognitive stimulation is effective irrespective of whether or not ACHEIs are prescribed, and any effects are in addition to those associated with the medication. One study (Requena 2006) reported comparative results from a control group who received neither cognitive stimulation nor an ACHEI, but this was not a randomly allocated condition so that the additive effects of these two treatment approaches were not established compared with no treatment. Such a study would perhaps not now be seen as ethical in a context where ACHEIs are seen as standard treatment for Alzheimer-type dementia. In general, it appears that outcome measures rated by staff are less likely to indicate positive change than those that are completed by the person with dementia directly with an assessor blinded to the treatment received. In a care home or hospital context, it is well known that achieving consistent staff ratings in research studies such as these is a major challenge. It can often be impossible to have the same staff member rate the person at each time point due to staff turn-over and sickness. There can be a 'drift' in ratings over time, even with the same rater. The observation period and opportunities for observation may vary over time. Maintaining staff blind to treatment allocation is more challenging than for an assessor who only visits the facility to carry out the assessments. The one area of staff-rated behaviour showing change in this review relates to communication and social interaction, an area that has not been highlighted in previous reviews of this type of intervention. Given the social emphasis of cognitive stimulation groups, this is an area of behaviour that is most closely linked to the content of the intervention. Indeed, Spector 2010 have demonstrated that in their study language function was an area of specific cognitive improvement. Inevitably where the control condition is 'no treatment', the question is raised as to whether any benefits associated with the treatment arise from non-specific effects such as meeting as a group, socialising, increased attention and so on. A few early studies did include an 'attention' control group (Woods 1979) or diversional occupational therapy (Wallis 1983) and one study offered physical rehabilitation sessions to both groups (Baldelli 2002). In the Requena 2006 study where stimulation materials were presented on a TV screen to the group, control participants watched TV elsewhere. Although we cannot provide a definitive conclusion on this matter, our results did not indicate any effects of these different control conditions on outcome. In a mediation analysis, Woods 2006 demonstrated that in the Spector 2003 RCT the improvements in quality of life were mediated by improvements in cognition, suggesting that it is the cognitive focus of the cognitive stimulation therapy programmes (rather than merely the social contact and attention) which lead to improved well-being. Further work is required to explore the relationship between changes in cognition and the changes in well-being and quality of life and communication that are emerging from the current review. The quality of the included studies is variable, and generally low, with lack of clarity regarding randomisation procedures being evident in around half the studies (including some of the more recent studies). CONSORT type diagrams depicting the flow of participants through the trial are provided by four of the recent studies (Bottino 2005; Buschert 2011; Onder 2005; Spector 2003) and, along with remote, independent randomisation, will be a minimum expectation in future trials. The number of participants included has increased markedly from an average of 23 per study prior to 2000 to 64 in the nine more recent studies. Larger sample sizes will necessitate multi-centre trials, for example Spector 2003 recruited from 23 centres. This will have implications for analyses, with cluster effects within a centre, even though participants are individually randomised. Spector 2003 accordingly included centre as a covariate in their analyses. Studies do not yet appear to have taken account of clustering effects arising from a group intervention. This occurs where changes in group members are not entirely independent. Group leaders often report that some groups seem to work much better than others for example. Intention-to-treat analyses were only described by two (recent) studies, although in most cases details of attrition were reported. The need for assessors to be blind to treatment allocation is widely recognised and attempted in most studies. More attention may need to be given in future studies to demonstrating the extent to which the cognitive stimulation is delivered as planned. Well-developed treatment manuals will help with assuring the replicability of the intervention. In general, there is a clear improvement in overall quality of the included studies over time, for example in the more consistent information given regarding the diagnosis of participants and the criteria used, as well as the use of consistent outcome measures and larger sample size. The studies included in the review utilised therapists with a variety of backgrounds, experience and training. They included volunteers, family caregivers, speech and language therapists, occupational therapists, nurses, care workers and research staff. There are no indications from this review of the amount or type of training required to deliver cognitive stimulation, although there is broad agreement that whilst training is needed the therapist does not need a professional qualification. 11 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Given the concerns regarding RO being delivered in a mechanical, dehumanising fashion (Dietch 1989), training and supervision in person-centred care would be seen as a prerequisite for delivery of cognitive stimulation. In this review, there were no reported side effects or adverse effects of any of the cognitive stimulation interventions. Attrition was due to expected reasons in studies of this nature: illness, death, transfer to another facility and occasional refusal to complete follow-up assessments. With only one study utilising an individual approach, in contrast to offering group cognitive stimulation, it is not possible to draw on any evidence on the strengths and benefits of these different treatment modalities. The range of severity of dementia experienced by patients included in the studies reviewed here ranged from mild to moderate. There was no indication that the two studies with the lowest mean MMSE scores of participants (Spector 2001; Spector 2003) had any different effects than those with more mildly impaired participants. Most studies included a mixture of participants with mild and moderate dementia. Onder 2005 indicated that there was no differential benefit to either group, with no significant interaction between severity of dementia and treatment on change in either MMSE or ADAS-Cog scores. More work may be needed to define if there are people with dementia, or subgroups, who are more or less likely to benefit from a cognitive stimulation intervention. No analyses have been attempted here seeking to establish whether different subtypes of the dementias show specific responses to cognitive stimulation. One study (Baldelli 2002) included only participants who had had a cerebrovascular accident (CVA), although less than half the participants had a diagnosis of vascular dementia. Most recent studies have included those who are receiving AChEI medication, with a probable Alzheimer's diagnosis, whereas Spector 2003 included all types of dementia. Given that the average age of participants across the review was almost 80 years (over 85 in the Spector 2003 study), it is highly likely that neurodegenerative and vascular changes are co-occurring in the majority of participants, and so the distinction may be of limited pragmatic utility. The findings of this review are broadly in line with other more wide-ranging reviews of non-pharmacological interventions in dementia. Livingston 2005 gives a relatively strong recommendation for cognitive stimulation in relation to its effects on neuropsychiatric symptoms, including mood, but notes some inconsistencies in the evidence on these outcomes. Sitzer 2006 included 17 studies in their meta-analysis of 'cognitive training' in Alzheimer’s disease, four of which met our definition of 'cognitive stimulation' and three of which are included in this review. Sitzer 2006 concludes that there is a medium effect size (0.47) across all types of 'training' over the whole range of outcome measures, making 'cognitive training' a promising intervention. However, it is noteworthy that general stimulation techniques were prominent in four of the five trials reporting the most beneficial results, with the review describing these as 'restorative strategies'. Olazaran 2010 reviewed 179 RCTs across 26 categories of non-pharmacological interventions. They concluded that there was 'Grade B' evidence (consistent evidence from lower quality RCTs) for cognitive stimulation in relation to cognition, behaviour and psychological well-being. They did not identify any 'Grade A' evidence (consistent evidence from high quality RCTs) for any interventions with people with dementia, although some caregiver interventions did reach this level. The 2011 World Alzheimer's Report (Prince 2011) concludes, from a wide-ranging systematic review, "We found strong evidence (multiple RCTs) that acetylcholinesterase inhibitors (for cognitive function, functional impairment), and cognitive stimulation (for cognitive function) are effective interventions in mild dementia" and makes the following recommendation in relation to interventions for early-stage dementia: "Acetylcholinesterase inhibitors and cognitive stimulation may enhance cognitive function in people with mild Alzheimer’s disease, and these interventions should therefore be routinely offered." Finally, consideration should be given to the possibility of publication bias in this domain. By reviewing only the studies on cognitive stimulation that have been published in peer-reviewed journals, it must be acknowledged that these could represent a biased sample of the studies undertaken world-wide on this topic. In many fields of endeavour, trials that are not successful (that is do not produce the expected positive findings) are less likely to be published. This may be especially the case with smaller trials. The welcome trend to pre-registration of trials, and the publication of trial protocols, makes this less likely to occur in the future in relation to larger, well-funded trials. The meta-analyses here have been influenced strongly by the larger trials included (such as Spector 2003 and Onder 2005), and a funnel plot of the cognition outcome appears reasonably symmetrical (Figure 18) suggesting that possible publication bias is not a strong factor for this outcome at least. Authors' conclusions Implications for practice The evidence base for the effectiveness of cognitive stimulation therapy for dementia in relation to cognitive function has been consistently demonstrated, with small changes reported in multiple trials on commonly used brief measures of cognitive function; adverse effects have not been reported. There is now evidence from a small number of studies that cognitive stimulation may also be associated with improvements in quality of life and communication. These benefits are over and above any medication effects. This review is consistent with the NICE-SCIE 2006 Guideline recommendation that all people with mild to moderate dementia should have the opportunity to participate in cognitive stimulation groups, irrespective of whether or not they are receiving acetylcholinesterase inhibiting medication (ACHEIs). This recommendation was recently reinforced by the World Alzheimer's Report (Prince 2011). Although more research is needed, results from one study suggest that continuing involvement in cognitive stimulation may be beneficial. Implications for research There are a number of areas, relating to both theory and practice, where further research is required. Now that its effects are becoming better established, the theoretical basis of cognitive stimulation would benefit from fuller investigation. This would involve studying cognitive changes both in relationship to neural processes and pathways; and their linkage, if any, with 12 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia outcomes such as mood, quality of life, day-to-day function and behaviour. There is a clear need for more randomised controlled trials of cognitive stimulation exploring the long term benefits of this intervention. The effects of severity of dementia and different modalities (for example group versus with caregiver) need to be systematically evaluated. These RCTs may be of particular value if used in conjunction with more qualitative studies in order to understand the relationships between the different outcome measures. The clinical meaningfulness of any benefits needs to be examined, particularly in relation to the impact of any cognitive changes. Are there benefits in terms of increased participation in activities of importance to the person, increased social inclusion or of individual goals being attained? Only one cost-effectiveness study has been identified to date and this gap should be addressed, particularly in relation to clinically meaningful benefits. Finally, the implementation of cognitive stimulation in real-life settings needs to be addressed. The key issue here is whether the results obtained by those who attend brief training in the methods or make use of one of the treatment manuals that have been developed, or both, are comparable to those obtained in the context of research studies and RCTs. Acknowledgements Maintenance Cognitive Stimulation Programme (ISRCTN26286067) is part of the Support at Home - Interventions to Enhance Life in Dementia (SHIELD) project (Application No RP-PG-0606-1083) which is funded by the NIHR Programme Grants for Applied research funding scheme. The grant holders are Professors Orrell (UCL), Woods (Bangor), Challis (Manchester), Moniz-Cook (Hull), Russell (Swansea), Knapp (LSE) and Dr Charlesworth (UCL). The views and opinions expressed in this review are those of the authors and do not necessarily reflect those of the Department of Health/NIHR. The authors wish to thank Joanne Knowles for acting as consumer reviewer for this review. Contributions of authors BW: correspondence; drafting review versions; search for trials; selection of trials; extraction of data; entry of data; data analysis; interpretation of data analyses; updating review. AS: selection of trials; extraction of data; interpretation of data analyses; updating review. EA: search for trials; obtaining copies of trial reports; entry of data; data analysis; interpretation of data analyses. MO: selection of trials; extraction of data; interpretation of data analyses; updating review. Declarations of interest The authors have produced various training materials in dementia care, including cognitive stimulation therapy manuals, in order to disseminate research findings to care workers and others. Royalties for the manuals are received by the Dementia Services Development Centre Wales. AS receives fees for providing training in cognitive stimulation approaches. Differences between protocol and review Published notes This review replaces the review of Reality Orientation for dementia (Spector A, Orrell M, Davies S, Woods B. Reality orientation for dementia. The Cochrane Database of Systematic Reviews 2000, Issue 3. Art. No.: CD001119. DOI: 10.1002/14651858.CD001119). Characteristics of studies Characteristics of included studies Baines 1987 13 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Cross-over design: (only results from first phase are included in this review) Treatment 1: 4 weeks; 4 week 'wash-out' period'; Treatment 2: 4 weeks Participants N=15 (14F, 1M) 'Moderate to severe Impairment of cognitive functioning' Mean age=81.5 (range 72-90) Living in care home Interventions RO Reminiscence Treatment as usual Outcomes Cognitive: Information/Orientation & Mental Ability (CAPE) Behaviour: Behavioural Rating Scale (CAPE) Well being: Life Satisfaction Index; Problem Behaviour Rating Scale Communication : Holden Communication Scale 4 week follow-up data available Staff completed 'Personal Information Questionnaire', evaluating staff knowledge of residents Notes Treatment duration 30 minute sessions, 5 days a week for 4 weeks Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Unclear risk Support for judgement No details of the randomisation method used reported in the paper. Allocation concealment (selection bias) Unclear risk No details of the randomisation method used reported in the paper. Blinding (performance bias and detection bias) Low risk RO groups held in separate areas; Blinding of outcome assessment (detection bias) Low risk Incomplete outcome data (attrition bias) Low risk No attrition at follow-up assessment Other bias Unclear risk Cognitive assessments made by an independent psychologist; other ratings made by staff not involved in the therapy groups. Baldelli 1993a 14 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N=23 (23F/0M) Alzheimer’s (SDAT) Mean MMSE 20.6 (sd 4.9) Mean age 84.5 (range 75-94) All resident in institution Interventions RO Treatment at usual Outcomes Cognition: MMSE; Berg Orientation Scale Mood: GDS-30 ADL: Stewart ADL scale 3 month follow-up data on cognitive measures Notes 60 minutes, 3 times a week for 3 months Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Unclear risk Support for judgement Stated by e-mail that their trials were randomised (with no detail of the methods used) Stated by e-mail that their trials were randomised (with no detail of the methods used) Allocation concealment (selection bias) Unclear risk Blinding (performance bias and detection bias) Unclear risk No evidence of blinding in the paper Blinding of outcome assessment (detection bias) Unclear risk No details of who assessors were Incomplete outcome data (attrition bias) Low risk Other bias Unclear risk Zero attrition at 3 month post-treatment assessment; no attrition reported at follow-up 3 months later Baldelli 2002 15 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N= 87 (61F/26M) 'Degenerative senile dementia of the Alzheimer’s type (SDAT)' (N=46) and “vascular multi-infarct dementia” (N=41) Mean MMSE 20.7 (sd 3.0) Mean age 80.0 (range 65-97) Resident in sub-acute care nursing home All had at least elementary schooling 'All had comorbid conditions consisting of vascular accidents with acute motor deficits of recent onset' Interventions RO + physical therapy programme. Physical therapy programme only Outcomes Cognition: MMSE Mood: Geriatric Depression Scale (GDS 30) ADL: Barthel Notes 60 minutes, 5 days per week for one month Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Unclear risk Support for judgement Stated by e-mail that their trials were randomised (with no detail of the methods used) Stated by e-mail that their trials were randomised (with no detail of the methods used) Allocation concealment (selection bias) Unclear risk Blinding (performance bias and detection bias) Unclear risk No evidence of blinding in the paper Blinding of outcome assessment (detection bias) Unclear risk No details of assessors given Incomplete outcome data (attrition bias) Low risk No attrition reported Other bias Unclear risk Bottino 2005 16 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N=13 (9F / 4M) 'Mildly impaired probable Alzheimer’s diagnosis' All participants taking rivastigmine 6-12mg/day for 2 months Mean MMSE 22.31 (sd 3.61; range 16-28) Age 73.7 (range 62-83) Out-patients Interventions 'cognitive rehabilitation' plus rivastigmine; carers attended a support group at same time Treatment as usual: rivastigmine plus 30 minute monthly consultation with doctor in relation to medication Outcomes Participants: Cognition: MMSE; ADAS-Cog, ADL (rated by carer) Carers' mood: Hamilton Anxiety and Montgomery-Asberg Depression Rating Scales Notes 90 minutes, once a week, for 5 months Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Low risk Support for judgement Randomised blocks design, randomly allocated to either group by telephone by an assessor blind to the patient group Randomised blocks design, randomly allocated to either group by telephone by an assessor blind to the patient group Allocation concealment (selection bias) Low risk Blinding (performance bias and detection bias) Blinding of outcome assessment (detection bias) Unclear risk Low risk Assessment made by assessors blinded to group allocation Incomplete outcome data (attrition bias) Low risk No attrition reported Other bias Unclear risk Breuil 1994 17 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N= 61 (37F / 24M) Diagnosis of dementia (DSM-III) (90% have Alzheimer's Disease) Age 77.1 (range 61-93) Mean MMSE 21.5 (range 9-29) Out-patients Interventions Cognitive stimulation Treatment as usual Outcomes Cognition: MMSE, CERAD ADl: ECA scale rated by family members Notes 60 minutes, 2 times a week, for 5 weeks Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Unclear risk Allocation concealment (selection bias) Unclear risk Blinding (performance bias and detection bias) Blinding of outcome assessment (detection bias) Unclear risk Incomplete outcome data (attrition bias) Low risk Other bias Unclear risk Support for judgement No details of randomisation reported No details of randomisation reported Low risk Cognitive assessments made by an assessor blind to group allocation; ADL assessment open Five patients excluded as did not attend all training and evaluation sessions (3 from treatment group, 2 from controls) Buschert 2011 18 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N=39 24 amnestic MCI; 15 mild Alzheimer's disease (only data on Alzheimer's patients reported in this review) 8F/7M Mean MMSE 24.9 (sd 1.6; range 22-27) All on stable doses of AChEIs or memantine Age 75.9 (sd 8.1) Out-patients Interventions Outcomes Multi-component cognitive group intervention - for AD group emphasis on cognitive stimulation (for MCI group more emphasis on cognitive training); Control group had pencil and paper exercises for self-study and monthly meetings Cognition: MMSE; ADAS-Cog, Trail Making Test, RBANS story memory & recall Quality of life: QoL-AD Mood: Montgomery Asberg Depression Rating Scale Notes 2 hours, once a week for 6 months (20 sessions) Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Low risk Allocation concealment (selection bias) Low risk Blinding (performance bias and detection bias) Blinding of outcome assessment (detection bias) Unclear risk Incomplete outcome data (attrition bias) Low risk Other bias Support for judgement Blocked randomisation procedure; participants pooled in pairs with respect to age, gender, education and ApoE genotype, then randomly assigned pairs to intervention or control using a computerised random number generator. Blocked randomisation procedure Unclear risk Assessors blind to group allocation No attrition Unclear risk Chapman 2004 19 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N= 54 (29F / 25M) probable AD, on stable dose of donepezil for at least 3 months Mean MMSE 20.87 (sd 3.55, range 12-28) Age 76.4 (range 54-91) Living at home initially Interventions cognitive stimulation + donepezil Donepezil only Outcomes Cognition: MMSE; ADAS-Cog; ADL: Texas Functional Living Scale Behavioural problems: NPI - Irritability and Apathy Quality of Life: QoL-AD Global functioning: CBIC Verbalisation: Composite discourse score Carer distress - derived from the NPI 10 month follow up data available Notes 90 minutes, once a week, for 8 weeks Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Low risk Support for judgement Independent randomisation, using an SAS procedure Allocation concealment (selection bias) Low risk Remote telephone randomisation Blinding (performance bias and detection bias) Unclear risk Carer ratings not blind to allocation Blinding of outcome assessment (detection bias) Low risk Incomplete outcome data (attrition bias) Low risk Other bias Low risk All raters underwent extensive training; assessors blinded to group allocation Intention to treat analysis used. 24% attrition rate at end of study Programme led by trained speech therapist, weekly meetings held in order to ensure the programme is implemented as designed. Coen 2011 20 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N = 27 (14F/13M) Dementia - MMSE 10-23 MMSE: 16.9 (sd 5.0) Age: 79.8 (sd 5.6) Groups ran in 2 long term care facilities and a private nursing home Interventions Cognitive stimulation No treatment Outcomes Cognition: MMSE; ADAS-Cog Quality of life: QoL-AD Communication: Holden Communication Scale Mood: Geriatric Depression Scale (14 item); RAID (Rating of Anxiety in Dementia) Behaviour: Behaviour Rating Scale (CAPE) Clinical Dementia Rating (sum of boxes) Notes 45 minutes, 2 times a week for 7 weeks Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Unclear risk Support for judgement Stated that participants were randomly assigned. Author confirms computerised randomisation and random number tables were used Allocation concealment (selection bias) Unclear risk Stated that participants were randomly assigned. Author confirms computerised randomisation and random number tables were used Blinding (performance bias and detection bias) Unclear risk Staff running groups also involved in other activities, involving control participants Blinding of outcome assessment (detection bias) Unclear risk Tests administered by staff blind to group membership. Not clear if staff ratings were made by staff who wre blinded Incomplete outcome data (attrition bias) Unclear risk No attrition Other bias Unclear risk Sessions run by occupational therapists and activity coordinator Ferrario 1991 21 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N=19 (8F / 11M) elderly patients with cognitive disturbances MMSE range 18-25 Age 82.5 (sd 5.2) Resident in institution Interventions RO No treatment Outcomes Cognition: CAPE I/O Self-care: MOSES Behaviour problems: MOSES - irritable, withdrawn Mood: MOSES Notes 60 minutes, 5 times a week, for 21 weeks Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Unclear risk Allocation concealment (selection bias) Unclear risk Blinding (performance bias and detection bias) Unclear risk Blinding of outcome assessment (detection bias) Unclear risk Incomplete outcome data (attrition bias) Low risk Other bias Low risk Support for judgement Stated by e-mail that the trial was randomised (with no detail of the methods used) Stated by e-mail that the trial was randomised (with no detail of the methods used) No information given in relation to where groups were held, but RO materials were in evidence on the ward - may have been accessed by control participants? MOSES completed by nursing staff - not clear if raters were blind 2 dropouts (/21). 1 in each group (pneumonia and stroke). (Information provided by the author) RO administered by volunteers trained by physicians and psychologist Onder 2005 22 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N = 156 (113F/ 43M) Probable Alzheimer's Disease, on Donepezil for at least 3 months MMSE 20.1 (sd 3.1) Age 75.8 (sd 7.1) Living at home Interventions RO + Donepezil Donepezil only Outcomes Cognition:MMSE; ADAS-Cog ADL: Barthel; IADL Behaviour problems: NPI Family caregiver outcomes: Hamilton anxiety and depression scales; Caregiver Burden Inventory; SF-36 Notes 30 minutes, 3 times a week, for 25 weeks; plus informal contacts 2 or 3 times a day Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Low risk Support for judgement Computerised block randomisation process Computerised block randomisation process Allocation concealment (selection bias) Low risk Blinding (performance bias and detection bias) Blinding of outcome assessment (detection bias) Unclear risk Low risk Assessment made by blind assessors Incomplete outcome data (attrition bias) Low risk Attrition data reported: 9 from RO group, 10 from control group i.e. 19% Other bias Low risk Family caregivers trained by a multi-disciplinary team Requena 2006 23 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N = 86 (61F / 25M) Alzheimer-type dementia (severe dementia excluded) MMSE 21.3 Age 77 (sd 7.5) Attending daycare centre Interventions 1) Cognitive stimulation + Donepezil 2) Donepezil only 3) Cognitive stimulation only 4) No treatment Outcomes Cognition: MMSE, ADAS-Cog Mood: GDS-30 12 month and 24 month data reported Notes 45 minutes, 5 times a week for 24 months 'No treatment' group were not part of the randomisation process. Comparison of interest to this review is cognitive stimulation + donepezil v donepezil alone Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Unclear risk Allocation concealment (selection bias) Unclear risk Blinding (performance bias and detection bias) Low risk Blinding of outcome assessment (detection bias) Low risk Incomplete outcome data (attrition bias) Low risk Other bias Unclear risk Support for judgement Randomisation by registration order: 'subjects were randomly distributed in groups at the time they arrived at the Centre' Randomisation by registration order Spanish paper stated that groups were led by an independent member of the research team Spanish paper states 'Evaluator was blind to treatment allocation' Attrition reported: 6/20 in CS + donepezil group; 10/30 in donepezil alone group i.e. 32% over 2 year period Spector 2001 24 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N = 35 Diagnosis of dementia according to DSM-IV criteria MMSE 13.1 (sd 4.4) Age 85.7 (sd 6.7) Living at home: 12; living in residential home: 23 Interventions Cognitive stimulation Treatment as usual Outcomes Cognition: MMSE; ADAS-Cog Communication: Holden Communication Scale Mood: Cornell Scale for Depression in Dementia; RAID Behaviour: Behaviour Rating Scale (CAPE). Family caregivers: Relatives Stress Scale; GHQ Notes 45 minutes, 2 times a week, for 7 weeks Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Low risk Support for judgement Randomly allocated to either group by drawing names from a sealed container Allocation concealment (selection bias) Low risk Randomly allocated to either group by drawing names from a sealed container Blinding (performance bias and detection bias) Unclear risk Not clear whether staff and carer ratings were made blind to treatment allocation Blinding of outcome assessment (detection bias) Low risk Cognitive assessments made by a blind assessor Incomplete outcome data (attrition bias) Low risk Attrition reported: 4 in CS group, 4 in control group i.e. 23% Other bias Low risk Groups led by a member of the research team in a separate room for the programme in each of the centres Spector 2003 25 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N = 201 (158F / 43M) Dementia (DSM-IV criteria) - MMSE 10-24 MMSE: 14.4 (sd 3.8) Age: 85.3 (sd 7.0) Groups ran in 18 residential homes; 5 day centres Interventions Cognitive stimulation No treatment Outcomes Cognition: MMSE; ADAS-Cog Quality of life: QoL-AD Communication: Holden Communication Scale Mood: Cornell Scale for Depression in Dementia Behaviour: Behaviour Rating Scale (CAPE) Notes 45 minutes, 2 times a week, for 7 weeks Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Low risk Support for judgement Randomly allocated to either group by drawing names from a sealed container Randomly allocated to either group by drawing names from a sealed container - would have been preferable for randomisation to have been carried out independently Allocation concealment (selection bias) Low risk Blinding (performance bias and detection bias) Low risk Members of staff involved in groups did not carry out ratings, but ratings by other staff may not have been blind Blinding of outcome assessment (detection bias) Low risk Cognitive assessments and quality of life interview conducted by a blind assessor Incomplete outcome data (attrition bias) Low risk Other bias Low risk 34/201 did not complete study (18 CS / 16 controls); 17% attrition Groups led by a member of the research team in a specific room for the programme in each of the centres, with a member of staff Wallis 1983 26 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N = 60 31 'Demented / organic'; 29 'functional' not included in this review Age 69.8 (range 38-95) All residents in long-stay psychiatric hospital Interventions RO groups Diversional Occupational Therapy - group and individual Outcomes Cognition: Royal College of Physicians mental test score Behaviour: Crichton Behaviour Rating Scale One month follow up data available Notes 30 minutes, 5 times a week, for 3 months Risk of bias table Bias Random sequence generation (selection bias) Authors' judgement Low risk Support for judgement Drawing from a hat and consecutive allocation Allocation concealment (selection bias) Low risk Drawing from a hat and consecutive allocation Blinding (performance bias and detection bias) Low risk Setting of treatment separate to assessment settings Blinding of outcome assessment (detection bias) Low risk Assessors unaware of group allocation Incomplete outcome data (attrition bias) Unclear risk Other bias Low risk Eliminated those attending less than 20% of sessions; 12/31 participants in 'organic' group lost i.e. 39% Occupational therapists trained to carry out RO Woods 1979 Methods RCT Participants N = 18 'disorientated', significant memory impairment Age 76.6 (range 61-90) All living in specialist residential homes for people with dementia Interventions RO groups Social Therapy groups No treatment (in a different home, so not included in this review) Outcomes Cognition: Wechsler Memory Scale; composite Information & Orientation test Behaviour: modified Crichton Behaviour Rating Scale Notes 30 minutes, 5 times a week, for 20 weeks Risk of bias table 27 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Bias Random sequence generation (selection bias) Authors' judgement Low risk Support for judgement Stated that drawing from a hat was used Allocation concealment (selection bias) Unclear risk Stated that drawing from a hat was used Blinding (performance bias and detection bias) Low risk 'Social therapy' was perceived by staff as an active therapy Blinding of outcome assessment (detection bias) Low risk Groups held in separate areas and assessors bind to group allocation Incomplete outcome data (attrition bias) Low risk 4/18 dropped out: i.e. 22.2% attrition Other bias Low risk Checks were made in order to ensure compliance with therapeutic protocol, including rating tape-recorded sessions Footnotes Characteristics of excluded studies Arcoverde 2008 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Diagnoses varied, not purely dementia. Basak 2008 Reason for exclusion Intervention described doesn't meet the inclusion criteria for cognitive stimulation; better fit for cognitive training. Brook 1975 Reason for exclusion Does not include a measure of cognitive function. Carlson 2008 Reason for exclusion Intervention described doesn't meet the inclusion criteria for cognitive stimulation but for cognitive training. Diagnoses varied, not purely dementia. Cassinello 2008 Reason for exclusion Doesn't report an RCT, reports results from Tarraga 2001. Cheng 2006 Reason for exclusion Intervention described doesn't meet the inclusion criteria for cognitive stimulation. Constantinidou 2008 Reason for exclusion Intervention described doesn't meet the inclusion criteria for cognitive stimulation Corbeil 1999 Reason for exclusion Although intervention is described as "cognitive stimulation", it focuses on specific cognitive modalities. Primary reports outcomes for family caregivers, no measure of cognitive function. Relates to Quayhagen, 1995. Croisile 2006 28 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Reason for exclusion Doesn't report results from an RCT. Davis 2001 Reason for exclusion Cognitive stimulation (delivered for 30 minutes a day, 6 days a week by family caregivers) confounded with cognitive training-spaced retrieval and face name associations. Eckroth-Bucher 2009 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Diagnoses varied, not purely dementia. Eggermont 2009a Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Eggermont 2009b Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Evans 2009 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Faggian 2007 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Fanto 2002 Reason for exclusion No mention of randomisation; available only as a conference abstract. Farina 2006a Reason for exclusion Non randomised allocation; comparison is with an active treatment group. Farina 2006b Reason for exclusion Non randomised allocation; comparison is with an active treatment group. Forbes 2004a Reason for exclusion Commentary on Spector 2003. Gerber 1991 Reason for exclusion Eligible study, but no extractable data provided. Only data available is for a composite cognitive and behavioural scale. Goldstein 1982 Reason for exclusion Around 25% of participants appear not to have dementia, other diagnoses include schizophrenia, epilepsy and ruptured aneurysm. Gonzalez-Abraldes 2010 29 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Green 2009 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Greenaway 2008 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Hanley 1981 Reason for exclusion Eligible study, but no extractable data available Hernandez, 2007 Reason for exclusion Reports on Requena 2006 study results. Article in Spanish. Hirsch 2004 Reason for exclusion Reports on Spector 2003 results. Holden 1978 Reason for exclusion Diagnoses varied, not purely dementia. Not clear that participants were randomised to the intervention and control groups. Johnson 1981 Reason for exclusion Allocation of patients to treatment was not random for various practical reasons. Leach 2004 Reason for exclusion Commentary on Spector 2003. Matsuda 2007 Reason for exclusion Non-randomised study. Meza-Kubo 2009 Reason for exclusion Not a randomised control trial. Milev 2008 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation - relates to 'snoezelen'. Moniz-Cook 2006 Reason for exclusion Reference to other studies in cognitive stimulation (e.g. Spector 2003) but not a new RCT. Mudge 2008 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Newson 2006 30 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Diagnoses varied, not dementia. Olazaran 2004 Reason for exclusion 12/84 participants with a diagnoses of MCI; results not presented separately for those with Alzheimer's disease.Interventions include additional elements as physical exercise. Orrell 2000b Reason for exclusion Describes study aims for Spector 2003. Orrell 2005 Reason for exclusion Allocation to intervention and control groups not random for maintenance study. Quayhagen 1995 Reason for exclusion Although intervention is described as cognitive stimulation, it appears to focus on specific cognitive modalities, and so fits better with cognitive training definition. Quayhagen 2000 Reason for exclusion Although intervention is described as cognitive stimulation, it appears to focus on specific cognitive modalities, and so fits better with cognitive training definition. Raggi 2007 Reason for exclusion Not RCT. Reeve 1985 Reason for exclusion No indication of random allocation to groups. Riegler 1980 Reason for exclusion Comparin of RO plus music versus RO. No control groups without RO. Ruiz Sanchez de Leon 2007 Reason for exclusion No RCT and intervention doesn't meet the criteria for inclusion under cognitive stimulation. Salmon 2006 Reason for exclusion No RCT, reports on Spector 2003 trial results. Schmitter-Edgecombe 2008 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Schreiber 1998 Reason for exclusion Cognitive training, targeting specific cognitive modalities, rather than cognitive stimulation. Allocation to groups alternate, not random. Schreiber 1999 31 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Reason for exclusion See Schreiber 1998. Scott 2003 Reason for exclusion Doesn't report on a study intervention. No RCT. Smith 2009 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation. Spector 2008 Reason for exclusion Reports on Spector 2003 results. Tadaka 2004 Reason for exclusion The intervention combines elements of Reality Orientation (RO) and reminiscence. The RO element appears to be only an orientation board, used to reinforce orientation for time, place and person. The reminiscence element appears to be predominant, with a variety of reminiscence based triggers, and so the study would be a better fit for a review of reminiscence work with people with dementia. Tarraga 2005a Reason for exclusion Published as Tarraga 2006. Tarraga 2005b Reason for exclusion Published as Tarraga 2006. Tarraga 2006 Reason for exclusion Allocation to groups is not entirely random. For the comparison of interest, integrated psychostimulation program versus medication only control, allocation is clearly nonrandom. Tarraga 2007 Reason for exclusion As in Tarraga 2006. Thickpenny-Davis 2007 Reason for exclusion Intervention doesn't meet the inclusion criteria for cognitive stimulation, participants included with other diagnosis than dementia. Tsai 2008 Reason for exclusion Not RCT. Wenisch 2005 Reason for exclusion Participants included with a diagnosis of MCI and not dementia. Wenisch 2007 Reason for exclusion As in Wenisch 2005. Wettstein 2004 32 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Reason for exclusion Does not report an intervention study. Williams 1987 Reason for exclusion Not a RCT, compares two wards, not cognitive stimulation, involves environmental modification and informal RO. Woods 2006 Reason for exclusion Report on Spector 2003 study results. Zanetti 1995 Reason for exclusion Allocation non-randomised. Zanetti 2004 Reason for exclusion As in Zanetti 1995. Zepelin 1981 Reason for exclusion Not a RCT, compares residents at one home with those in another. Zientz 2007 Reason for exclusion Does not report an intervention study. Footnotes Characteristics of studies awaiting classification Buettner 2011 Methods RCT Participants N=77 (15M/62F) Community dwelling Mild memory loss - probable early stage Alzheimer's MMSE 25.3 Interventions Classroom style 'Mentally Stimulating Activities' v Structured early-stage social support programme Outcomes Cognition: MMSE, Trail Making B Quality of life: Cornell-Brown QoL Depression: PHQ-9 Apathy Evaluation Scale Notes Sessions 1 hour, twice weekly for 4 weeks Fernandez-Calvo 2010 33 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Methods RCT Participants N=45 Probable Alzheimer's MMSE 18.97 Interventions Outcomes Individual multimodal cognitive stimulation versus group multimodal cognitive stimulation versus no treatment control Cognition: ADAS-Cog NPI Cornell Depression Scale Notes In Spanish Niu 2010 Methods RCT Participants N=32 mild to moderate Alzheimer's with marked neuropsychiatric symptoms MMSE 17.1 Inpatients in military sanatorium All on donepezil Interventions Individual sessions, task based including reality orientation, fluency, and memory tasks Placebo control - communication exercise Outcomes Cognition: MMSE NPI, apathy, depression Notes 45 minutes, twice a week for 10 weeks Footnotes Characteristics of ongoing studies Aguirre 2010 34 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Study name MCST - maintenance cognitive stimulation Methods RCT Participants Target N=230 Diagnosis of dementia with DSM-IV criteria Mild to moderate dementia Interventions Outcomes All participants receive 7 weeks of twice weekly cognitive stimulation; then randomised to recive 6 months of once weekly maintenance cognitive stimulation Cognition: ADAS-Cog, MMSE Quality of Life: QoL-AD, DEMQOL Starting date Contact information e.aguirre@ucl.ac.uk Notes ISRCTN 26286067 Vidovich 2011 Study name PACE-AD Methods RCT Participants Target N=128 probable Alzheimer's Interventions Outcomes Cognitive activity groups for person with dementia and companion together v companions alone Cognition: ADAS-Cog Companion quality of life, mood, and general health Starting date Contact information vidovichm@meddent.uwa.edu.au Notes Trial registration: ACTRN 12610000653066 Footnotes Summary of findings tables Additional tables References to studies Included studies Baines 1987 Baines S, Saxby P, Ehlert K. Reality orientation and reminiscence therapy A controlled cross-over study of elderly confused people. British Journal of Psychiatry 1987;151:222-31. Baldelli 1993a Baldelli MV, Pirani A, Motta M, Abati E, Mariani E, Manzi V. Effects of reality orientation therapy on elderly patients in the community. Archives of Gerontology and Geriatrics 1993a;17(3):211-8. Baldelli 2002 35 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Baldelli MV, Boiardi R, Fabbo A, Pradelli JM, Neri M. The role of reality orientation therapy in restorative care of elderly patients with dementia plus stroke in the subacute nursing home setting. Archives of Gerontology and Geriatrics 2002;35 Suppl 8:15-22. Bottino 2005 Bottino CMC, Carvalho IAM, Alvarez AM, Avila R, Zukauskas PR, Bustamante SEZ, et al. Cognitive rehabilitation combined with drug treatment in Alzheimer's disease patients: a pilot study. Clinical Rehabilitation 2005;19:861-9. Breuil 1994 Breuil V, De Rotrou J, Forette F, Tortrat D, Ganansia Ganem A, Frambourt A, et al. Cognitive stimulation of patients with dementia: Preliminary results. International Journal of Geriatric Psychiatry 1994;9(3):211-7. Buschert 2011 Buschert VC, Friese U, Teipel SJ, Schneider P, Merensky W, Rujescu D, et al. Effects of a newly developed cognitive intervention in amnestic mild cognitive impairment and mild alzheimer's disease: a pilot study. Journal of Alzheimer's Disease 2011;25:679-94. [10.3233/JAD-2011-100999] Chapman 2004 Chapman SB, Weiner MF, Rackley A, Hynan LS, Zientz J. Effects of cognitive-communication stimulation for Alzheimer's disease patients treated with donepezil. Journal of Speech, Language, and Hearing Research 2004;47(5):1149-63. Coen 2011 Coen RF, Flynn B, Rigney, E, O'Connor E, Fitzgerald L, Murray C, et al. Efficacy of a cognitive stimulation therapy programme for people with dementia. Irish Journal of Psychological Medicine 2011;28(3):145-7. Ferrario 1991 Ferrario E, Cappa G, Molaschi M, Rocco M, Fabris F. Reality orientation therapy in institutionalized elderly patients: Preliminary results. Archives of Gerontology and Geriatrics 1991;12 Suppl 2:139-42. Onder 2005 Onder G, Zanetti, O, Giacobini E, Frisoni GB, Bartorelli L, et al. Reality orientation therapy combined with cholinesterase inhibitors in Alzheimer's disease: randomised controlled trial. British Journal of Psychiatry 2005;187:450-5. Requena 2006 Requena C, Lopez-Ibor MI, Maestu F, Campo P, Lopez-Ibor JJ, Ortiz T. Effects of cholinergic drugs and cognitive training on dementia. Dementia and Geriatric Cognitive Disorders 2004;18:50-4. * Requena C, Maestu F, Fernandez A, Ortiz T. Effects of cholinergic drugs and cognitive training on dementia: 2-year followup. Dementia and Geriatric Cognitive Disorders 2006;22:339-45. Spector 2001 Spector A, Orrell M, Davies S, Woods B. Can reality orientation be rehabilitated? Development and piloting of an evidencebased programme of cognition-based therapies for people with dementia. Neuropsychological Rehabilitation 2001;11(34):377-97. Spector 2003 Spector A, Thorgrimsen L, Woods B, Royan L, Davies S, Butterworth M, et al. Efficacy of an evidence-based cognitive stimulation therapy programme for people with dementia: randomised controlled trial. British Journal of Psychiatry 2003;183:248-54. Wallis 1983 Wallis GG, Baldwin M, Higginbotham P. Reality orientation therapy-a controlled trial. British Journal of Medical Psychology 1983;56(3):271-7. Woods 1979 Woods RT. Reality Orientation and Staff attention: A Controlled Study. British Journal of Psychiatry 1979;134:502-7. Excluded studies Arcoverde 2008 Arcoverde C, Deslandes A, Rangel A, Rangel A, Pavllo R, Nigri F, Engelhardt E, Laks J. Role of physical activity on the maintenance of cognition and activities of daily living in elderly with Alzheimer's disease. Alzheimer. Arq. Neuropsiquiatry 2008;66(2b):323-7. Basak 2008 Basak C, Boot WR, Voss MW, Kramer AF. Can training in a real-time strategy video game attenuate cognitive decline in older adults? Psychology and Aging 2008;23(4):765-77. Brook 1975 36 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Brook P, Degun G, Mather M. Reality orientation, a therapy for psychogeriatric patient: a controlled study. British Journal of Psychiatry 1975;127:42-5. Carlson 2008 Carlson MC, Saczynski JS, Rebok GW, Seeman T, Glass TA, McGill S, et al. Exploring the effects of an "everyday" activity program on executive function and memory in older adults: Experience Corps. The Gerontologist 2008;48(6):793-801. Cassinello 2008 Cassinello DZ, Tarraga L, Fernandez-Ballesteros R. Cognitive plasticity in Alzheimer's disease patients receiving cognitive stimulation programmes. Psychology in Spain 2009;13(1):48-54. * Cassinello DZ, Tarraga L, Fernandez-Ballesteros R. Cognitive plasticity in Alzheimer's disease patients receiving cognitive stimulation programs. Psicothema 2008;20:432-7. Cheng 2006 Cheng ST, Chan ACM, Yu ECS. An exploratory study of the effect of Mahjong on the cognitive functioning of persons with dementia. International Journal of Geriatric Psychiatry 2006;21(7):611-7. Constantinidou 2008 Constantinidou F, Thomas RD, Robinson L. Benefits of categorization training in patients with traumatic brain injury during post-acute rehabilitation: additional evidence from a randomized controlled trial. The Journal of Head Trauma Rehabilitation 2008;23(5):312-28. Corbeil 1999 Corbeil RR, Quayhagen MP, Quayhagen M. Intervention effects on dementia caregiving interaction: A stress-adaptation modeling approach. Journal of Aging and Health 1999;11(1):79-95. Croisile 2006 Croisile B. Memory stimulation. What rational? What exercises? Revue de Geriatrie 2006;31(6):421-33. Davis 2001 Davis RN, Massman PJ, Doody RS. Cognitive intervention in Alzheimer disease: A randomized placebo-controlled study. Alzheimer Disease and Associated Disorders 2001;15(1):1-9. Eckroth-Bucher 2009 Eckroth-Bucher M, Siberski J. Preserving cognition through an integrated cognitive stimulation and training program. American Journal of Alzheimer's Disease and other Dementias 2009;24(3):234-45. Eggermont 2009a Eggermont LH, Knol DL, Hol EM, Swaab DF, Scherder EJ. Hand motor activity, cognition, mood, and the rest-activity rhythm in dementia: a clustered RCT. Behavioural Brain Research 2009;196(2):271-8. Eggermont 2009b Eggermont LH, Swaab DF, Hol EM, Scherder EJ. Walking the line: a randomised trial on the effects of a short term walking programme on cognition in dementia. Journal of Neurology, Neurosurgery and Psychiatry 2009;80(7):802-4. Evans 2009 Evans JJ, Greenfield E, Wilson BA, Bateman A. Walking and Talking Therapy: Improving cognitive-motor dual-tasking in neurological illness. Journal of the International Neuropsychological Society 2009;15(1):112-20. Faggian 2007 Faggian S. An intervention protocol on cognitive abilities for subjects with severe cognitive impairment. Giornale di Gerontologia 2007;55(3):134-43. Fanto 2002 Fanto F, Tisci C, Molaschi M, Ostacoli L, Furlan PM, Riondato A, et al. Long-term reality orientation therapy for subjects with dementia in an Italian suburban setting. In: Proceedings of the 8th International Conference on Alzheimer's Disease and Related Disorders; 2002 July 20-25, Stockholm, Sweden. 2002:Abstract No 1966. Farina 2006a Farina E, Mantovani F, Fioravanti R, Pignatti R, Chiavari L, Imbornone E, et al. Evaluating two group programmes of cognitive training in mild-to-moderate AD: is there any difference between a 'global' stimulation and a 'cognitive-specific' one? Aging & Mental Health 2006;10(3):211-8. Farina 2006b Farina E, Mantovani F, Fioravanti R, Rotella G, Villanelli F, Imbornone E, et al. Efficacy of Recreational and Occupational Activities Associated to Psychologic Support in Mild to Moderate Alzheimer Disease - a Multicenter Controlled Study. Alzheimer Disease & Associated Disorders 2006;20(4):275-82. 37 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Forbes 2004a Forbes D. Cognitive stimulation therapy improved cognition and quality of life in dementia. Evidence-Based Nursing 2004a;7(2):54-5. Gerber 1991 Gerber GJ, Prince PN, Snider HG, Atchison K, Dubois L, Kilgour JA. Group activity and cognitive improvement among patients with Alzheimer's disease. Hospital and Community Psychiatry 1991;42(8):843-5. Goldstein 1982 Goldstein G. An evaluation of reality orientation therapy. Journal of Behavioral Assessment 1982;4(2):165-78. Gonzalez-Abraldes 2010 Gonzalez-Abraldes I, Millan-Calenti JC, Balo-Garcia A,Tubio J, Lorenzo T, Maseda A. Accessibility and usability of computer-based cognitive stimulation: Telecognitio.. Revista Espanola de Geriatria y Gerontologia 2010;45(1):26-9. Green 2009 Green HA, Patterson K. Jigsaws - a preserved ability in semantic dementia. Neuropsychologia 2009;47(2):569-76. Greenaway 2008 Greenaway MC, Hanna SM, Lepore SW, Smith GE. A behavioral rehabilitation intervention for amnestic mild cognitive impairment. American Journal of Alzheimer's Disease and other Dementias 2008;23(5):451-61. Hanley 1981 Hanley IG, McGuire RJ, Boyd WD. Reality orientation and dementia: A controlled trial of two approaches. British Journal of Psychiatry 1981;138:10-4. Hernandez, 2007 Hernandez R, Unturbe FM, Martinez-Lage P, Lucas AF, Gregorio PG, Ortiz Alonso T. Effects of combined pharmacologic and cognitive treatment in the progression of moderate dementia: A two-year follow-up. Revista Espanola de Geriatria y Gerontologia 2007;42(1):3-10. Hirsch 2004 Hirsch CH. Cognitive stimulation therapy improved cognition and quality of life in dementia. Evidence-Based Medicine 2004;9(2):49. Holden 1978 Holden UP, Sinebruchow A. Reality orientation therapy: a study investigating the value of this therapy in the rehabilitation of elderly people. Age and Ageing 1978;7(2):83-90. Johnson 1981 Johnson CH, McLaren SM, McPherson FM. The comparative effectiveness of three versions of 'classroom' reality orientation. Age and Ageing 1981;10(1):33-5. Leach 2004 Leach L. Cognitive stimulation therapy improves cognition and quality of life in older people with dementia. Evidence-Based Mental Health 2004;7(1):19. Matsuda 2007 Matsuda O. Cognitive stimulation therapy for Alzheimer's disease: the effect of cognitive stimulation therapy on the progression of mild Alzheimer's disease in patients treated with donepezil. International Psychogeriatrics 2007;19(2):241-52. Meza-Kubo 2009 Meza-Kubo V, Gonzalez-Fraga A, Moran AL, Tentori M. Augmenting Cognitive Stimulation Activities in a Nursing Home Through Pervasive Computing. In: La-Web: 2009 Latin American Web Congress. 2009:8-15. Milev 2008 Milev RV, Kellar T, McLean M, Mileva V, Luthra V, Thompson S, Peever L. Multisensory stimulation for elderly with dementia: a 24-week single-blind randomized controlled pilot study. American Journal of Alzheimer's Disease and Other Dementias 2008;23(4):372- 6. Moniz-Cook 2006 Moniz-Cook E. Cognitive stimulation and dementia. Aging & Mental Health 2006;10:207-10. Mudge 2008 Mudge AM, Giebel AJ, Cutler AJ. Exercising body and mind: An integrated approach to functional independence in hospitalized older people. Journal of the American Geriatrics Society 2008;56(4):630-5. Newson 2006 38 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Newson R, Kemps E. The influence of physical and cognitive activities on simple and complex cognitive tasks in older adults. Experimental Aging Research 2006;32(3):341-62. Olazaran 2004 Olazaran J, Muniz R, Reisberg B, Pena-Casanova J, del Ser T, Cruz-Jentoft AJ, et al. Benefits of cognitive-motor intervention in MCI and mild to moderate Alzheimer disease. Neurology 2004;63:2348-53. Orrell 2000b Orrell M. Evidence based psychological therapy package for dementia. National Research Register 2000b. Orrell 2005 Orrell M, Spector A, Thorgrimsen L, Woods B. A pilot study examining the effectiveness of maintenance Cognitive Stimulation Therapy (MCST) for people with dementia. International Journal of Geriatric Psychiatry 2005;20:446-51. Quayhagen 1995 Quayhagen MP, Quayhagen M, Corbeil RR, Roth PA, Rodgers JA. A dyadic remediation program for care recipients with dementia. Nursing Research 1995;44(3):153-9. Quayhagen 2000 Quayhagen MP, Quayhagen M, Corbeil RR, Hendrix RC, Jackson JE, Snyder L, et al. Coping with dementia: Evaluation of four non pharmacologic interventions. International Psychogeriatrics 2000;12(2):249-65. Raggi 2007 Raggi A, Iannaccone S, Marcone A, Ginex V, Ortelli P, Nonis A, et al. The effects of a comprehensive rehabilitation program of Alzheimer's Disease in a hospital setting. Behavioural Neurology 2007;18(1):1-6. Reeve 1985 Reeve W, Ivison D. Use of environmental manipulation and classroom and modified informal reality orientation with institutionalized, confused elderly patients. Age and Ageing 1985;14(2):119-21. Riegler 1980 Riegler J. Comparison of a reality orientation program for geriatric patients with and without music. Journal of Music Therapy 1980;17(1):26-33. Ruiz Sanchez de Leon 2007 Ruiz Sanchez De Leon JM, Llanero Luque M. Computer-based cognitive training and donepezil: Combined therapy effects in cognitive impairment. Mapfre Medicina 2007;18(1):25-33. Salmon 2006 Salmon N. Cognitive stimulation therapy versus acetyl cholinesterase inhibitors for mild to moderate dementia: a latter-day David and Goliath? British Journal of Occupational Therapy 2006;69(11):528-30. Schmitter-Edgecombe 2008 Schmitter-Edgecombe M, Howard JT, Pavawalla SP, Howell L, Rueda A. Multidyad memory notebook intervention for very mild dementia: a pilot study. American Journal of Alzheimer's Disease & Other Dementias 2008;23(5):477-87. Schreiber 1998 Schreiber M, Lutz K, Schweizer A, Kalveram KT, Jancke L. Development and evaluation of an interactive computer-based training as a rehabilitation tool for dementia. Psychologie Beitrage 1998;40(1):85-102. Schreiber 1999 Schreiber M, Schweizer A, Lutz K, Kalveram KT, Jancke L. Potential of an interactive computer-based training in the rehabilitation of dementia: an initial study. Neuropsychological Rehabilitation 1999;9(2):155-67. Scott 2003 Scott J, Clare L. Do people with dementia benefit from psychological interventions offered on a group basis? Clinical Psychology and Psychotherapy 2003;10(3):186-96. Smith 2009 Smith GE, Housen P, Yaffe K, Ruff R, Kennison RF, Mahncke HW, Zelinski EM. A cognitive training program based on principles of brain plasticity: Results from the improvement in memory with plasticity- based adaptive cognitive training (IMPACT) study. Journal of the American Geriatrics Society 2009;57(4):594-603. Spector 2008 Spector A, Woods B, Orrell M. Cognitive stimulation for the treatment of Alzheimer's disease. Expert Review of Neurotherapeutics 2008;8(5):751-7. Tadaka 2004 39 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Tadaka E, Kanagawa K. A randomized controlled trial of a group care program for community-dwelling elderly people with dementia. Japan Journal of Nursing Science 2004;1:19-25. Tarraga 2005a Tarraga L. Randomised pilot study to evaluate the efficacy of an interactive multimedia system for the cognitive stimulation of Alzheimer's disease patients. In: 1st International Consensus Conference Non pharmacological Therapies for Alzheimer's, Madrid, May 12-13. 2005:35-6. Tarraga 2005b Tarraga L, Badenas S, Modinos G, Espinosa A, Diego S, Balcells J, et al. Randomized Pilot Study To Evaluate the Efficacy of an Interactive Multimedia Internet-Based Tool for the Cognitive Stimulation of Patients with Alzheimer's Disease. In: 57th Annual Meeting of the American Academy of Neurology, Miami Beach, April 2005. 2005:P01.138. Tarraga 2006 Tarraga L, Boada M, Modinos G, Espinosa A, Diego S, Morera A, et al. A randomised pilot study to assess the efficacy of an interactive, multimedia tool of cognitive stimulation in Alzheimer's disease. Journal of Neurology, Neurosurgery & Psychiatry 2006;77:1116-21. Tarraga 2007 Tarraga L, Boada M, Modinos G, Espinosa A, Diego S, Morera A, et al. Interactive cognitive stimulation with a computer and Alzheimer's disease. Neuroscientist 2007;13(2):97. Thickpenny-Davis 2007 Thickpenny-Davis KL, Barker-Collo SL. Evaluation of a structured group format memory rehabilitation program for adults following brain injury. The Journal of Head Trauma Rehabilitation 2007;22:303-13. Tsai 2008 Tsai AY, Yang M, Lan C, Chen C. Evaluation of effect of cognitive intervention programs for the community-dwelling elderly with subjective memory complaints. International Journal of Geriatric Psychiatry 2008;23(11):1172-4. Wenisch 2005 Wenish E, Stoker A, Bourrellis C, Pasquet C, Gauthier E, Corcos E, et al. A global intervention program for institutionalized demented patients. Revue Neurologique 2005;161(3):290-8. Wenisch 2007 Wenisch E, Cantegreil-Kallen I, De Rotrou J, Garrigue P, Moulin F, Batouche F, et al. Cognitive stimulation intervention for elders with mild cognitive impairment compared with normal aged subjects: preliminary results. Aging Clinical & Experimental Research 2007;19(4):316-22. Wettstein 2004 Wettstein A, Schmid R, Konig M. Who participates in psychosocial interventions for caregivers of patients with dementia? Dementia and Geriatric Cognitive Disorders 2004;18(1):80-6. Williams 1987 Williams R, Reeve W, Ivison D, Kavanagh D. Use of environmental manipulation and modified informal reality orientation with institutionalized, confused elderly subjects: a replication. Age and Ageing 1987;16(5):315-8. Woods 2006 Woods B, Thorgrimsen L, Spector A, Royan L, Orrell M. Improved quality of life and cognitive stimulation therapy in dementia. Aging & Mental Health 2006;10(3):219-26. Zanetti 1995 Zanetti O, Frisoni GB, De Leo D, Dello Bueno M, Bianchetti A, Trabucchi M. Reality orientation therapy in Alzheimer disease: Useful or not? A controlled study. Alzheimer Disease and Associated Disorders 1995;9(3):132-8. Zanetti 2004 Zanetti O, Calabria M, Cotelli M. Effectiveness of the association of donepezil and Reality Orientation Therapy. Giornale di Gerontologia 2004;52(5):480-411. Zepelin 1981 Zepelin H, Wolfe CS, Kleinplatz F. Evaluation of a year long reality orientation program. Journal of Gerontology 1981;36(1):70-7. Zientz 2007 Zientz J, Rackley A, Chapman SB, Hopper T, Mahendra N, Cleary S. Evidence-based practice recommendations: Caregiveradministered active cognitive stimulation for individuals with Alzheimer's disease.. Journal of Medical Speech-Language Pathology 2007;15(3):27-34. 40 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Studies awaiting classification Buettner 2011 Buettner LL, Fitzsimmons S, Atav S, Sink K. Cognitive stimulation for apathy in probable early-stage Alzheimer's. Journal of Aging Research 2011. [10.4061/2011/480890] Fernandez-Calvo 2010 Fernandez-Calvo B, Contador I, Serna A, de Lucena VM, Ramos F. The effect of an individual or group intervention format in cognitive stimulation of patients with Alzheimer's disease. [El efecto del formato de intervencion individual o grupal en la estimulacion cognitiva de pacientes con enfermedad de Alzheimer]. Revista de Psicopatología y Psicología Clínica 2010;15:115-23. Niu 2010 Niu YX, Tan JP, Guan JQ, Zhang ZQ, Wang LN. Cognitive stimulation therapy in the treatment of neuropsychiatric symptoms in Alzheimer's disease: a randomized controlled trial. Clinical Rehabilitation 2010;24:1002-1011. [10.1177/0269215510376004] Ongoing studies Aguirre 2010 Aguirre E, Spector A, Hoe J, Russell IT, Knapp M, Woods RT, Orrell M. Maintenance Cognitive Stimulation Therapy (CST) for dementia: A single-blind, multi-centre, randomized controlled trial of Maintenance CST vs. CST for dementia. Trials 2010;11:46. Vidovich 2011 Vidovich MR, Shaw J, Flicker L, Almeida OP. Cognitive activity for the treatment of older adults with mild Alzheimer's disease (AD)--PACE AD: study protocol for a randomised controlled trial.. Trials 2011;12:47. Other references Additional references Alexopoulos 1988 Alexopoulos GS, Abrams RC, Young RC, Shamoian CA. Cornell Scale for Depression in Dementia. Biological Psychiatry 2008;23:271-84. APA 1997 American Psychiatric Association. Practice guideline for the treatment of patients with Alzheimer's disease and other dementias of late life. American Journal of Psychiatry 1997;154 Suppl(5):1-39. Burton 1982 Burton M. Reality orientation for the elderly: a critique. Journal of Advanced Nursing 1982;7:427-33. Clare 2003 Clare L, Woods RT, Moniz Cook ED, Orrell M, Spector A. Cognitive rehabilitation and cognitive training for early-stage Alzheimer's disease and vascular dementia. Cochrane Database of Systematic Reviews 2003, Issue Issue 4 Art. No.: CD003260.. Art. No.: CD003260 DOI: 10.1002/14651858.CD003260. Clare 2004 Clare L, Woods RT. Cognitive training and cognitive rehabilitation for people with early-stage Alzheimer's disease: a review. Neuropsychological Rehabilitation 2004;14:385-401. Dietch 1989 Dietch JT, Hewett LJ, Jones S. Adverse effects of reality orientation. Journal of American Geriatrics Society 1989;37:974-6. Folstein 1975 Folstein MF, Folstein SE, McHugh PR. 'Mini Mental State': a practical method for grading the cognitive state of patients for the clinician. Journal of Psychiatric Research 1975;12:189-98. Holden 1995 Holden UP, Woods RT. Positive approaches to dementia care. 3rd edition. Edinburgh: Churchill Livingstone, 1995. Hughes 1982 Hughes CP, Berg L, Danziger WL, Coben LA, Martin RL. A new clinical scale for the staging of dementia. The British Journal of Psychiatry 1982;140:566-72. Knapp 2006 Knapp M, Thorgrimsen L, Patel A, Spector A, Hallam A, Woods B, Orrell M. Cognitive stimulation therapy for people with dementia: cost-effectiveness analysis. British Journal of Psychiatry 2006;188:574-80. 41 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Livingston 2005 Livingston G, Johnston K, Katona C, Paton J, Lyketsos CG. Systematic review of psychological approaches to the management of neuropsychiatric symptoms of dementia. American Journal of Psychiatry 2005;162(11):1996-2021. McGilton 2003 McGilton KS, Rivera TM, Dawson P. Can we help persons with dementia find their way in a new environment? Aging & Mental Health 2003;7(5):363-71. Mohs 2000 Mohs R. Neuropsychological assessment of patients with Alzheimer's disease. In: Bloom FE, Kupfer DJ, editors(s). Psychopharmacology: 4th generation of progress. American College of Neuropsychopharmacology, 2000:http://www.acnp.org/publications/psycho4generation.aspx. NICE-SCIE 2006 NICE-SCIE. Dementia: Supporting people with dementia and their carers in health and social care: Clinical Guideline 42.. London: NICE-SCIE, 2006. Olazaran 2010 Olazaran J, Reisberg B, Clare L, Cruz I, Pena-Casanova J, del Ser T, et al. Non-pharmacological therapies in Alzheimer's disease: a systematic review of efficacy. Dementia and Geriatric Cognitive Disorders 2010;30:161-78. Powell-Proctor 1982 Powell-Proctor L, Miller E. Reality orientation: a critical appraisal. British Journal of Psychiatry 1982;140:457-63. Prince 2011 Prince M, Bryce R, Ferri C. World Alzheimer Report 2011: the benefits of early diagnosis and intervention. London: Alzheimer's Disease International, 2011. [ Other: http://www.alz.co.uk/research/WorldAlzheimerReport2011.pdf] Rosen 1984 Rosen WG, Mohs RC, Davis KL. A new rating scale for Alzheimer's disease. American Journal of Psychiatry 1984;141:135664. Sitzer 2006 Sitzer D, Twamley E, Jeste D. Cognitive training in Alzheimer's disease: a meta-analysis of the literature. Acta Psychiatrica Scandinavica 2006;114(2):75-90. Small 2002 Small GW. What we need to know about age related memory loss. BMJ 2002;324:1502-5. Spector 2000b Spector A, Orrell M, Davies S, Woods RT. Reality orientation for dementia. Cochrane Database of Systematic Reviews 2000, Issue Issue 3. Art. No.: CD001119. Art. No.: CD001119 DOI: 10.1002/14651858.CD001119. Spector 2006 Spector A, Thorgrimsen L, Woods B, Orrell M. Making a difference: an evidence-based group programme to offer cognitive stimulation therapy (CST) to people with dementia. London: Hawker Publications, 2006. Spector 2010 Spector A, Orrell M, Woods B. Cognitive Stimulation Therapy (CST): effects on different areas of cognitive function for people with dementia. International Journal of Geriatric Psychiatry 2010;25:1253-8. Taulbee 1966 Taulbee LR, Folsom JC. Reality orientation for geriatric patients. Hospital & Community Psychiatry 1966;17:133-5. Woods 1977 Woods RT, Britton PG. Psychological approaches to the treatment of the elderly. Age & Ageing 1977;6:104-12. Woods 2002 Woods B. Editorial: Reality Orientation: a welcome return? Age & Ageing 2002;31:155-6. Yesavage 1983 Yesavage JA, Brink TL, Rose TL. Development and validation of a geriatric depression scale: a preliminary report. Journal of Psychiatric Research 1983;17:37-49. Other published versions of this review Spector 2000a Spector A, Davies S, Woods B, Orrell M. Reality orientation for dementia: a systematic review of the evidence for its 42 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia effectiveness. Gerontologist 2000;40(2):206-212. Classification pending references Baines 1987 Baines S, Saxby P, Ehlert K. Reality orientation and reminescence therapy A controlled cross-over study of elderly confused people. British Journal of Psychiatry 1987;151:222-31. Baldelli 1993a Baldelli MV, Pirani A, Motta M, Abati E, Mariani E, Manzi V. Effects of reality orientation therapy on elderly patients in the community. Archives of Gerontology and Geriatrics 1993a;17(3):211-8. Baldelli 2002 Baldelli MV. The role of reality orientation therapy in restorative care of elderly patients with dementia plus stroke in the subacute nursing home setting. Archives of Gerontology and Geriatrics 2002;35(Suppl 8):15-22. Breuil 1994 Breuil V, De Rotrou J, Forette F, Tortrat D, Ganansia Ganem A, Frambourt A, Moulin F, Boller F. Cognitive stimulation of patients with dementia: Preliminary results. International Journal of Geriatric Psychiatry 1994;9(3):211-7. Brook 1975 Brook P, Degun G, Mather M. Reality orientation, a therapy for psychogeriatric patient: a controlled study. British Journal of Psychiatry 1975;127:42-5. Chapman 2004 Chapman SB, Weiner MF, Rackley A, Hynan LS, Zientz J. Effects of cognitive-communication stimulation for Alzheimer's disease patients treated with donepezil. Journal of speech, language, and hearing research JSLHR 2004;47(5):1149-63. Corbeil 1999 Corbeil RR, Quayhagen MP, Quayhagen M. Intervention effects on dementia caregiving interaction: A stress-adaptation modeling approach. Journal of Aging and Health 1999;11(1):79-95. Davis 2001 Davis RN, Massman PJ, Doody RS. Cognitive intervention in Alzheimer disease: A randomized placebo-controlled study. Alzheimer Disease and Associated Disorders 2001;15(1):1-9. Fanto 2002 Fanto F, Tisci C, Molaschi M, Ostacoli L, Furlan PM, Riondato A, Trevisan A, Iachino A, Pignata M, Perino B, Santoro M, Prolo P, Gorlato S. Long-term reality orientation therapy for subjects with dementia in an Italian suburban setting. Proceedings of the 8th International Conference on Alzheimer's Disease and Related Disorders; 2002 July 20-25, Stockholm, Sweden 2002:Abstract No 1966. Ferrario 1991 Ferrario E, Cappa G, Molaschi M, Rocco M, Fabris F. Reality orientation therapy in institutionalized elderly patients: Preliminary results. Archives of Gerontology and Geriatrics 1991;12(Suppl 2):139-42. Forbes 2004a Forbes D. Cognitive stimulation therapy improved cognition and quality of life in dementia. Evidence-Based-Nursing 2004a;7(2):54-5. Gerber 1991 Gerber GJ, Prince PN, Snider HG, Atchison K, Dubois L, Kilgour JA. Group activity and cognitive improvement among patients with Alzheimer's disease. Hospital and Community Psychiatry 1991;42(8):843-5. Goldstein 1982 Goldstein G. An evaluation of reality orientation therapy. Journal of Behavioral Assessment 1982;4(2):165-78. Hanley 1981 Hanley IG, McGuire RJ, Boyd WD. Reality orientation and dementia: A controlled trial of two approaches. British Journal of Psychiatry 1981;138:10-4. Holden 1978 Holden UP, Sinebruchow A. Reality orientation therapy: a study investigating the value of this therapy in the rehabilitation of elderly people. Age and Ageing 1978;7(2):83-90. Johnson 1981 Johnson CH, McLaren SM, McPherson FM. The comparative effectiveness of three versions of 'classroom' reality orientation. Age and Ageing 1981;10(1):33-5. 43 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Kiemle 1987 Kiemle G. Social and cognitive reorientation in psychogeriatric patients. Social and cognitive reorientation in psychogeriatric patients 1987. Leach 2004 Leach L. Cognitive stimulation therapy improves cognition and quality of life in older people with dementia. Evidence-BasedMental-Health 2004;7(1):19. Orrell 2000b Orrell M. Evidence based psychological therapy package for dementia. National Research Register 2000b. Peoples 1982 Peoples M. Validation therapy versus reality orientation as treatment for disorientated institutionalised elderly. Masters dissertation, College of Nursing, University of Akron 1982. Quayhagen 1995 Quayhagen MP, Quayhagen M, Corbeil RR, Roth PA, Rodgers JA. A dyadic remediation program for care recipients with dementia. Nursing Research 1995;44(3):153-9. Quayhagen 2000 Quayhagen MP, Quayhagen M, Corbeil RR, Hendrix RC, Jackson JE, Snyder L, Bower D. Coping with dementia: Evaluation of four non pharmacologic interventions. International Psychogeriatrics 2000;12(2):249-65. Reeve 1985 Reeve W, Ivison D. Use of environmental manipulation and classroom and modified informal reality orientation with institutionalized, confused elderly patients. Age and Ageing 1985;14(2):119-21. Riegler 1980 Riegler J. Comparison of a reality orientation program for geriatric patients with and without music. Journal of Music Therapy 1980;17(1):26-33. Schreiber 1998 Schreiber M, Lutz K, Schweizer A, Kalveram KT, Jancke L. Development and evaluation of an interactive computer-based training as a rehabilitation tool for dementia. Psychologie Beitrage 1998;40(1):85-102. Spector 2001b Spector A, Orrell M, Davies S, Woods. Can reality orientation be rehabilitated? Development and piloting of an evidencebased programme of cognition-based therapies for people with dementia. Neuropsychological Rehabilitation 2001;11(34):377-97. Spector 2003 Spector A, Thorgrimsen L, Woods B, Royan L, Davies S, Butterworth M, Orrell M. Efficacy of an evidence-based cognitive stimulation therapy programme for people with dementia: randomised controlled trial. British journal of psychiatry the journal of mental science, The 2003;183:248-54. Tarraga 2005a Tarraga L. Randomised pilot study to evaluate the efficacy of an interactive multimedia system for the cognitive stimulation of Alzheimer's disease patients. In: 1st International Consensus Conference Non pharmacological Therapies for Alzheimer's, Madrid, May 12-13 2005. 2005:35-6. Tarraga 2005b Tarraga L, Badenas S, Modinos G, Espinosa A, Diego S, Balcells J, Lopez OL, Becker JT, Boada M. Randomized Pilot Study To Evaluate the Efficacy of an Interactive Multimedia Internet-Based Tool for the Cognitive Stimulation of Patients with Alzheimer's Disease. In: 57th Annual Meeting of the American Academy of Neurology, Miami Beach, April 2005. 2005:P01.138. Wallis 1983 Wallis GG, Baldwin M, Higginbotham P. Reality orientation therapy-a controlled trial. British Journal of Medical Psychology 1983;56((Pt 3)):271-7. Wettstein 2004 Wettstein A, Schmid R, Konig M. Who participates in psychosocial interventions for caregivers of patients with dementia? Dementia and geriatric cognitive disorders 2004;18(1):80-6. Williams 1987b Williams R, Reeve W, Ivison D, Kavanagh D. Use of environmental manipulation and modified informal reality orientation with institutionalized, confused elderly subjects: a replication. Age and Ageing 1987;16(5):315-8. 44 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Woods 1979 Woods RT. Reality Orientation and Staff attention: A Controlled Study. British Journal of Psychiatry 1979;134:502-7. Zanetti 1995 Zanetti O, Frisoni GB, De Leo D, Dello Bueno M, Bianchetti A, Trabucchi M. Reality orientation therapy in Alzheimer disease: Useful or not? A controlled study. Alzheimer Disease and Associated Disorders 1995;9(3):132-8. Zepelin 1981 Zepelin H, Wolfe CS, Kleinplatz F. Evaluation of a yearlong reality orientation program. Journal of gerontology 1981;36(1):707. Data and analyses 1 Cognitive stimulation versus no cognitive stimulation: post-treatment Outcome or Subgroup Studies Participants Statistical Method 1.1 Cognition 14 658 1.1.1 ADAS-Cog 7 434 1.1.2 Wechsler Memory Scale 1 10 1.1.3 Global cognitive score (includes MMSE & CERAD) 1 56 1.1.4 MMSE 2 110 1.1.5 CAPE-I/O 2 29 1.1.6 RCP Cognition 1 19 Effect Estimate Std. Mean Difference(IV, Fixed, 95% 0.41[0.25, 0.57] CI) Std. Mean Difference(IV, Fixed, 95% 0.37[0.17, 0.56] CI) Std. Mean Difference(IV, Fixed, 95% 0.47[-0.80, 1.74] CI) Std. Mean Difference(IV, Fixed, 95% 0.63[0.09, 1.17] CI) Std. Mean Difference(IV, Fixed, 95% 0.64[0.17, 1.10] CI) Std. Mean Difference(IV, Fixed, 95% 0.29[-0.48, 1.06] CI) Std. Mean Difference(IV, Fixed, 95% 0.13[-0.78, 1.03] CI) 1.2 MMSE 10 1.2.1 One to twelve months of CS 10 1.2.2 24 months of CS 1 600 29 Mean Difference(IV, Fixed, 95% CI) Subtotals only Mean Difference(IV, Fixed, 95% CI) 1.74[1.13, 2.36] Mean Difference(IV, Fixed, 95% CI) 5.99[-1.58, 13.56] 1.3 ADAS-Cog 7 1.3.1 One to twelve months of CS 7 1.3.2 24 months of CS 1 434 29 Mean Difference(IV, Fixed, 95% CI) Subtotals only Mean Difference(IV, Fixed, 95% CI) 2.27[0.99, 3.55] Mean Difference(IV, Fixed, 95% CI) 11.94[-0.97, 24.85] 1.4 Other cognitive measure: Information/Orientation 5 81 1.4.1 CAPE I/O 2 29 1.4.2 RCP Cognition 1 19 1.4.3 Berg Orientation Scale 1 23 1.4.4 Information / Orientation 1 10 4 223 3 204 1 19 1.6 Well-being & Quality of Life 4 219 1.6.1 Life Satisfaction Index 1 10 1.6.2 QoL-AD 3 209 1.5 Comunication and social interaction 1.5.1 Holden Communication Scale 1.5.2 MOSES - Withdrawn behaviour Std. Mean Difference(IV, Fixed, 95% 0.45[-0.01, 0.90] CI) Std. Mean Difference(IV, Fixed, 95% 0.29[-0.48, 1.06] CI) Std. Mean Difference(IV, Fixed, 95% 0.13[-0.78, 1.03] CI) Std. Mean Difference(IV, Fixed, 95% 0.87[0.00, 1.74] CI) Std. Mean Difference(IV, Fixed, 95% 0.60[-0.68, 1.89] CI) Std. Mean Difference(IV, Fixed, 95% 0.44[0.17, 0.71] CI) Std. Mean Difference(IV, Fixed, 95% 0.47[0.18, 0.75] CI) Std. Mean Difference(IV, Fixed, 95% 0.10[-0.86, 1.07] CI) Std. Mean Difference(IV, Fixed, 95% 0.38[0.11, 0.65] CI) Std. Mean Difference(IV, Fixed, 95% -0.23[-1.48, 1.01] CI) Std. Mean Difference(IV, Fixed, 95% 0.41[0.13, 0.69] CI) 45 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 1.7 Mood: Self-reported 5 1.7.1 Geriatric Depression Scale (GDS-30) One to twelve months of 3 CS 1.7.2 Geriatric Depression Scale (14 item) One to twelve months of 1 CS 1.7.3 Montgomery Asberg Depression Rating Scale (MADRS) 1 One to twelve months of CS 1.8 Mood: Staff-reported 4 1.8.1 Cornell Scale for Depression 2 in Dementia 1.8.2 MOSES - Depressed / 1 anxious mood 1.8.3 Rating of Anxiety in 1 Dementia (RAID) 201 Std. Mean Difference(IV, Fixed, 95% 0.22[-0.09, 0.53] CI) 160 Std. Mean Difference(IV, Fixed, 95% 0.34[-0.01, 0.70] CI) 26 Std. Mean Difference(IV, Fixed, 95% -0.39[-1.16, 0.39] CI) 15 Std. Mean Difference(IV, Fixed, 95% 0.31[-0.72, 1.33] CI) 239 194 19 26 1.9 ADL scales 4 260 1.10 Behaviour, general 8 416 1.10.1 CAPE - BRS 4 231 1.10.2 Crichton BRS (modified) 2 29 1.10.3 MOSES Self-care 1 19 1.10.4 Instrumental ADL 1 137 1.11 Behaviour, problem 3 166 1.11.1 Problem Behaviour Rating 1 Scale 10 1.11.2 MOSES - Irritable 1 19 1.11.3 NPI 1 137 1.12 Caregiver outcome 3 1.12.1 Hamilton anxiety 2 150 1.12.2 Depression 2 150 1.12.3 Carer stress/burden 2 147 1.12.4 General Health Questionnaire (GHQ-12) 1 10 Std. Mean Difference(IV, Fixed, 95% 0.05[-0.21, 0.31] CI) Std. Mean Difference(IV, Fixed, 95% 0.01[-0.28, 0.30] CI) Std. Mean Difference(IV, Fixed, 95% -0.01[-0.98, 0.96] CI) Std. Mean Difference(IV, Fixed, 95% 0.38[-0.40, 1.16] CI) Std. Mean Difference(IV, Fixed, 95% 0.21[-0.05, 0.47] CI) Std. Mean Difference(IV, Fixed, 95% 0.13[-0.07, 0.32] CI) Std. Mean Difference(IV, Fixed, 95% 0.12[-0.14, 0.38] CI) Std. Mean Difference(IV, Fixed, 95% 0.33[-0.42, 1.07] CI) Std. Mean Difference(IV, Fixed, 95% 0.00[-0.97, 0.97] CI) Std. Mean Difference(IV, Fixed, 95% 0.12[-0.22, 0.46] CI) Std. Mean Difference(IV, Fixed, 95% -0.14[-0.44, 0.17] CI) Std. Mean Difference(IV, Fixed, 95% 0.40[-0.86, 1.66] CI) Std. Mean Difference(IV, Fixed, 95% 0.12[-0.85, 1.09] CI) Std. Mean Difference(IV, Fixed, 95% -0.20[-0.54, 0.13] CI) Std. Mean Difference(IV, Fixed, 95% Subtotals only CI) Std. Mean Difference(IV, Fixed, 95% 0.11[-0.21, 0.44] CI) Std. Mean Difference(IV, Fixed, 95% 0.04[-0.28, 0.36] CI) Std. Mean Difference(IV, Fixed, 95% -0.03[-0.35, 0.29] CI) Std. Mean Difference(IV, Fixed, 95% 0.94[-0.41, 2.29] CI) 2 Cognitive stimulation versus no cognitive stimulation: follow-up Outcome or Subgroup Studies Participants Statistical Method 2.1 Cognition 4 2.1.1 One to three months follow3 up Information/ Orientation 52 2.1.2 Ten months follow-up MMSE 1 54 2.1.3 Ten months follow-up ADAS1 Cog 54 Effect Estimate Std. Mean Difference(IV, Fixed, 95% Subtotals only CI) Std. Mean Difference(IV, Fixed, 95% 0.57[0.01, 1.14] CI) Std. Mean Difference(IV, Fixed, 95% 0.18[-0.35, 0.72] CI) Std. Mean Difference(IV, Fixed, 95% 0.12[-0.41, 0.66] CI) 46 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 2.2 Well-being & Quality of Life 2 1 10 1 54 2.3 Behaviour, general 3 2.3.1 One month follow-up 2 29 2.3.2 Ten months follow-up Texas 1 Functional Living Scale 54 2.4 Behaviour, problem 2 1 10 1 54 1 54 2.2.1 One month follow-up Life Satisfaction Index 2.2.2 Ten months follow-up QoLAD 2.4.1 One month follow-up Problem Behaviour Rating Scale 2.4.2 Ten month follow-up NPI severity 2.4.3 Ten-month follow up NPI (Caregiver Distress) 2.5 Communication and social 2 interaction 2.5.1 One month follow-up Holden 1 Communication Scale 2.5.2 Ten month follow-up 1 'Relevance of discourse' 10 54 Std. Mean Difference(IV, Fixed, 95% Subtotals only CI) Std. Mean Difference(IV, Fixed, 95% -0.03[-1.27, 1.21] CI) Std. Mean Difference(IV, Fixed, 95% 0.34[-0.19, 0.88] CI) Std. Mean Difference(IV, Fixed, 95% Subtotals only CI) Std. Mean Difference(IV, Fixed, 95% 0.44[-0.30, 1.18] CI) Std. Mean Difference(IV, Fixed, 95% 0.43[-0.11, 0.97] CI) Std. Mean Difference(IV, Fixed, 95% Subtotals only CI) Std. Mean Difference(IV, Fixed, 95% 0.39[-0.87, 1.65] CI) Std. Mean Difference(IV, Fixed, 95% 0.29[-0.24, 0.83] CI) Std. Mean Difference(IV, Fixed, 95% 0.41[-0.13, 0.95] CI) Std. Mean Difference(IV, Fixed, 95% Subtotals only CI) Std. Mean Difference(IV, Fixed, 95% 0.20[-1.05, 1.44] CI) Std. Mean Difference(IV, Fixed, 95% 0.15[-0.39, 0.68] CI) Figures Figure 1 (Analysis 1.1) 47 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation, outcome: Cognition. Figure 2 (Analysis 1.3) 48 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation, outcome: ADAS-Cog. Figure 3 (Analysis 1.2) Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation, outcome: MMSE. Figure 4 (Analysis 1.4) Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation, outcome: Other cognitive measure: Information/Orientation. Figure 5 (Analysis 1.5) 49 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation: post-treatment, outcome: Comunication and social interaction. Figure 6 (Analysis 1.6) Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation, outcome: QoL-AD. Figure 7 (Analysis 1.7) 50 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation, outcome: Geriatric Depression Scale (GDS30) Figure 8 (Analysis 1.8) Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation: post-treatment, outcome: Mood: Staffreported. Figure 9 (Analysis 1.9) Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation, outcome: ADL. Figure 10 (Analysis 1.10) 51 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation, outcome: Behavior, Other. Figure 11 (Analysis 1.11) Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation: post-treatment, outcome: Behaviour, problem. Figure 12 (Analysis 1.12) 52 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Caption Forest plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation: post-treatment, outcome: Caregiver outcome. Figure 13 (Analysis 2.1) Caption Forest plot of comparison: 2 Cognitive stimulation vs No Cognitive Stimulation: follow-up, outcome: Cognition. Figure 14 (Analysis 2.2) 53 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Caption Forest plot of comparison: 2 Cognitive stimulation vs No Cognitive Stimulation: follow-up, outcome: Well-being & Quality of Life. Figure 15 (Analysis 2.3) Caption Forest plot of comparison: 2 Cognitive stimulation vs No Cognitive Stimulation: follow-up, outcome: Behaviour, general. Figure 16 (Analysis 2.4) Caption Forest plot of comparison: 2 Cognitive stimulation vs No Cognitive Stimulation: follow-up, outcome: Behaviour, problem. Figure 17 (Analysis 2.5) 54 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia Caption Forest plot of comparison: 2 Cognitive stimulation vs No Cognitive Stimulation: follow-up, outcome: Communication and social interaction. Figure 18 (Analysis 1.1) Caption Funnel plot of comparison: 1 Cognitive Stimulation vs No Cognitive Stimulation: post-treatment, outcome: 1.1 Cognition. Sources of support Internal sources Bangor University, UK University College London, UK External sources NIHR, UK EA was supported by the Support at Home - Interventions to Enhance Life in Dementia (SHIELD) project (Application No RP-PG-0606-1083) which is funded by the NIHR Programme Grants for Applied Research funding scheme. Feedback Appendices 1 Search: December 2011 Source Search strategy 55 / 69 Hits retrieved Cognitive stimulation to improve cognitive functioning in people with dementia 1. ALOIS (www.medicine.ox.ac.uk/alois) (all dates) cognitive stimulation OR reality orientation OR 139 memory therapy OR memory groups OR memory support OR memory stimulation OR global stimulation OR cognitive psychostimulation 2. MEDLINE In-process and other non-indexed citations and 1. exp Dementia/ MEDLINE 1950-present (Ovid SP) 2. Delirium/ 3. Wernicke Encephalopathy/ 4. Delirium, Dementia, Amnestic, Cognitive Disorders/ 5. dement*.mp. 6. alzheimer*.mp. 7. (lewy* adj2 bod*).mp. 8. deliri*.mp. 9. (chronic adj2 cerebrovascular).mp. 10. ("organic brain disease" or "organic brain syndrome").mp. 11. ("normal pressure hydrocephalus" and "shunt*").mp. 12. "benign senescent forgetfulness".mp. 13. (cerebr* adj2 deteriorat*).mp. 14. (cerebral* adj2 insufficient*).mp. 15. (pick* adj2 disease).mp. 16. (creutzfeldt or jcd or cjd).mp. 17. huntington*.mp. 18. binswanger*.mp. 19. korsako*.mp. 20. or/1-19 21. "cognitiv* stimul*".mp. 22. "reality orientation".mp. 23. (memory adj2 therapy).mp. 24. "memory group*".mp. 25. "memory support".mp. 26. (memory adj2 stimulat*).mp. 27. "global stimulation".mp. 28. ("cognitive psycho-stimulation" or "cognitive psychostimulation").mp. 29. *Psychomotor Performance/ 30. or/21-29 31. 20 and 30 32. (2010* OR 2011*).ed. 33. 31 and 32 34. randomized controlled trial.pt. 35. controlled clinical trial.pt. 36. randomized.ab. 37. placebo.ab. 38. drug therapy.fs. 39. randomly.ab. 40. trial.ab. 41. groups.ab. 56 / 69 40 Cognitive stimulation to improve cognitive functioning in people with dementia 42. or/34-41 43. (animals not (humans and animals)).sh. 44. 42 not 43 45. 33 and 44 3. EMBASE 1. exp dementia/ 1980-2011 Dec 05 (Ovid SP) 2. Lewy body/ 239 3. delirium/ 4. Wernicke encephalopathy/ 5. cognitive defect/ 6. dement*.mp. 7. alzheimer*.mp. 8. (lewy* adj2 bod*).mp. 9. deliri*.mp. 10. (chronic adj2 cerebrovascular).mp. 11. ("organic brain disease" or "organic brain syndrome").mp. 12. "supranuclear palsy".mp. 13. ("normal pressure hydrocephalus" and "shunt*").mp. 14. "benign senescent forgetfulness".mp. 15. (cerebr* adj2 deteriorat*).mp. 16. (cerebral* adj2 insufficient*).mp. 17. (pick* adj2 disease).mp. 18. (creutzfeldt or jcd or cjd).mp. 19. huntington*.mp. 20. binswanger*.mp. 21. korsako*.mp. 22. CADASIL.mp. 23. or/1-22 24. "cognitiv* stimul*".mp. 25. "reality orientation".mp. 26. (memory adj2 therapy).mp. 27. "memory group*".mp. 28. "memory support".mp. 29. (memory adj2 stimulat*).mp. 30. "global stimulation".mp. 31. ("cognitive psycho-stimulation" or "cognitive psychostimulation").mp. 32. *psychomotor performance/ 33. or/24-32 34. 23 and 33 35. (2010* OR 2011*).em. 36. 34 and 35 4. PsycINFO 1. exp Dementia/ 1806-November week 5 2011 (Ovid SP) 2. exp Delirium/ 3. exp Huntingtons Disease/ 4. exp Kluver Bucy Syndrome/ 5. exp Wernickes Syndrome/ 57 / 69 29 Cognitive stimulation to improve cognitive functioning in people with dementia 6. exp Cognitive Impairment/ 7. dement*.mp. 8. alzheimer*.mp. 9. (lewy* adj2 bod*).mp. 10. deliri*.mp. 11. (chronic adj2 cerebrovascular).mp. 12. ("organic brain disease" or "organic brain syndrome").mp. 13. "supranuclear palsy".mp. 14. ("normal pressure hydrocephalus" and "shunt*").mp. 15. "benign senescent forgetfulness".mp. 16. (cerebr* adj2 deteriorat*).mp. 17. (cerebral* adj2 insufficient*).mp. 18. (pick* adj2 disease).mp. 19. (creutzfeldt or jcd or cjd).mp. 20. huntington*.mp. 21. binswanger*.mp. 22. korsako*.mp. 23. ("parkinson* disease dementia" or PDD or "parkinson* dementia").mp. 24. or/1-23 25. "cognitiv* stimul*".mp. 26. "reality orientation".mp. 27. (memory adj2 therapy).mp. 28. "memory group*".mp. 29. "memory support".mp. 30. (memory adj2 stimulat*).mp. 31. "global stimulation".mp. 32. ("cognitive psycho-stimulation" or "cognitive psychostimulation").mp. 33. "psychomotor performance".mp. 34. or/25-33 35. 24 and 34 36. random*.mp. 37. trial.mp. 38. placebo.mp. 39. group*.mp. 40. exp Clinical Trials/ 41. or/36-40 42. 35 and 41 43. (2010* OR 2011*).up. 44. 42 and 43 58 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 5. CINAHL (EBSCOhost) S1 (MH "Dementia+") S2 (MH "Delirium") or (MH "Delirium, Dementia, Amnestic, Cognitive Disorders") S3 (MH "Wernicke's Encephalopathy") S4 TX dement* S5 TX alzheimer* S6 TX lewy* N2 bod* S7 TX deliri* S8 TX chronic N2 cerebrovascular S9 TX "organic brain disease" or "organic brain syndrome" S10 TX "normal pressure hydrocephalus" and "shunt*" S11 TX "benign senescent forgetfulness" S12 TX cerebr* N2 deteriorat* S13 TX cerebral* N2 insufficient* S14 TX pick* N2 disease S15 TX creutzfeldt or jcd or cjd S16 TX huntington* S17 TX binswanger* S18 TX korsako* S19 S1 or S2 or S3 or S4 or S5 or S6 or S7 or S8 or S9 or S10 or S11 or S12 or S13 or S14 or S15 or S16 or S17 or S18 S20 TX "cognitiv* stimul*" S21 TX "reality orientation" S22 TX memory N2 therapy S23 TX "memory group*" S24 TX "memory support" S25 TX memory N2 stimulat* S26 TX "global stimulation" S27 TX "cognitive psycho-stimulation" OR "cognitive psychostimulation" S28 (MM "Psychomotor Performance") S29 S20 or S21 or S22 or S23 or S24 or S25 or S26 or S27 or S28 S30 S19 and S29 S31 EM 2010 S32 EM 2011 S33 S31 or S32 S34 S30 and S33 S35 TX random* S36 (MH "Clinical Trials+") S37 AB group S38 TI study S39 S35 or S36 or S37 or S38 S40 S34 and S39 59 / 69 34 Cognitive stimulation to improve cognitive functioning in people with dementia 6. Web of Science (1945-present) (WOK) Topic=(dement* OR alzheimer* OR "lew* bod*" OR 135 deliri* OR creutzfeldt OR cjd OR jcd OR huntington* OR binswanger* OR korsako*) AND Topic=("cognitiv* stimul*" OR CST OR "reality orienation" OR "memory therapy" OR "memory group*" OR "memory support" OR "psychomotor performance" OR "global stimulation" OR "cognitive performance") AND Topic=(random* OR trial* OR RCT OR "cross-over" OR cross-over) AND Year Published=(2010-2011) Timespan=All Years. Databases=SCI-EXPANDED, SSCI, A&HCI, CPCI-S, CPCI-SSH. Lemmatization=On 7. LILACS (BIREME) "cognitiv$ stimul$" OR "reality orienation" OR 16 "memory therapy" OR "memory group$" OR "memory support" OR "psychomotor performance" OR "global stimulation" OR "cognitive performance" [Words] and dementia OR alzheimer$ OR demenc$ OR AD OR demência [Words] 8. CENTRAL (The Cochrane Library) (Issue 3 of 4, Oct 2011) #1 dement* #2 alzheimer* #3 deliri* #4 chronic adj2 cerebrovascular #5 (lewy* bod*) #6 "organic brain disease" or "organic brain syndrome" #7 (pick* disease) #8 creutzfeldt or jcd or cjd #9 huntington* #10 binswanger* #11 korsako* #12 (#1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11) #13 "cognitiv* stimul*" #14 "reality orientation" #15 "memory therapy" #16 "memory group*" #17 "memory support" #18 "memory stimulat*" #19 "global stimulation" #20 "cognitive psycho-stimulation" #21 "cognitive psychostimulation" #22 MeSH descriptor Psychomotor Performance explode all trees #23 (#13 OR #14 OR #15 OR #16 OR #17 OR #18 OR #19 OR #20 OR #21 OR #22) #24 (#12 AND #23) #25 (#24), from 2010 to 2011 60 / 69 35 Cognitive stimulation to improve cognitive functioning in people with dementia 9. Clinicaltrials.gov (www.clinicaltrials.gov) #1 Intervention: Cognitive stimulation AND Interventional studies AND First rec: 01/01/201012/05/2011 = 30 51 #2 Intervention: reality orientation AND Interventional studies AND First rec: 01/01/2010-12/05/2011 = 1 #3 Interventional Studies | dementia OR alzheimers OR AD OR alzheimer's OR alzheimer OR lewy OR FTLD OR FLD | memory therapy OR memory training | received from 01/01/2010 to 12/05/2011=20 10. ICTRP Search Portal (http://apps.who.int/trialsearch) [includes: Australian New Zealand Clinical Trials Registry; ClinicalTrilas.gov; ISRCTN; Chinese Clinical Trial Registry; Clinical Trials Registry – India; Clinical Research Information Service – Republic of Korea; German Clinical Trials Register; Iranian Registry of Clinical Trials; Japan Primary Registries Network; Pan African Clinical Trial Registry; Sri Lanka Clinical Trials Registry; The Netherlands National Trial Register] Advanced search: (dementia OR alzheimer OR 86 alzheimers OR alzheimers) AND (cognitive stimulation OR reality orientation OR memory therapy OR memory training OR cognitive training) AND (2010-2011) TOTAL before de-duplication 804 TOTAL after de-dupe and first-assess 41 Graphs 1 - Cognitive stimulation versus no cognitive stimulation: post-treatment 61 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 62 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 63 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 64 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 65 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 66 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 2 - Cognitive stimulation versus no cognitive stimulation: follow-up 67 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 68 / 69 Cognitive stimulation to improve cognitive functioning in people with dementia 69 / 69