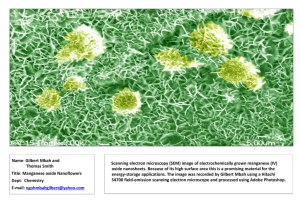
AN ABSTRACT OF THE THESIS OF
advertisement

AN ABSTRACT OF THE THESIS OF Sudarshan Lai Sabharwal (Name) in Title: Horticulture (Major) for the Doctor of Philosophy (Degr&e) presented on ^^/^i^/^^ /^// //^ /jj&te) RESPONSE OF STRAWBERRY PLANTS TO MANGANESE NUTRITION Abstract approved: Ralph Garren, Jr. The effects of manganese nutrition on growth, mineral composition, carbohydrate and nitrogen metabolism, IAA oxidase and phenols of 'Northwest' strawberry plants are reported. Growth and mineral composition response were investigated in sand culture and Hoagland's solution 2 modified to contain 0. 0, 0. 5, 125. 0 ppm substrate manganese. 2. 5, 12. 5, 25. 0, 62. 5 and The remaining aspects of the study were conducted using the Hoaglands solution in a hydroponics system with manganese levels of 0. 0, 0. 5, 2. 5, 1 2. 5 and 25. 0 ppm in the nutrient solution. In sand culture, symptoms of manganese toxicity occurred first in old leaves after three weeks of 125. 0 ppm Mn treatment; after five weeks at 62. 5 ppm; and after 13 weeks at 25. 0 ppm substrate Mn. Tox- icity symptoms appeared as necrotic spots along the margins of leaves; these spots enlarged greatly within a few days and ultimately killed all the leaves. All the plants treated with 62. 5 and 125. 0 ppm Mn died within 7 and 11 weeks, respectively. Young leaves of runners from the plants treated with 0. 0 ppm Mn developed interveinal chlorosis and cupping after 12 weeks. Later, these leaves developed chlorotic spots which expanded to chlorotic bands. Plant weight and total leaf area of the plants harvested after 20 weeks was greatest with 2. 5 ppm Mn treatment, and decreas-e.d both at lower or higher substrate manganese. The most leaves and runners were produced at 25. 0 ppm treatment; whereas area per leaf was highest at 0. 5 ppm Mn. Manganese present in tissue was directly correlated to substrate manganese levels and was also related to the plant part. Accumulation of manganese occurred in all tissues at high substrate Mn levels, indicating that the tolerance of strawberry plants to high Mn is not because of low uptake but due to their ability to endure large amounts of accumulated Mn. In Mn deficient plants, the differences in the con- centration of Mn in the various tissues were small, suggesting the possibility of redistribution of the element under conditions of deficiency. Highest levels of tissue iron were found in 0. 0 ppm Mn treatment followed in order by 2. 5, 0. 5, 1 2. 5 and 25. 0 ppm substrate Mn. A strong inverse relationship was noted between the tissue iron/ manganese ratio and the substrate Mn. The deficiency and excess of Mn resulted in a decrease in the concentration of Mg and P in the leaves . Highest levels of these elements were observed with 2. 5 ppm Mn treatment. The most vigorous plants were produced at 2. 5 ppm Mn treatment when old and young leaves contained 955. 7 and 617.4 ppm Mn, respectively. Typical toxicity symptoms were shown by plants when Mn in young leaves reached about 4, 000 and in old leaves 9, 000 ppm. In the hydroponic system, manganese deficiency and toxicity resulted in a decrease in sugar as well as starch content in the leaves, except in old leaves at 25. 0 ppm Mn treatment which contained the highest levels of starch. Total and insoluble N increased whereas nitrate N decreased with the increase in substrate Mn. Lowest levels of soluble and soluble reduced N were found at 2. 5 and 0. 5 ppm Mn treatment, respectively. Both the excess and deficiency of Mn resulted in the accumulation of amino acids. The highest concentrations of nitrate and lowest concen- trations of insoluble N observed in Mn deficient plants indicate that the Mn is not involved in nitrate N absorption but may be concerned in protein synthesis. High IAA oxidase activity was found in enzyme extracts of young and old leaves of deficient and toxic plants. Total and bound phenols, in general, were directly related to substrate manganese. Low levels of free phenols were found in conditions of excess and deficiency of Mn. The increase in the level of LAA oxidase activity at high level of tissue Mn was accompanied by simultaneous decrease in the concentration of o-dihydroxyphenols. These results indicate the possible involvement of phenols in the inhibition of IAA oxidase in the leaves of strawberry plants and manganese seems to affect the levels of the inhibitor(s). Response of Strawberry Plants to Manganese Nutrition by Sudarshan Lai Sabharwal A THESIS submitted to Oregon State University in partial fulfillment of the requirements for the degree of Doctor of Philosophy June 1972 APPROVED: " v^-c-T -y^--— ^T faO^. tf fc-M-JC" ■■■^r Professor of Horticulture in charge of major •y^ »• •^ Head of Department of Horticulture ^^ Dean of Graduate School Date thesis is presented ^/yy^V g/(>\. /<?"7/ Typed by Barbara Eby/fpg Sudarshan Lai Sabharwal ACKNOWLEDGEMENT I would like to express my sincere appreciation to Dr. Ralph Garren, Jr. for his interest, guidance, encouragement and support during the entire graduate program and to Dr. O. C. Compton for the suggestions and inspiration throughout the period of this study. I would like to express my sincere thanks to committee members Dr. N. I. Bishop, Dr. R. H. Converse, and Dr. T. L. Jackson and to Dr. E. Hansen for thoughtful criticism of the manuscript. I would also like to express my thanks and appreciation to the staff members of the Department of Horticulture for their guidance, cooperation, and help during my period of study. TABLE OF CONTENTS INTRODUCTION GROWTH AND MINERAL COMPOSITION RESPONSE OF STRAWBERRY PLANTS TO MANGANESE NUTRITION Materials and Methods Plant material Sand culture studies Purification of nutrient solution Sampling of plants Chemical analysis Results Manganese toxicity and deficiency symptoms Percentage of leaves shewing deficiency or toxicity symptoms Effect of substrate manganese on plant growth Concentration and distribution of tissue manganese Concentration and distribution of tissue iron Iron manganese ratio and distribution Concentration and distribution of tissue magnesium Concentration and distribution of tissue phosphorous Discussion Literature Cited 1 PART I. INFLUENCE OF MANGANESE NUTRITION ON CARBOHYDRATE AND NITROGEN METABOLISM OF STRAWBERRY LEAVES Materials and Methods Water culture studies Analytical methods Results and Discussion Carbohydrate content Total nitrogen Insoluble and soluble nitrogen Soluble reduced and nitrate nitrogen Amino acids Literature Cited 2 3 3 4 5 5 6 6 6 8 8 11 13 13 16 16 18 23 PART II. 26 29 29 29 30 30 33 33 35 36 41 PART III. INFLUENCE OF MANGANESE NUTRITION ON IAA OXIDASE AND PHENOLS OF STRAWBERRY LEAVES Materials and Methods Solution culture studies Extraction and purification for assay of IAA oxidase LAA oxidase assay Assay for co-factor Extraction for determination of phenols Estimation of phenols Determination of manganese Results and Discussion Tissue manganese Characterization of IAA oxidase enzyme assay Effect of Mn on IAA oxidase activity of dialyzed extract Effect on phenols Interaction of tissue Mn, o-dihydroxyphenols and IAA oxidase Literature Cited 45 47 47 47 48 49 49 49 50 50 50 50 55 58 61 63 LIST OF FIGURES Figure 1. 1 Page Effect of manganese on the growth of strawberry plants. 10 Effect of manganese on the magnesium content in strawberry plants. 17 Effect of manganese on the phosphorous content in strawberry plants. 17 Carbohydrate content in relation to manganese nutrition. 32 Nitrogen content in relation to manganese nutrition. 32 Soluble and insoluble nitrogen in relation to manganese nutrition. 34 Soluble reduced and nitrate nitrogen in relation to manganese nutrition. 34 IAA oxidase activity of dialyzed and nondialyzed extracts of strawberry leaves. 52 3. 2 Effect of time of dialysis on enzyme activity. 52 3. 3 Effect of enzyme concentration on oxygen uptake. 54 3. 4 Effect of co-factors on IAA oxidase activity. 54 3. 5 pH optimum of IAA oxidase of strawberry leaves. 56 3. 6 Effect of substrate IAA on IAA oxidase activity. 56 3. 7 Effect of substrate manganese on IAA oxidase activity of strawberry leaves. 57 1. 2 1. 3 Z. 1 2. 2 2. 3 2. 4 3. 1 Figure 3. 8 3. 9 3. 10 Page Effect of substrate manganese on total and bound phenols of strawberry leaves. 59 Effect of substrate manganese on free phenols of strawberry leaves. 59 Interaction of tissue manganese, o-dihydroxyphenol and IAA' oxidase. 62 LIST OF TABLES Table 1. 1 1. 2 1. 3 1. 4 2. 1 2. 2 3. 1 Page Percentage of leaves showing deficiency or toxicity symptoms. 9 Effect of manganese on the tissue manganese of strawberry plants. 12 Effect of manganese on the tissue iron of strawberry plants. 14 Effect of substrate manganese on the iron/ manganese ratio of strawberry plants. 15 Influence of substrate manganese on the amino acids in young strawberry leaves. 37 Influence of substrate manganese on the amino acids in old strawberry leaves. 39 Manganese content of young and old leaves. 51 RESPONSE OF STRAWBERRY PLANTS TO MANGANESE NUTRITION INTRODUCTION A wide variation in the concentration of manganese in strawberry leaves has been observed in Oregon from samples taken from various locations. The effect of this on the growth and yield of the strawberry is not known. The purpose of this study was to investigate the relation- ship between growth and biochemical status of strawberry plants grown in media varying from deficient to toxic levels of manganese. Since manganese has been observed to act as an antagonist or synergist to other elements, the mineral composition status of other elements was also examined. The dissertation is divided into three parts. In Part I, the effects of substrate manganese on 'Northwest' strawberry plants are described. Since the reports on manganese deficiency and/or toxicity for the crop are limited, the symptoms of deficiency and toxicity are verified, and the optimum requirements of Mn indicated. In Part II, the changes in carbohydrate and nitrogenous compounds as affected by substrate manganese are examined. The concentration of these compounds is correlated with levels of the nutrient manganese. In Part III, the interrelationship of tissue manganese, IAA oxidase and phenols is investigated in an attempt to explain growth differences in plants grown at manganese levels ranging from deficient to toxic. GROWTH AND MINERAL COMPOSITION RESPONSE OF STRAWBERRY PLANTS TO MANGANESE NUTRITION Strawberries are grown successfully on soils that vary widely in texture and reaction, yet little information is available on the relationships between growth and tissue manganese. Poor plant growth has been reported due both to low and excess tissue manganese (24, 25). A number of workers have shown that the availability of manganese in soil increases or decreases as soil acidity increases or decreases. Mulder and Gerretsen (26) stated that soils with pH less than 5. 5 contain most of their manganese in a water-soluble or exchangeable form. However, as the pH of the soil is increased, the manganese is converted to the less available manganic oxide. Most manganese toxicity occurs on highly acid soils. Hewitt (10) stated that the deleterious effects of high acidity were the results of a complex of factors of which manganese toxicity was most importa'nt. Fried and Peech (7) indicated that poor plant growth on acid soils was primarily because of the toxic effects of soluble manganese and not necessarily a lack of calcium. It has been shown that the plants grown in a solution or sand culture containing toxic quantities of Mn developed abnormalities similar to those observed in plants grown on acid soils (1, 2, 16, 25). The manganese content of strawberry leaves growing under acidic conditions is often very high, and yet the plants sho-w no signs of abnormalities. Mn levels up to 1. 8% have been observed without visible symptoms of toxicity (29). Lohnis (21) stated that the tolerance to high available Mn may be caused by either weak absorption, as observed in mustard and marigold, or to strong tolerance within the plant tissue as appears to be true for strawberry, potato and probably for broadbeans. Gerloff et al. (8) also concluded that species with high concen- trations of Mn in their tissues showed tolerance of the element. Lohnis (20) found that the strawberry is not only resistant to Mn toxicity but also to Mn deficiency. Johanson and Walker (18) observed the first deficiency symptoms in solution cultures after 88 days; whereas, Dennis and O'Brien (5) noticed growth retardation after two months in sand culture and symptoms of deficiency after four months. The present work was undertaken to study the effects of substrate and tissue manganese on growth and foliage-nutrient composition of 'Northwest' strawberry plants. Materials and Methods Plant material. All the strawberry plants for this study were of the cultivar Northwest and "certified" free of diseases and pests by the Oregon State Department of Agriculture. Before separating the plants into different weight categories, they were washed with tap water; the leaves were removed; and the roots were trimmed to four inches. Plants handled as above and -weighing six to eight grams were held in storage at 32 F for four months prior to use. At the initiation of the experiment, plants were removed from storage and grown in the greenhouse at 75 F day and 60 F night temperature with a 16-hour day length. Plants were grown in 11 liter plastic containers filled with distilled water and aerated at a constant pressure. The water in the containers was changed twice daily. Sand culture studies. Plants grown for 15 days under the above described conditions, having 2-3 leaves, were selected for uniformity and transferred to two-gallon, glazed crocks having a single drainage hole. Crocks were filled with acid-washed quartz sand after the manner of Hewitt (11). Five plants were planted in each container, which was individually connected to an 18 liter, gray-painted glass bottle containing Hoagland Solution #2, minus manganese. The plants were irrigated automatically, by a time-clock mechanism, four times a day by pumping air into the 18 liter bottles for 14 seconds at a time. Mn treatments were started ten days after transplanting into the sand. The levels of substrate manganese as MnSO were 0. 0, 0. 5, 2. 5, 12. 5, 25. 0, 62. 5, and 125. 0 mg per liter of solution. treatments were replicated four times. The seven Nutrient solutions were adjusted to pH 5. 5 and were changed fortnightly to assure a constant supply of nutrients. Iron was supplied as iron tartrate, at the rate of 1 ml per liter (1 ppm), twice a week, and the level of solutions was 1 • Supplied as amendments to the Hoagland Solution #2. brought to the 18 liter mark. Iron was not supplied as chelated iron because it has been reported that chelated iron interferes with Mn uptake (35). Purification of nutrient solution. The zero level of manganese used in the study required that all nutrient salts be purified. The macro-nutrient stock solutions were purified by the calcium carbonate phosphate coprecipitation procedure (11). Further removal of man- ganese was carried out by extraction with 8-hydroxyquinoline in chloroform at pH 7 (12). Iron was purified by a modified isopropyl ether extraction technique described by Piper (27). Ferric chloride was dissolved in concentrated hydrochloric acid and extracted three times with isopropyl ether. The iron was recovered from the ether fraction by adding deionized water and heating to drive off the ether. The solution was then analyzed for iron and brought to volume by the addition of five grams of tartaric acid and five milliliters of concentrated H SO per five grams of pure ferric chloride. Other micro-nutrients ■were purified by recrystallization. Sampling of plants. Blossoms were removed as they occured, but all runners were permitted to develop. The plants were observed twice a week for deficiency and toxicity symptoms in relation to substrate levels. The time of appearance and the distribution of the symp- toms was recorded. One plant from each pot was harvested after every four weeks for observations on roots. The last plants 'were harvested after ZO weeks of treatment. Samples were washed in EDTA (ethylene diamine tetra acetic acid) detergent solution; rinsed with demineralized water; separated into roots, petioles and leaves; and dried in an oven at 65 C. total weight. The dry -weight of each fraction was combined to give the The roots, petioles and leaves were ground separately. The area of the leaves was determined by the method described by Darrow and Waldo (4) using the length and width measurements of each leaflet. Chemical analysis. The oven-dried samples were ground in a Wiley mill to pass through a 40 mesh screen. An aliquot of 0. 25 to 1. 0 gram of each sample was weighed and ashed in an electric muffle o o furnace at 300 C for one hour and then at 55 0 C for three hours. Ashed samples were cooled, taken up in 0. IN HCl, digested with 10 ml of 2. 5 HCl and brought to volume in 0. IN HCl. The determination of Mg, Mn and Fe was made with an atomic absorption spectrophotometer. Phosphorous was determined co.lori.metrica.Uy by the molybdate method (6). Results Manganese toxicity and deficiency symptoms. Plants grown at the 125. 0 ppm Mn level died after 7 weeks, while the plants at the 62. 5 ppm Mn level survived up to 11 weeks. Thus, the analytical results are largely confined to the 0. 0, 0. 5, 2. 5, 12. 5, and 25. 0 ppm Mn treatments. Symptoms of manganese toxicity developed three weeks after treatment with 125. 0 ppm Mn. Symptoms of toxicity were also observed in plants with 62. 5 and 25. 0 ppm manganese treatment after 5 and 13 weeks, respectively. The first deficiency symptoms were noticed in some plants after 12 weeks in 0. 0 ppm (minus) manganese treatment. No symptoms, either of toxicity or deficiency, were observed in plants at substrate manganese levels of 0. 5 (control), 2. 5 and 1 2. 5 ppm. Symptoms of toxicity appeared first in old leaves as small necrotic spots around the margin of leaves and enlarged greatly within a few days. Necrotic spots appeared also around the mid rib. The marginal necrosis extended inward, and ultimately killed the whole leaf. The deficiency symptoms which developed first in the young leaves of the runner plants were interveinal chlorosis and cupping of leaves. The leaves developed chlorotic spots which expanded to form the chlorotic band. No deficiency symptoms were observed in leaves arising from the parent plant throughput the duration of the experiment. The substrate Mn also affected the development of roots, and there were practically no new roots after 4 and 8 weeks in plants treated with 125. 0 and 62. 5 ppm Mn, respectively. In these two treat- ments, roots taken at various intervals showed a remarkable correlation between their browning and the number of leaves showing toxicity 8 symptoms. Slight browning of roots was also observed in 16 and 20- week old plants treated with 25. 0 ppm manganese. Percentage of leaves showing deficiency or toxicity symptoms. The percentage of leaves showing toxicity symptoms was directly related to the concentration of substrate manganese, and the rate of appearance of toxicity symptoms was highest at high substrate manganese concentrations (Table 1. 1). Within four weeks of the appear- ance of the first toxicity symptoms, 100 percent of the leaves were affected in plants treated with 125. 0 ppm Mn, while in other plants treated with 62. 5 ppm Mn showed toxicity symptoms after six weeks. On the other hand, only 11. 0% of leaves showed toxicity even after six weeks in plants treated with 25. 0 ppm Mn. However, there was not much change in the percentage of leaves showing deficiency symptoms even after six weeks of the appearance of the first deficient leaf. Effect of substrate manganese on plant growth. Plant vigor implies a well balanced growth which can be quantatized by various indices. In this investigation leaf and runner number, area of leaves, dry plant weight and tissue analysis were used to measure quantitative growth. Increase in substrate Mn resulted in an increase in the number of leaves and runners as shown in Figure 1. la. Leaf numbers increased subs'tantiallly with each increment of added Mn but only the highest Mn levels produced significantly more runners. The increase Table 1.1. Substrate Manganese (ppm) Percentage of leaves showing deficiency or toxicity symptoms. 0 Percentage Leaves Affected Weeks After the Appearance of First Symptom 1 2 3 4 5 6 Toxicity Symptoms 125.0 5.4 18.8 47.5 81.4 100.0 62. 5 2. 5 5.6 21.0 37.8 69.0 92.5 100. 0 25. 0 0. 5 1.9 3. 7 9.5 13.0 12.4 11. 0 4.2 5.2 5.5 Deficiency Symptoms 0. 0 0.5 1.2 2.0 3.1 Figure 1.1a. Effect of manganese on the number of runners and leaves in strawberry plants. Vertical lines indicate L. S. D. at . 05 level. Figure 1.1b. Effect of manganese on leaf area and dry weight of strawberry plants. Vertical lines indicate L. S. D. at . 05 level. oo <► o> TOTAL LEAF AREA Cm2xlOO ro LEAF AREA (Cm2) PLANT WEIGHT (gm) ro OJ 01 ro ^ oo o NUMBER OF LEAVES NUMBER OF RUNNERS 01 11 in the number of leaves and runners seems to be a direct response to the increase in the level of substrate Mn. The data on size of leaf, leaf area per plant and total dry weight per plant are shown in Figure 1.1b. Aside from a slight but non- significant increase at 0. 5 ppm Mn, leaf size decreased progressively with increase in substrate Mn, signficance being attained only at the two highest levels. Total leaf area per plant was greatest at 2. 5 ppm Mn and decreased with either lower or higher Mn. Changeis in total dry weight per plant were similar to those for total leaf area. The most vigorous plants were produced at 2. 5 ppm Mn. Concentration and distribution of tissue manganese. The man- ganese content of all the fractions increased with each increase in substrate Mn (Table 1. 2). The old leaves treated with 1 2. 5 and 25. 0 ppm Mn accumulated 2 to Zj times more manganese than the young leaves, while in the remaining treatments they accumulated only 1. 5 times more Mn. The concentration of Mn in the petioles was lowest of all the fractions sampled. The 1 2. 5 and 25. 0 ppm treatments produced signif- icantly higher petiole Mn than did 0. 0, 0. 5 and 2. 5 ppm Mn. However, the Mn content of petioles at the highest substrate level was only 3 times the control; whereas, in leaves the range was 10 (young leaves) to 1 6 (old leaves) times the control. 12 Table 1. 2. Substrate Manganese (ppm) Effect of manganese on tissue manganese of strawberry plants. Manganese Content of Tissue (ppm) Old Leaves Young Leaves Petiole Roots 0. 0 283. 0 e 203. 4 e 206. 5 c 368. 0 d 0.5 641.5 d 450.3 d 220.4 c 418.9 d 2.5 955.7 c 617.4 c 284.0 c 590.7 c 12.5 2645.6 b 1252.8 b 401.9 b 775.4 b 25.0 9691.1 a 4308.0 a 628.1 a 1825.7 a Each figure is the mean of four replications. 2 Means followed by the same letter in each column are not significantly different at the 0. 05 level of probability. 13 In 2. 5, 1 2. 5 and 25. 0 ppm substrate manganese, the roots contained significantly more Mn than the 0. 0 and 0. 5 ppm Mn treatments. The roots of the minus Mn treatment contained 368. 0 ppm Mn as compared to 418. 9 ppm for the control. Roots of plants treated with 2. 5, 12. 5 and 25. 0 ppm Mn contained 1.4, 1.9 and 4. 4 times the Mn as in the control. Concentration and distribution of tissue iron. Decreasing the substrate Mn from 0. 5 to 0. 0 resulted in an increase in the iron content of leaves and roots and a decrease in the iron content of petioles (Table 1. 3). Also, an increase in substrate Mn to 2. 5 ppm resulted in an increase in the iron content of leaves (young and old) and petioles but a small decrease in the iron content of the roots. Any further increase in substrate levels of Mn resulted in an appreciable decrease in iron content of all the fractions sampled. The highest concentration of iron was found in roots, followed by old leaves, young leaves and petioles, except for the 25. 0 ppm Mn treatment where young leaves contained more iron than the old leaves. Iron manganese ratio and distribution. A strong inverse relation- ship was noted between the tissue iron/manganese ratio and the substrate Mn level (Table 1.4). The highest iron/.manganese ratio always occurred in the root fraction regardless of the substrate Mn level. Next in order was young leaves, followed by old leaves and the petioles. 14 Table 1. 3. Substrate Manganese (ppm) Effect of manganese on tissue iron of strawberry plants. Old Leaves 0.0 1301.2 0.5 651.9 2.5 I a 2 Iron Content of Tissue (ppm) Young Lea/ves Petiole Roots 821.0 a 137. 4 ab 3958.7 a b 582.7 b 163.7 a 3894.6 a 702.5 b 602.4 b 177. 2 a 3863.2 a 12.5 481.0 c 375.1 c 144. 2 ab 3654.9 ab 25.0 201.4 d 243.7 d 109.0 b 3486.0 b Each figure is the mean of four replications. 2 Means followed by the same letter in each column are not significantly different at the 0. 05 level of probability. 15 Table 1.4. Substrate Manganese (ppm) Effect of manganese on the iron/manganese ratio of strawberry plants. Fe/Mn Ratio Nutrient Solution Old Leaves Plant Fractions Young Petiole Leaves Roots 0.0 200.00 4.60 4.04 0.67 10.76 0. 5 2.00 1. 02 1. 29 0. 74 9. 30 2.5 0.40 0.73 0.98 0.62 6.54 12. 5 0. 08 0. 18 0. 30 0. 36 4. 71 25. 0 0. 04 0. 02 0. 06 0. 17 1. 91 Each figure is the mean of four replications. Figure 1. 2. Effect of manganese on the magnesium content in strawberry plants. Vertical lines indicate L. S. D. at . 05 level. Figure 1. 3. Effect of manganese on the phosphorous content in strawberry plants. Vertical lines indicate L, S. D. at . 05 level. 17 oOLD LEAVES ^YOUNG LEAVES AROOTS»PETIOLES .2 5^^ 0" 0.5 2.5 LOG Mn 12.5 25.0 12.5 25.0 (PPM) >8 0" 0.5 2.5 LOG Mn (PPM) 18 and Mg were similar in that they did not show wide variations among the different fractions. Discussion The work described above was designed to find the critical substrate and tissue manganese levels associated with the deficiency and toxicity symptoms in strawberry plants. Further studies were made to determine the effect of tissue manganese on growth and mineral composition of various fractions. The results showed that the minus and high substrate Mn levels restricted growth and altered the mineral composition. The visual symptoms of deficiency in strawberry plants are difficult to establish. The first symptom of deficiency was noticed after 12 weeks of minus manganese treatment while, after 18 weeks, only 5. 5% leaves showed deficiency symptoms, which developed only on the runner leaflets. Iwakiri and Scott (14) noticed no Mn deficiency symptoms in 'Temple' strawberry. . However, Johanson and Walker (18) reported the appearance of the first Mn deficiency symptom after 88 days on runner leaflets and within another two weeks in the intermediate leaves of parent plants. Dennis and O'Brien (5) observed small reddened and crinkled leaves after four months in sand culture; whereas, Lott (22) observed, similarly, yellowish-green coloration of leaves -with veins remaining darker green but with no purple stippling. 19 This effect, however, was more pronounced in old leaves (3). Lohnis (20) found that strawberry is resistant not only to Mn deficiency but also to Mn toxicity. The resistance to Mn toxicity seemed to be correlated with lower Mn uptake (3). However, when the strawberry plants were grown -with a high substrate Mn, the plants developed the toxicity symptoms which closely paralleled the characteristic chlorosis, necrotic spots and cupping of leaves observed in other crops. Plants showed the typical toxicity symptoms when Mn in young and old leaves reached about 4000 and 9000 ppm, respectively. Thus, the results indicate that the 'Northwest' strawberry is rather tolerant of high concentrations of substrate Mn not because of low uptake but because of its ability to endure large amounts of accumulated Mn(17, 20). Increase in substrate Mn from 0. 0 to 2. 5 ppm resulted in greatly increased growth as measured by the dry weight of plants, whereas decreases in growth were experienced with more than 2. 5 ppm. dry weight of the minus Mn plants was 71. 1% of the control. The The decrease in growth capacity was in part due to leaf chlorosis and to reduction in photosynthetic area (9). The reduction in growth of plants grown with 1 2. 5 and 25. 0 ppm Mn was correlated to excess Mn. Medappa and Dana (23) stated that dry weight of cranberry shoots was not affected by Mn concentration in the external solution; whereas, 20 Hiatt and Ragiand (13) and others (1, 32) observed that the dry weight was inversely related to Mn concentration in substrate or tissue. Numerous reports show that a direct relationship exists between substrate and tissue manganese. Results of this study were consistent with this relation (2, 16, 24, 26, 34). Manganese concentrations in different tissues were found to vary over a wide range. contained more Mn than roots and petioles. The leaves From the comparison of the tissue manganese between the 0. 0 and 0. 5 ppm Mn plants, this is evident that the difference in the amount of manganese required for good growth and that which results in deficient plants is small. addition, In these results indicate that as the substrate manganese be- came limiting, there was an increase in translocation of available manganese to the plant parts which were most active physiologically. This is supported by the observation that in the 0. 0 ppm Mn plants, there was not much difference in the levels of Mn in young and old leaves as well as in petioles and roots . Application of Mn to foliage has been shown to result in redistribution, mainly to the developing regions (30, 34). Jackson (5) concluded that the transfer of Mn from old leaves could occur but the rate of redistribution was very sluggish. The excess of Mn has been reported to hinder the translocation of iron by causing the iron in the plants to be converted to an insoluble form (31). It was suggested that, with a low level of Mn in the plant, iron is in the ferrous form and may cause iron toxicity, -while at a high 21 Mn level, iron may be in the ferric form and Fe deficiency may result (26). Hiatt and Ragland (13) found that substrate Mn did not change the concentration of iron in tobacco tissue. In this study, the inverse relationship between tissue Mn and iron, as reported by Somers and Shive (31), was found in all plant fractions except in leaves which contained little more iron at 2. 5 ppm Mn than in the control (0. Oppm). Rieckels and Lingle (28) found that increasing the Mn in the nutrient solution increased Fe uptake in tomato plants at low levels of Mn but decreased Fe uptake at high levels. Kirsch et al. (19) suggested that Mn does not interfere with iron utilization in the leaf but produces chlorosis by depressing the absorption of iron by the roots. An inverse relationship was found between the substrate Mn level and the iron/manganese ratio in the plant fractions studied. The ratio varied in the leaves from a value of more than 4. 6 with 0. 0 ppm substrate manganese to less than 0. 02 with 25. 0 ppm substrate manganese. The ratio in the roots was of a smaller order than that reported by Vlamis and Williams (3 3) for rice roots. However, the ratio only varied by a factor of 230 and 67 in the young and old leaves of the strawberry compared to as much as 1000 times in rice leaves. Somers and Shive (31) found that for good growth (without any deficiency symptoms) in soybean, the Fe/Mn ratio could only fluctuate slightly from 2. 0 in the nutrient solution. Iron/Mn ratios as high as 200 (minus manganese) and as low as 0. 04 (25. 0 ppm Mn) were imposed 22 in this experiment, but the detrimental effects (symptoms of deficiency and toxicity) were not observed in leaves when the Fe/Mn ratio ranged from 0. 08 to 2. 0 in the nutrient solution. This indicates that in 'Northwest' strawberry the substrate balance between Mn and Fe in the nutrient solution is not as critical as for the soybean. Similar to these results, Johanson (17) reported that Mn deficiency reduced the phosphorous level in the leaf blade of strawberry by 50 percent and magnesium by 35 percent. According to Lohnis (20, 21), there is a clear uptake antagonism between Mn and Mg, so that the addition of Mg prevents the appearance of injury symptoms in a .medium with an excess of manganese. In the results presented above, highest levels of P and Mg were observed when the Mn in leaves ranged from 617. 4 (young leaves) to 995. 7 (old leaves) ppm. Also, the maximum plant growth, as measured by the plant dry weight, was observed at these levels of Mn in the leaves. 23 LITERATURE CITED 1. Berger, K. C. , and G. C. Gerloff. 1947. Manganese toxicity of potato in relation to strong soil acidity. Soil Sci. Soc. Amer. Proc. 12:310-314. 2. Bolle-Jones, E. W. 1955. The effect of varied nutrient levels on the concentration and distribution of manganese with potato plant. Plant Soil 6;45-60. 3. Boyce, B. R. , and D. L. Matlock. 1967. Strawberry nutrition, p. 518-598. In: N. F. Childers [ed. ] Nutrition of Fruit Crops. Rutgers-the State University, New Brunswick, N. J. 4. Darrow, G. M. , and G. F. Waldo. 1932. Effect of fertilizers on plant growth, yield and decay of strawberries in North Carolina. Proc. Amer. Soc. Hort. Sci. 29:318-324. 5. Dennis, R. W. G. , and D. G. O'Brien. 1937. Boron in agriculture. West Scot. Agr. College, Plant Husbandry Dept. Res. Bui #5, p. 98. 6. Fiske, C. H. , and Y. Subbarow. 1925. The co.lorimetric determination of phosphorous. Jour. Biol. Chem. 66:375-400. 7. Fried, M. , and M. Peech. 1946. The comparative effects of lime and gypsum upon plants grown in acid soils. J. Amer. Soc. Agron. 38:614-623. 8. Gerloff, G. C. , D. G. Moore, and J. T. Curtis. 1966. Selective absorption of mineral elements by native plants of Wisconsin. Plant and Soil 25:39 3-405. 9. Gerretsen, F. C. 1949. Manganese in relation to photosynthesis. Plant and Soil 1:346-35 8. 10. Hewitt, E. J. 195 2. A biological approach to the problems of soil acidity. Trans. Congr. Intern. Soc. Soil Sci. (Dublin) 1:107-117. 11. Hewitt, E. J. 1966. Sand and water culture methods used in the study of plant nutrition. Technical Communication No. 22. Commonwealth Bureau of Horticulture and Plantation Crops, East Mailing, Maidstone, Kent. p. 547. 24 12. Hewitt, E. J. , and E. W. Jones. 1947. The production of molybdenum deficiency in plants in sand culture with special reference to tomato and brassica crops. J. Pomol. 23:254-262. 13. Hiatt, A. J. , and J. L. Ragland. 1963. burley tobacco. Agron. J. 55;47-49. 14. Iwakiri, B. , and L. E. Scott. 1951. Mineral deficiency symptoms of the Temple strawberry grown in sand culture. Proc. Amer. Soc. Hort. Sci. 57:45-52. 15. Jackson, W. A. 1967. Physiological effects of soil acidity. p. 43-124. In: R. W. Pearson and F. Adams [ed. ] Soil acidity and liming. Monograph 12. Amer. Soc. Agro. Madison, Wisconsin. 16. Jackobson, H. G. M. , and T. R. Swanback. 1932. Manganese content of certain Connecticut soils and its relation to growth of tobacco. J. Amer. Soc. Agron. 24:237-245. 17. Johanson, F. D. 1963. Nutrient deficiency symptoms, p. 7580. In: C. R. Smith and N. F. Childers [ed. ] The Strawberry. Rutgers-the State University, New Brunswick, N. J. 18. Johanson, F. D. , and R. B. Walker. 1963. Nutrient deficiency and foliar composition of strawberries. Proc. Amer. Soc. Hort. Sci. 83:431-439. 19. Kirsch, R. K. , M. E, Harward.and R. G. Peterson. I960. Interrelationships among iron, manganese and molybdenum in the growth and nutrition of tomato grown in culture solution. Plant Soil 12:259-275. 20. Lohnis, M. P. 1950. Symptoms of Mn toxicity in cultivated crops. T. N. O. Nieuws 5:150-155. 21. Manganese toxicity of ' Lohnis, M. P. I960. Effect of magnesium and calcium supply on the uptake of manganese by various crop plants. Plant Soil 12:339-376. 22. Lott, W. L. 1946. Strawberries need small amounts of Mn and Zn. N. Car. Agr. Exp. Sta. Ann. Rept. 69, p. 66. 23. Medappa, K. C. , and M. N. Dana. 1970. Tolerance of cranberry plants to manganese, iron and aluminum. J. Amer. Soc. Hort. Sci. 95:107-110. 25 ?4. Morris,, H. D. 1969. The soluble manganese content of acid soils and the relation to growth and manganese content of sweet clover and lespedeza. Soil Sci. Soc. Amer. Proc. 13:362-371. 25. Morris, H. D. , and W. H. Pierre. 1949. Minimum concentrations of manganese necessary for injury to various legumes in culture solutions. Agron. J. 41:107-113. 26. Mulder, E. G. , and F. C. Gerretsen. 195 2. Soil manganese in relation to plant growth, p. 221-227. In: A. G. Norman [ed. ] Advances in Agronomy Vol. IV. Academic Press Inc. N. Y. 27. Piper, C. S. 1941. Marsh spot of peas. disease. J. Agric. Sci. 31:448-452. 28. Riekels, J. W. , and J. C. Lingle. 1966. Iron uptake and translocation by tomato plants as influenced by root temperature and manganese nutrition. Plant Physiol. 41:1095-1101. 29. Roberts, A. N. , and A. L. Kenworthy. 1956. Growth and composition of the strawberry plant in relation to root temperature and intensity of nutrition. Proc. Amer. Soc. Hort. Sci. 68:157-168. 30. Romney, E. M. , and S. J. Toth. 1954. Plant and soil studies with radioactive manganese. Soil Sci. 77:107-117. 31. Somers, L I., and J. W. Shive. 1942. The iron-manganese relation in plant metabolism. Plant Physiol. 17:5 82-602. 32. Sutton, C. G. , and E. G. Hallsworth. 1958. Studies on the nutrition of forage legumes. I. Toxicity of low pH and high manganese supply to lucerne as affected by climatic factors and calcium supply. Plant Soil 9:305-317. 33. Vlamis, J. , and D. E. Williams. 1964. Iron and manganese relations in rice and barley. Plant and Soils 20:221-231. 34. Vose, P. B. 1963. The translocation and redistribution of manganese in Avena. J. Exp. Bot. 14:448-45 7. 35. "Wallace, A. 1958. Effect of chelated iron and manganese on the manganese content of soybeans grown in solutions culture. Agro. J. 50:285-286. A manganese deficiency 26 INFLUENCE OF MANGANESE NUTRITION ON CARBOHYDRATE AND NITROGEN METABOLISM OF STRAWBERRY LEAVES The effects of manganese on the metabolism of plants are numerous. The metal serves as an activator or a co-factor for a wide variety of enzyme systems involved in carbohydrate and nitrogen metabolism (13, 25). Although the effects of Mn deficiency have been studied in a number of crops, information is lacking on the effects of Mn, ranging from near deficient to near toxic levels on the nitrogen and carbohydrate metabolism of strawberry plants. Manganese plays a primary role in photosynthesis and is involved mainly with oxygen evolution in photosynthesis (9, 18, 37). The inhibi- tion of photosynthesis in Mn deficient plants has been observed to be independent of any influence' oh chlorophyll level, and occurs in the early stage of manganese deficiency prior to any real chlorosis (27, 28). Manganese deficient plants are characterized by low levels of soluble carbohydrates (37). Reduction in levels of starch and sugar were found in Mn deficient leaves of oats and tomatoes (10, 32, 37); also Hewitt (12) observed that total carbohydrate in normal chlorella cells was higher than in Mn deficient cells. In the leaves, Mn pro- motes the development of an effective carbohydrate apparatus only during the early formative period; and a deficiency at that time cannot be made good by the subsequent addition of more Mn (27, 37). 27 Mn is also believed to be involved in the chain of reactions beginning with the reduction of NO plants. and ending in protein synthesis in higher Burstrom (4) concluded that Mn deficiency resulted in nitrate accumulation for lack of a carbon skeleton due to a decrease in carbohydrate supplies following impaired photosynthesis. Nance (23) sug- gested that Mn was involved in reduction of nitrite rather than nitrate. Later it was shown that Mn is involved as a constituent of the flavoprotein of hydroxy.la.mine reductase (17, 24). Steward and Margolis (34) concluded that Mn is not implicated in reduction of NO immediate conversion to organic compounds. or its Mn does promote a rapid build-up of soluble N compounds through its effects on the supply of N acceptor compounds via the citric acid cycle. In Mn deficient plants,,the assimilation of both NO disturbed. Because of Mn deficiency,NO and NH is assimilation in algae was inhibited (30); whereas it accumulated to abnormally high levels in cauliflower leaves (14). Accumulation of soluble N compounds was also observed in plants deficient in Mn (16, 27, 33). Cain and Holly (5) found, in lime-induced chlorotic leaves of blueberries, a higher accumulation of soluble nitrogenous compounds, as compared to green leaves. A number of workers have reported the accumulation of amino acids in Mn deficient plants. Bar-Akiva (2) found that Mn deficient citrus leaves accumulated more free arginine, asparagine, alanine 28 and serine. Accumulations of aspartic acid and proline and a reduction in "y-amino butyric acid were also observed in citrus leaves (35). Gilfillan _et al. (11) found that Mn deficient chlorotic leaves of macadamia contained significantly higher amounts of homoserine; whereas Labanauskas e_t al. (19) reported a significantly higher accumulation of free aspartic acid, threonine, giutamic acid, proline, alanine, valine, isoleucine, leucine, tyrosine and phenyla.lanine. Scheffer et al. (31), working with oats and wheat, observed phenyla.lanine and histidine only in normal plants; whereas alanine was totally absent in Mn deficient plants, and proline was found only in plants showing toxicity of Mn. Lljin (16) studied N metabolism of various fruit plants affected by lime-induced chlorosis and found that 3 to 7 times more amino acids accumulated in the chlorotic leaves than in green leaves. However, a decrease in total free amino acids was observed by Lewis e_t al. (20) in pear leaves in conditions of Mn deficiency and excess. The effects of Mn deficiency or excess on the carbohydrate and N metabolism, of plant's, in general, and strawberry plants in particular, are still obscure. The purpose of this study was to evaluate the effects of Mn deficiency and excess in strawberry leaves on the carbohydrate and N fractions of plants grown in water culture. 29 Materials and Methods Water culture studies. Strawberry plants as described in a pre- vious experiment were transplanted in ■water culture solution after growing 15 days in distilled water (29). Five young plants were trans- planted to each 11 liter colored plastic container. Each container was fitted with a wooden lid having five holes drilled in a circular pattern and plants were placed through the holes and supported by packed, sterile glass wool. through an air stone. immediately. The solutions were aerated with filtered air passed The manganese treatments were imposed The substrate levels of manganese were 0. 0, 0. 5, 2. 5, 12. 5 and 25. 0 pprn with three replications in each treatment. The nutrient solution, made from purified salts (29), was completely changed after a week and iron was supplied as iron tartrate on the 3rd and 5th day of change of solutions (15). trusses were removed periodically. Runners and flower Plants were harvested after 84 days, immediately fractionated into young and old leaves, and lyophilized. Analytical methods. with 100 ml of 80% ethanol. Samples of 0. 5 to 1. 0 gram were extracted The alcohol fraction was made to 25 0 ml volume and stored under refrigeration for further analysis. The con- centrations of nitrate N, soluble reduced N, total soluble N, and soluble carbohydrates 'were determined in the alcohol fraction. 30 Total hot-water-soluble carbohydrate (starch) was determined from the oven-dried residue of 80% ethanol extract. The mixture of 25-50 mg of dried residue, plus 2 ml 80% ethanol and 25 ml of water, was placed on a steam bath for 30 minutes. This was transferred quantitatively to a centrifuge tube and centrifuged. was brought to 100 ml by addition of water. The supernatant Carbohydrates were deter- mined colorimetrically by the phenol sulfuric acid method (7, 22). Total N was determined by Kjeldahl, the reduced soluble N by the micro Kjeldahl and NO (6). - N by the phenol disulfonic acid method The total soluble N was obtained by adding the nitrate and soluble reduced N. The total insoluble N was the difference between total N and soluble N. For the determination of free amino acids, the leaf material was extracted according to the method of Thompson et aU (38). The individ- ual amino acids in each sample were then separated and quantitatively determined by the Technicon Amino Acid Autoanalyzer using two buffers as outlined by Efron (8). Results and Discussion Carbohydrate content. Both the substrate Mn concentration and the type of leaf affected the 80% ethanol-soluble (sugar) and hot-watersoluble (starch) carbohydrate fractions of strawberry leaves harvested after 84 days of Mn treatment (Figure 2. 1 ). 31 The sugar content of young as well as old leaves increased with increases in substrate Mn but only up to 1 2. 5 ppm Mn treatment. The lowest sugar content was observed in Mn deficient leaves (0. 0 ppm). However, at low (0. 0 and 0. 5 ppm) and high (25. 0 ppm) substrate Mn, the sugar content was higher in old leaves than in young leaves. In young leaves, the starch content increased in treatments up to 2. 5 ppm Mn, but no definite trend was observed in old leaves. Young and old leaves in all Mn treatments contained substantially more starch than the minus Mn treatment. The low levels of sugars and starch observed in the Mn deficient leaves agree with the findings of other workers. Gerretsen (10) observed low levels of both sugar and starch in Mn deficient leaves of oats, while Miller (21) observed only low levels of soluble carbohydrate under similar conditions. The reduction in the content of carbo- hydrates in the Mn deficient leaves may have been due to low photosynthesis. A decrease in the amount of reducing sugar in potato and tomato on fertilization with Mn was observed by Abutalybov et al. .(1). The levels of Mn in these plants prior to treatment were not indicated. It may be that the treatment caused an excess of Mn and a decrease in carbohydrates. leaves, also. This effect of excess Mn is apparent in the strawberry Figure 2. 1. Carbohydrate content in relation to manganese nutrition. Figure 2. 2. Nitrogen content in relation to manganese nutrition. 32 IZ UJ -1 I 28 o UJ UJ 24 V) o o dzo 16 - -^ ^ YOUNG LEAVES o OLD LEAVES SUGAR STARCH r 0.5 2.5 12.5 LOG. 25.0 Mn (PPM) 3.3 r- 2.5 LOG. Mn 12.5 (PPM) 25.0 33 Total nitrogen. The N content was also affected by the type of leaf and substrate Mn levels (Figure 2. 2). Increase in the substrate concentration of Mn was associated with increase in the total N content of young as well as old leaves. However, the young leaves contained more total N than the old leaves at all levels of substrate Mn. The increases in total N were considerably greater when Mn was increased from 0. 0 to 2. 5 ppm than from 2. 5 to 25. 0 ppm. Voth ^t al. (39) also found a positive correlation between the total N and Mn in leaf tissue of a number of strawberry varieties. They attributed increases in Mn at high N application either to its release" from the exchange complex by the NH ions produced by nitrification. ions of the fertilizer or by H Later, they found the relationship true irrespective of soil pH (40). In the majority of treatments, the highest carbohydrate contents were at intermediate levels of leaf Mn and N, similar to the observations of Blatt (3). Insoluble and soluble nitrogen. Insoluble N increased with increases in the amounts of Mn in the substrate in young as well as old leaves (Figure 2. 3). In both types of leaves, the insoluble N was substantially lower in Mn deficient plants (0. 0 ppm). On the other hand, high levels of soluble N were observed both at low and high levels of substrate Mn. The lowest level of soluble nitrogen was at 2. 5 ppm. In all treatments, young leaves contained higher soluble and insoluble N than old leaves; the differences in the amount of insoluble N bet-ween Figure 2. 3. Soluble and insoluble nitrogen in relation to manganese nutrition. Figure 2. 4. Soluble reduced and nitrate nitrogen in relation to manganese nutrition. 34 i56 2.5 LOG Mn(PPM) O - \ x [\\ ro O a. / v ££ ^^^^ x tf " 0.5 s o s x s ' \ 37 cr en or -- ' S -'-$■'' 1 y \ ' "—^~——^ -* " ' ^~~~~~~~~~-*-^z-' ^ " ^ "* ^^^^*v\ ^ " ^^^*^^*»^ -"' ^-^ " X6^^ -*4S ' V \ \\ \\^ \V\ \\ \ 13 S.R.N \ A \ I A 40O " N \\ i - NO3-N - O O 25.0 YOUNG LEAVES OLD LEAVES ^ 17 K 12.5 1 H34 ^^^ 1 2.5 12.5 LOG Mn (PPM) 31 25.0 a. 35 the young and old leaves were smaller at 0. 0 ppm Mn. These results indicate that Mn deficiency resulted in a decrease in protein (insoluble N fraction) synthesis. Steward ej: aX. Nason et al.(25) and (32, 33) proposed that protein synthesis was affected adversely in the Mn deficient leaves of cauliflower and tomato. Higher concentrations of insoluble N in young leaves of cotton plants grown in minus Mn nutrient solution were found by Taylor (36) who proposed that the Mn •was not involved in protein synthesis. Accumulations of soluble N were also observed in tomato plants by Steward and Margolis (34) under conditions of Mn deficiency. Soluble reduced and nitrate nitrogen. High levels of soluble reduced N 'were found in leaf samples taken from plants treated -with low as well as high levels of Mn (Figure 2. 4). In young leaves, the lowest levels of soluble reduced N were found at 0. 5 ppm and in old leaves at 2. 5 ppm. The accumulation of soluble reduced N in leaves with Mn deficiency has been reported in cauliflower by Hewitt et aA. (14) and in tomato plants by Steward and Margolis (34). The levels of nitrate in the leaves were negatively correlated with substrate Mn and were higher in young than old leaves. et al. (14), working with caulifower, required for NO this conclusion. Hewitt concluded that Mn was not reduction, and Steward and Durzan (33) supported Jones et al. (17) showed that Mn was involved as a co-factor with hydroxy.lamine reductase. The high level of NO 36 observed at low Mn indicates that Mn is not involved in NO but in its utilization. A high turnover rate at high Mn levels resulted in the low level of NO Amino acids. absorption observed. The concentrations of free amino acids in young strawberry leaves were greatly increased by Mn deficiency or excess as shown in Table 2. 1. Mn deficient leaves contained more glycine, valine, aspartic acid, lysine, arginine, serine, phenylalanine, asparagine and glutamine than leaves with more Mn. The concentrations of alanine, threonine, glutamic acid, proline and Y-amino butyric acid increased slightly; whereas leucine, isoleucine, histidine, methionine and tyrosine decreased a little. Mn treatments above 0. 5 ppm produced variable influence on the content of free amino acids in strawberry leaves. There was a decrease in the concentration of glycine, leucine and arginine when the substrate Mn was increased to Z. 5 ppm. All other amino acids increased a slight amount. Further increase in substrate Mn resulted in an increase of all amino acids and the highest amounts were found in young leaves taken from the 25. 0 ppm Mn treatment. Table 2. 2 gives the results of the analysis of free amino acids in old leaves of strawberry. As in young leaves, the concentrations of amino acids in old leaves also were greatly increased by Mn deficiency and excess. Mn deficiency in old leaves resulted in an increase of all 37 Table 2. 1. Influence of substrate manganese on amino acids in young strav/berry leaves. Amino Acid 0.0 Substrate Manganese (ppm) 0.5 2.5 12.5 25.0 jiM/g glycine 2. 62 0. 79 0. 07 0. 69 0.97 alanine 10. 08 8. 28 6. 10 6. 01 13.41 valine 2. 15 0. 62 0. 94 2. 16 1. 84 leucine 1. 04 1. 27 1. 71 1. 11 1.92 isoleucine 0. 91 0. 96 0. 97 1. 60 1.88 threonine 3. 65 3. 31 3. 85 4. 07 5. 78 aspartic acid 7. 96 0. 51 0. 63 1. 05 7.90 glutamic acid 8. 79 5. 53 6. 13 6. 05 8. 19 lysine 0. 65 0. 06 0. 12 0. 45 0. 76 arginine 4. 69 1. 77 1. 60 4. 78 9.51 histidine 0. 09 0. 65 0. 83 1. 41 1.53 methonine 0. 10 0. 14 0. 14 0. 15 0. 21 proline 2. 04 1. 83 2. 23 2. 36 2.57 tyros ine 0. 14 0. 25 0. 25 0. 38 0. 39 20. 81 14. 28 16. 23 16. 84 18. 04 11. 45 3. 86 6. 32 10. 22 8.52 phenylalanine 0. 39 0. 05 0. 27 0. 32 0. 39 asparagine 8. 67 5. 23 4. 12 4. 92 5. 78 glutamine 5. 38 3. 17 3. 58 4. 69 7. 84 91. 66 52. 56 55. 46 69. 24 97.43 Y- amino butyric acid serine TOTAL Data given are average of three samples. 38 amino acids except leucine, arginine, histidine and tyrosine, which decreased slightly. The level of alanine, lysine, histidine, and tyrosine decreased when the substrate Mn concentration was increased from 0. 5 to Z. 5 ppm. Further increase in substrate Mn resulted in an increase of all free amino acids with very high concentrations at 25. 0 ppm Mn. Mn deficiency in strawberry leaves produced changes in the concentrations of free amino acids, which are partially in agreement with the results reported by Possingham (26), who found more aspartic acid, glutamic acid, asparagine, arginine, -y-amino butyric acid, valine and proline and less glutamine, histidine, lysine, serine, glycine, threonine, alanine, phenylalanine and leucine in Mn deficient tomato leaves. Hewitt et al. (14), Steward et al. (32, 33), and Labanauskas et al. (19) also reported similar results in experiments with other crops. Excess Mn in the plant tissue also produced an increase in all the amino acids. In contrast, Lewis et al. (20) reported increases only in glutamic acid, serine, and alanine and a decrease in arginine and asparagine in pear leaves grown with excess Mn. Scheffer et al. (31) also observed an accumulation of methionine, threonine, glutamic acid and proline and reduction of the remaining amino acids in leaves of wheat plants grown with 100 ppm Mn in the substrate. In strawberry leaves (Table 2. 1 and 2. 2) and also in other crops (16, 19),the total amino acid content increased tremendously under 39 Table 2. 2. Influence of substrate manganese on amino acids in old strawberry leaves. Amino Acid 0.0 Substrate Manganese (ppm) 0.5 2.5 12.5 25.0 M-M/g glycine 3.40. 0. 97 0. 69 1. 25 0. 97 alanine 19. 14 12. 32 10. 24 13. 01 19. 21 valine 3.98 1.61 1. 81 1. 84 3. 16 leucine 0.49 1. 28 1. 35 1.46 1.61 isoleucine 2. 16 1. 08 1.86 1. 48 1.97 threonine 7.94 4. 04 4. 38 4. 16 5. 01 aspartic acid 8. 14 3. 55 4. 05 4. 81 4.96 glutamic acid 3.64 2. 07 3. 15 2. 05 4. 35 lysine 1. 75 0. 86 0. 5 3 0. 89 0.94 arginine 0. 22 0. 73 0. 80 0.96 1. 26 histidine 0.0 3 0. 05 0. 01 0.04 0.07 methonine 0.46 0. 12 0. 13 0. 15 0. 17 proline 2. 68 1. 27 2.42 2.97 5.98 tyrosine 0. 27 0.41 0. 23 0.45 0. 71 21. 19 20. 76 20.99 22. 62 24. 04 11.03 9. 72 11. 16 11. 50 12. 05 phenylalanine 0. 51 0, 37 0.48 0. 56 0.69 asparagine 7.91 6.48 7. 64 9. 70 10. 77 g.lutamine 4. 83 3. 81 4. 57 5. 15 5. 89 82.19 85. 05 103. 81 •y amino butyric acid serine TOTAL 99. 77 71. 5 1 Data given are average of three samples. 40 conditions of deficiency and toxicity. The work of Lewis ej: al. (20) with pear leaves, on the other hand, found a decrease in the total amino acid content with an increase and a decrease in the Mn nutrition. Hewitt (12), reviewing the effects of micronutrients on the metabolism of free amino acids in plants, observed that a deficiency or toxicity of a plant nutrient could increase or decrease the contents of free amino acids in plants. Thus, the concentration of an individual amino acid or all amino acids cannot be used to distinguish between the deficiency or toxicity of Mn in leaves. 41 LITERATURE CITED 1. Abutalybov, M., I. Buniatov, and A. Mardanov. 1956. Importance of manganese in oxidation reduction processes in plant organism. Uchen. Zap. Azerbaidzh. Univ. No. 9, 47-57 [inter. Abst. Biol. Sci., 11:(2348), 1958]. 2. Bar-Akiva, A. 1964. Visible symptoms and chemical analysis vs. biochemical indicators as a means of diagnosing iron and manganese deficiencies in citrus plants, p. 9-24. In: C. Bould, P. Prevot, and J. R. Magness [ ed. ] Plant Analysis and Fertilizer Problems, IV. 3. B.latt, C. R. 1967. Response of 'Acadia' strawberry to two forms and three rates of nitrogen at two pH levels. Proc. Amer. Soc. Hort. Sci. 92;346-35 3. 4. Burstrom, H. 19 39. Uber die Schwermetallkatalyze der Nitratassimil'ation. Planta. 29;292-305. 5. Cain, J. C. , and R. W. Holly. 1955. A comparison of chlorotic and green blueberry leaf tissue with respect to free amino acid and basic cation contents. Proc. Amer. Soc. Hort. Sci. 65:49-53. 6. Chapman, H. D. , and P. F. Pratt. 1961. Methods of analysis for soils, plants and waters. University of California, Riverside, California. p. 309. 7. Dubois, M. , K. A. Gilles, J. K. Hamilton, P. A. Rebers, and F. Smith. 1956. Colorimetric method for determination of sugars and related substances. Anal. Chem. 28:350-356. 8. Efron, M. L. 1965. Quantitative estimation of amino acids in physiological fluids using a Technicon Amino Acid Analyzer. ' p. 637-642. In: L. T. Skegg [ ed. ] Automation in analytical chemistry. Technicon symposium. Mediad Incorporated, New York. 9. Eyster, C. , T. E. Brown, H. A. Tanner, and S. L. Hood. 1958. Manganese requirement with respect to growth, Hill reaction, and photosynthesis. Plant Physiol. 33:235-241. 42 10. Gerretsen, F. C. 1949. Manganese in relation to photosynthesis. I. Carbon dioxide assimilation and the typical symptoms of manganese deficiency in oats. Plant and Soil ls346-358. 11. Gilfillan, I. M. , and W. W. Jones. 1968. Effect of iron and manganese deficiency on the chlorophyll, amino acid and organic acid status of leaves of macadamia. Proc. Amer. Soc. Hort. Sci. 93:210-214. 12. Hewitt, B. R. 1959. Glucose assimilation in normal and manganese deficient chlorella cells. Physiol. Piantarum 12:45 2-455. 13. Hewitt, E. J. 1963. The essential nutrient elements: the requirements and interaction, p. 218-222. In: F. C. Steward [ ed. ] Plant Physiology: a treatise. III. Inorganic Nutrition of Plants. Academic Press, New York. 14. Hewitt, E. J. , E. W. Jone, and A. H. Williams. 1949. Relation of molybdenum and manganese to the free amino acid content of the cauliflower. Nature 163:681-682. 15. Hoagland, D. R. , and D. J. Arnon. 195 0. The water culture method for growing plants without soil. University of California Circ. 347:29-31. 16. Iljin, W. S. 1951. Metabolism of plants affected with limeinduced chlorosis. I. Nitrogen Metabolism. Plant and Soil 3:239-255. 17. Jones, L. H. , W. B. Shepardson, and C. A. Peters. 1949. The function of manganese in assimilation of nitrates. Plant Physiol. 24:300-306. 18. Kessler, E. 1955. On the role of manganese in the oxygen evolving system of photosynthesis. Arch. Biochem. Biophys. 59:527-529. 19. Labanauskas, C. K. , and M. F. Handy. 1970. The effects of iron and manganese deficiencies on accumulation of nonprotein and protein amino acids in macadamia leaves. Amer. Soc. Hort. Sci. 95:218-223. 20. Lewis, L. N. , N. E. Tolbert, and A. L. Kenworthy. 1963. Influence of mineral nutrition on the amino acid composition of Bartlett pear trees. Proc. Amer. Soc. Hort. Sci. 83:185-192. 43 21. Miller, L. P. 1933. Effect of manganese deficiency on the growth and sugar content of plants. Amer. J. Bot. 20:621-631. 22. Nalewaja, J. D, , and L. H. Smith. 1963. Standard procedure for the quantitative determination of individual sugars and total soluble carbohydrate materials in plant extracts. Argon. J. 55:523-525. 23. Nance, J. F. 1948. Role of oxygen in nitrate assimilation by wheat roots. Amer. J. Bot. 35:602-606. 24. Nason, A. 195 6. Enzymatic steps in the assimilation of nitrate and nitrite; in fungi and green plants, p. 109-136. In: W. D. McElroy and H. B. Glass [ ed. ] Inorganic nitrogen metabolism. Johns Hopkins Press, Baltimore, Maryland. 25. Nason, A., and W. D. McElroy. 1963. Modes of action of the essential mineral elements, p. 451-5 36. In: F. C. Steward [ ed. ] Plant physiology: a treatise. III. Inorganic Nutrition of Plants. Academic Press, New York. 26. Possingham, J. V. 1956. The effect of mineral nutrition on the content of free amino acids and amides in tomato plants. Austral. J. Biol. Sci. 9:539-551. 27. Prison, A. 1958. Manganese and its role in photosynthesis, p. 81-98. In: C. A. Lamb et al. [ ed. ] Trace elements. Academic Press, New York. 28. Ruck, H. C. , and B. D. Bolas. 1954. The effect of manganese on the assimilation and respiration rate of isolated rooted leaves. Ann. Bot. 18:267-297. 29. Sabharwal, S. L. , R. Garren, and O. C. Compton. 1971. Growth and mineral composition response of strawberry plants to manganese nutrition. (In Preparation). 30. Sadana, J. C. , and W. D. McElroy. 1957. Nitrate reductase from Achromobacter fischeri. Purification and properties: function of flavines and cytochrome. Arch. Biochem. Biophys. 67:16-33. 44 31. Scheffer, F., E. Przemeck, and V. K. Saolapurkar. 1970. Mineral nutrition and metabolic processes in young plants. II. Effects of manganese nutrition on the levels of free amino acids in oats and wheat. J. Ind. Soc. Soil Sci. 16; 135-141. 32. Steward, F. C. , F. Crane, K. Millar, R. M. Zacharius, R. Rabson, and D. Margolis. 1959. Nutritional and environmental effects on the nitrogen metabolism of plants. In; Utilization of Nitrogen and its Compounds of Plants, Symposia Soc. Exptl. Biol. No. 13:148-176. 33. Steward, F. C. , and D. J. Durzan. 1965. Metabolism of nitrogenous compounds, p. 397-602. In; F. C. Steward [ ed. ] Plant Physiology; a treatise, IV. A. Metabolism; Organic nutrition and nitrogen metabolism. Academic Press, New York. 34. Steward, F. C. , and D. Margolis. 1962. The effects of manganese upon free amino acids and amide of the tomato plants. Contrib. Boyce Thompson Inst. 21s393-409. 35. Stewart, L 1962. The effects of minor element deficiencies on free amino acids in citrus leaves. Proc. Amer. Soc. Hort. Sci. 81:244-249. 36. Taylor, D. 1965. The manganese nutrition of cotton. Thesis. Texas A and M University, p. 138. 37. Thomas, M. D0 1965. Photosynthesis (carbon assimilation): Environmental and metabolic relationship, p. 125-128. In: F. C. Steward [ ed. ] Plant Physiology: a treatise, IV. A. Metabolism: Organic nutrition and nitrogen metabolism. Academic Press, New York. 38. Thompson, J. F. , C. J. Morris, and R. K. Gering. 1959. Purification of plant amino acids for paper chromatography. Anal. Chem. 31:1028-1031. 39. Voth, V., E. L. Proebsting, and R. S. Bringhurst. 1961. Response of strawberries to nitrogen in Southern California. Proc. Amer. Soc. Hort. Sci. 78:270-274. 40. Voth, V., K. Uriu, and R. S. Bringhurst. 1967. Effect of high nitrogen application on yield, earliness, fruit quality and leaf composition of California strawberries. Proc. Amer. Soc. Hort. Sci. 91:249-256. Ph. D. 45 INFLUENCE OF MANGANESE NUTRITION OF IAA OXIDASE AND PHENOLS OF STRAWBERRY LEAVES Strawberry plants were observed to exhibit unusual growth response to deficient and toxic levels of manganese during an investigation of absorption and distribution of the metal (28). The quantita- tive plant growth as measured by the plant dry weight showed a decrease in plant vigor, both from deficiency and excess of manganese. Since the elongation of plant cells is known to be hormonally regulated, the amount of growth accomplished by the cell is quantitatively dependent on the concentration of such substances. Any factor influencing the concentration of one of these hormones,.such as IAA, would affect growth within cell, tissue or organ. Manganese and a number of phenolics are known to affect growth by their regulatory action on IAA oxidase both in vivo and in vitro (1, 10, 18, 20, 34, 37, 40). The role of Mn as a co-factor in the oxidation of IAA was first reported by Wagenknecht and Burris (39) and has since been observed by numerous workers (13, 14, 17, 29). Increases of two to three fold in the rate of IAA oxidation by horseradish peroxidase with additions of Mn have been reported (15). Goldacre (7) reported that Mn was an inhibitor of IAA oxidase; whereas Furuya and Galston (4) observed that the preincubation of pea tissue homogenates with Mn overcame the effects of IAA oxidase inhibitor (a g.lucoside of quercetin). 46 Goldacre, Galston and Weintraub (8) pointed out that IAA oxidation is usually activated by monophenols and inhibited by o-dipheno.ls, an observation since supported by numerous workers (1, 9, 21, 23, 30, 38, 40). Presumably, phenols which inhibit IAA oxidase activity have a sparing effect on the IAA and thereby promote growth and vice versa. Stutz (35) reported an obligate requirement for a phenolic co-factor and Mn in the IAA oxidase system of lupine and found Mn to be inactive in the absence of the phenolic co-factor. Kenten and Mann (15) stated that the manganous ions were oxidized to the manganic ions via a phenol by the peroxidase-peroxide system. In the system, Mn seems to function as a reducing agent for the oxidized peroxidase substrate, the Mn ion being subsequently reduced, possibly by IAA. Recently, Stonier et al„ (32) suggested that a high molecular weight auxin protector, found by them in Japanese morning glory and sunflower, and the phenolic compounds described by others are concerned with regulation of peroxidase catalyzed redox reactions. They further stated that the auxin protector (which is inactivated by Mn) acts as an antioxidant of IAA. Morgan et a.l. (22) reported an inverse correlation between high manganese and IAA oxidase inhibitor (gossypol andkaempferol) activity. The degree of IAA oxidase stimulation was associated with the severity of Mn toxicity. Taylor et al. (37) indicated that the biosynthesis or, at least, the activity of the native co- factors and inhibitors of IAA 47 oxidase system was influenced by the concentration of manganese in the leaf. Both high and low levels of Mn modified IAA oxidase inhibitor activity (22, 37). This suggests that there is a relationship among phenols, IAA oxidase and Mn nutrition of plants and would indicate an in vivo role for the enzyme; such a role is compatible with other evidence. This paper deals with the effects of manganese nutrition on IAA oxidase and phenols of strawberry leaves. Tissue manganese was varied by growing strawberry plants at substrate manganese concentrations ranging from deficient to toxic levels. The effects of the tissue Mn on IAA oxidase and phenols were tested with assay of enzyme, co-factors and total, bound, free and o-diphenols. Materials and Methods Solution culture studies. 'Northwest1 strawberry transplants were grown in solution culture in the greenhouse during 1969 and 1970, as described previously (28). Five treatments with five plants in each 11 liter plastic container were treated with 0. 0, 0. 5, 2. 5, 12. 5, and 25. 0 ppm Mn. The leaves were harvested after 84 days and divided into young and old leaves. The leaves for enzyme assay were kept in an ice bath or lyophilized for phenol and manganese determination. Extraction and purification for assay of IAA oxidase. Immedi- o ately on harvest, the leaves were homogenized in a cold room (40 F) 48 in four volumes (4 ml; 1 g fresh wt. ) of 0. 1 M KH PO -Na HPO Li Q Li Q buffer (pH 6.0), containing 0. 3M sucrose and 200 |j,g/m.l each of penicillin G and streptomycin sulfate, and then filtered through a double layer of cheesecloth. Crude homogenate was centrifuged at 10, 000 x g for 20 minutes at 40 F, and the resulting supernatant was used as enzyme extract. The homogenates were kept in an ice bath throughout the preparative procedure. One set of the prepared homogenates was dialyzed against a 200 fold volume of 0. 1M phosphate buffer (pH 6. 0) for 48 hours with three' changes of buffer at 12-hour intervals, except as noted otherwise; while the second set was not dialyzed. at 40 F in a dark room. The dialysis was carried out After dialysis the enzyme extracts were centrifuged as before and either assayed immediately or stored at a o temperature of 0 F. IAA oxidase assay. system (20, 29). IAA oxidase was assayed by a manometric The reaction mixtures as described by Taylor et a.l. (37) were used with slight modification. Each flask contained 0. 2 ml of 20% KOH in the center well with a filter paper wick, 1 ml containing SOJJIM of IAA in the side arm and 0. 5 ml of 0. 1M sodium, phospate (pH 6. 0), 0. 2|i.M of 2, 4-dichlorophenol, 0. 1 mM of MnCl , and 1 ml of leaf extract and deionized water in the main compartment to give a total volume of 3. 3 ml. Unless otherwise indicated, the assay was conducted at room temperature in light, after 15 minutes of equilibration. 49 Duplicate flasks of each extract were assayed. Assay for co-factor. The assay for co-factor activity was identi- cal to the IAA oxidase assay except that 2, 4-dichlorophenol was omitted from the reaction mixture. Appropriate blanks, containing boiled extracts, were run to determine oxygen uptake due to the constituents other than the co-factors. Extraction for determination of phenols. Freeze dried leaves were ground to pass a 40 mesh screen and extracted thrice with boiling 95% ethanol. The hot extract was filtered through a sintered glass and reduced to semi-dryness under vacuum. The residue was taken up in a small quantity of hot water for subsequent extraction of free and bound (acid and alkali hydrolysable) phenols (3, 12). Acid hydrolysis was performed using 2N HC1 and refluxing on a steam bath for an hour, and subsequently, extracted with ethyl ether in a continuous liquid-liquid extractor for 24 hours. Alkaline hydrolysis was carried out using 2N NaOH and the preparation allowed to stand at room temperature overnight in a nitrogen atmosphere and then acidified to pH 2. 0 with HCl prior to ether extraction (16). The ether extract of alkaline hydrolysates was washed with 5% NaHC O . The ether portion was discarded and the alkaline portion was acidified to pH 2. 0 and re-extracted with ether. Estimation of phenols. The phenolic content of strawberry leaves was estimated by the method of Rosenblatt and Peluso (27), 50 using the Folin-Denis reagent and calculated as chlorogenic acid. The Folin-Denis reagent used in this method lacks complete specificity for polyphenols but was used because of its simplicity and convenience. O-dihydroxyphenols were estimated by the method of Arnow (2). The Arnow reagent appears to be a modification of the Hoepfner reagent (11) and is specific for the o-dihydroxy configuration. Determination of manganese. Levels of manganese in the leaves were determined by atomic absorption spectrophotometer (28). Results and Discussion Tissue manganese. The Mn content of leaf fractions after 84 days varied from 129. 0 ppm of leaf tissue from the young leaves of plants grown with 0. 0 ppm Mn to 10,132. 5 ppm in old leaves in plants grown with 25. 0 ppm Mn (Table 3. 1). In all treatments, the concen- tration of Mn was higher in old than in young leaves. In a number of other plants Millikan and Romney et al. (18, 26) also found more Mn in mature than in younger leaves, and autoradiographic studies showed prominent localized areas of Mn concentrations in old leaves. Characterization of IAA oxidase enzyme assay. Enzyme extracts of young and old leaves of strawberry dialyzed for 48 hours showed a decrease in lag period and a substantial increase in O uptake when compared with similar non-dialyzed leaves (Figure 3. 1). O Low uptake was observed in non-dialyzed extracts of old as compared to 51 Table 1. Influence of substrate manganese on the manganese content of young and old leaves of strawberry. Substrate Manganese (ppm) Old Leaves (ppm) Young Leaves (ppm) 0.0 164. le 129. 0e 0.5 782. 4d 541. 2d 2. 5 1, ,137. 8 c 749. 1 c 12. 5 2, 9 36. 0 b 1, 448. 7 b 25. 0 10, 132. 5 a 4, 295. 4 a Each figure is the mean of five replications. 2 Means followed by the same letter in each column are not significantly different at the 0. 05 level of probability. Figure 3. 1„ IAA oxidase activity of dialyzed and nondia.lyzed extracts of strawberry leaves. Figure 3. 2. Effect of time of dialysis on enzyme activity. 52 200 C? o OLD DIALYZED • OLD NON DIALYZED a YOUNG DIALYZED A YOUNG NON DIALYZED 150 UJ CL 3 100 L±J O >X o 60 90 120 150 180 60 72 MINUTES 200r oOLD LEAVES ^ YOUNG LEAVES _• 150 UJ CL Z> 100 UJ o >X 50 o 12 24 36 48 DIALYSIS TIME - HOURS 53 young leaves. After dialysis the old leaves showed higher enzyme activity than young leaves as measured by O uptake. Decline in enzyme activity was also observed when dialysis was extended beyond 48 hours (Figure 3. 2). Taylor et al. (37) observed a similar lag in O uptake in old leaves of cotton. Dialysis for 72 hours removed heat- stable, endogenous inhibitors which were causing the delay in O uptake. At relatively low concentrations of enzyme extract, its activity was not proportional to concentration (Figure 3. 3). The enzyme activ- ity did increase from zero to 1. 0 ml of extract per reaction mixture. The deviation from direct proportionality at low enzyme levels may be due to product degradation (19, 29). In all other enzyme assay experi- ments, a concentration of 1. 0 ml (equivalent to 250 mg fresh weight of leaves) was used as a standard. Tang and Bonner (36) observed that at low concentrations of pea seedling enzyme, rate of reaction was proportional to enzyme concentration, while at high concentrations substrate became limiting. Morgan (20), working with cotton leaves, found 0. 2 ml of enzyme extract had a minimum lag period. Quantities above or below 0. 2 ml resulted in an increase in the lag period of O uptake. Both the dialyzed and undialyzed enzyme extract of strawberry leaves required Mn (supplied as MnCl^) and 2, 4-dichlorophenol for its maximum expression of activity (Figure 3. 4). In its requirement for Figure 3. 3. Effect of enzyme concentration on oxygen uptake. Figure 3„ 4. Effect of co-factors on IAA oxidase activity. 54 200 r o OLD LEAVES A YOUNG LEAVES ^ 150 LJ ?CL Z UJ CD > X 100 50 o .25 .50 .75 1.00 1.25 1.50 ENZYME EXTRACT (ml) 400 A o — D H300 A • UJ ■ CRUDE COMPLETE MINUS Mn. MINUS 2,4-D DIALYZED COMPLETE MINUS Mn. MINUS 2,4-D 0- 200 UJ o X "00 - o 120 180 MINUTES 360 55 Mn, enzyme extract in strawberry resembles the enzyme in bean and pineapple (9, 14, by even 10 -5 39) and differs from that in pea, which is inhibited M Mn (4). Phosphate borate buffer for determination of the pH optimum of the enzyme system of strawberry leaves was prepared as described by Moore (19). (Figure 3. 5). The optimum pH for the enzyme was found to be 6. 0 Strawberry leaf IAA oxidase differs strikingly from the enzyme in pineapple, having a pH optimum of 3. 0 (9) but is similar in range to IAA oxidase in peas and beans (36, 39). In separate experiments in which the concentrations of IAA were varied under otherwise optimum conditions (Figure 3. 6), the amount of IAA decarboxylation (measured as O uptake) was approximately a linear function of initial IAA concentration at low levels but became nonlinear at concentrations above 50 |JLM. This optimum concentration was far more than observed for enzyme extracted from cotton (21) but low as compared to enzyme obtained from omphalia and lupine (25, 35). Effect of Mn on IAA oxidase activity of dialyzed extract. The effect of different levels of nutrient Mn on the IAA oxidase activity of dialyzed extract of old and young leaves is shown in Figure 3. 7. At all manganese levels, IAA oxidase activity was found to be higher in the old than in the young leaves. Enzyme extracts of leaves from 25. 0 ppm Mn treated plants had the highest IAA oxidase activity. The order of activity of extracts from old leaves in the remaining treatments Figure 3. 5„ pH optimum of IAA oxidase of strawberry leaves. Figure 3„ 6. Effect of substrate IAA on IAA oxidase activity. 56 200, o OLD LEAVES a YOUNG LEAVES 150 UJ Q_ 100 3 LU O > X o 50 3.o 4.5 5.0 5.5 pH 6.0 6.5 7.0 60 70 200. o OLD LEAVES a YOUNG LEAVES "5. 150 UJ a. 3 100 UJ o 50 20 30 40 50 IAA CONCENTRATION ( pM) Figure 3. 7. Effect of substrate manganese on IAA oxidase activity of strawberry leaves. 57 300 /25.0PPM OLD LEAVES (PPM of Mn.) A 2.5 PPM ^^OOPPM 250 / / UJ 200 Q. 150 2.5PPM_^^ • UJ O >X 100 o 50 // ^^^^/^OSPPM l^^ ■ / / 15 i 30 45 200r i i 60 75 i /25.0PPM YOUNG LEAVES (PPMofMn.) / -5. 150-\ /- / 90 // /I2.5PPM ^^^6!OPPM UJ < 3 0.5 PPM/' 100 / / UJ o >X o ^^5PPM 50 eS*^ , 1 15 30 1 45 MINUTES 1 1 60 75 1 90 58 was 12. 5, 0. 0, 0. 5, and 2. 5 and in young leaves -was 12. 5, 0. 0, 2. 5 and 0. 5 ppm Mn. The differences in the enzyme activity between the extracts of old and young leaves from 0. 5 and 2. 5 ppm Mn treatments were small as compared to the other treatments. Results of this experiment are in agreement with the data of Taylor ej: al. (37) that a toxic as well as a deficient level of Mn in vivo results in higher activity of LAA oxidase enzyme system. Kenten et al. (15) and others (4, 10, 22, 29) in vitro tests observed an increase in the activity of IAA oxidase with added Mn; whereas Goldacre (7) reported that Mn was an inhibitor of IAA oxidase. The observed stimulation in IAA oxidase activity from toxic and deficient leaves may be considered as an acceleration of the normal aging process (6, 33). Taylor et al. (37) found that the deficient leaves needed no additional co-factor (2, 4-dichloropheno.l) for IAA oxidase activity; whereas the toxic leaves needed this co-factor but no additional Mn. Thus he concluded that the biosynthesis or, at least, the activity of the native co-factors and inhibitors of the IAA oxidase system was influenced by the concentration of manganese in the leaf, which probably is true in strawberry leaves. Effect on phenols. The relative amounts of free, bound and total phenols as affected by the substrate Mn concentration are shown in Figure (3. 8 and 3. 9). Both the total as well as bound phenol levels increased with an increase in the concentration of the substrate or tissue Figure 3. 8. Effect of substrate manganese on total and bound phenols of strawberry leaves. Figure 3. 9. Effect of substrate manganese on free phenols of strawberry leaves. 59 16 H 14 £ o • A A OLD LEAVES TOTAL PHENOLS OLD LEAVES BOUND PHENOLS YOUNG LEAVES TOTAL PHENOLS YOUNG LEAVES BOUND PHENOLS >Q |2 SX10 Q. E 8 C -5S- 0.5 2.5 12.5 25.0 12.5 25.0 LOG. Mn (PPM) 12 O o 10 >Q 8 ui X A YOUNG LEAVES 0.5 2.5 LOG. Mn (PPM) 60 manganese (Table 3. 1), except in 0. 5 ppm Mn treatments in which the old leaves contained less phenols (bound and total) than the old leaves from the 0. 0 ppm Mn treatment. In all treatments, the old leaves con- tained more phenols (total and bound) than young leaves. There were slight increases in the concentration of phenols when the concentration of substrate Mn was increased from 0. 0 to 2. 5 ppm. When substrate concentration of Mn was increased from 2. 5 to 25. 0 ppm, however, the increase in total and bound phenols was substantially more. In contrast to total and bound phenols, the highest levels of free phenols were observed at the intermediate substrate Mn in both young and old leaves. Also, the old leaves contained more free phenols at all levels of Mn substrate than young leaves. The presence of inhibitors of LAA oxidase in enzyme extracts obtained from various plants has been demonstrated by a number of workers (5, 9, 20, 21, 23, 29). Addition of boiled extract resulted in increased lag period (21, 22, 37), the duration of which was a function of the concentration of inhibitors. Maclachlan et_ al. (17) and Morgan (20) found that the activity of inhibitors followed total phenol levels. Morgan et al. (22) reported an inverse correlation between high Mn and IAA oxidase inhibitor, which they thought to be kaempferol. In this study, high IAA oxidase activity was observed both at deficient and toxic levels of manganese in the tissue, but correlations of total and bound phenols with IAA oxidase occurred only at concentrations 61 of Mn greater than 2. 5 ppm. A negative correlation was found in the levels of free phenols and IAA oxidase (Figure 3.7 and 3. 9). This indicates that free phenols are involved in the mechanism of IAA oxidation, and the observation is further supported by the fact that old leaves with higher IAA oxidase activity have a higher concentration of free phenols than do young leaves (Figure 3. 8). Interaction of tissue Mn, o-dihydroxyphenols and IAA oxidase. A short-term experiment was conducted to determine the relationship of tissue Mn, o-diphenols and IAA oxidase activity. Strawberry plants were grown with 125 ppm substrate Mn and leaf samples were taken at 5, 10, 15 and 20-day intervals. The levels of IAA oxidase activity and o-dihydroxyphenols increased slightly up to 15 days of Mn treatment, but later o-dihy- droxyphenols decreased and IAA oxidase, as indicated by oxygen uptake, increased substantially (Figure 3. 10). IAA oxidation is usually activated by monophenols and inhibited by o-diphenols (8). Recently, Stonier el al. (31, 32) stated that the auxin protector acts as an antioxidant of IAA. The auxin protector found by them in Japanese morning glory leaves as well as in sunflower shoots was a phenolic compound of very high molecular weight. Earlier, Furuya and Galston (4) considered a glucoside of quercetin to be an inhibitor of IAA oxidase. •with cotton, Morgan et al. (21, 22), working suggested that a low molecular •weight phenol, functioned Figure 3. 10. Interaction of tissue manganese, o-dihydroxyphenol and LAA oxidase. 62 -225 UJ Q_ UJ O >X o 15 (23694) (65670) TISSUE Mn (PPM) 63 as an inhibitor of IAA oxidase. The decrease in the level of o-dihy- droxyphenol at a high tissue level of Mn, accompanied by a simultaneous increase in the level of IAA oxidase, suggests that a phenol may be involved in the inhibition of IAA oxidase activity in leaves of strawberry. In this experiment no attempts were made to isolate and identify such a hypothetical compound. The study also indicates that the level of inhibition is functionally related to the tissue level of Mn. A more elaborate experiment is necessary to elicit this relationship. 64 LITERATURE CITED 1. Andreae, W. A., and G. Collet. 1968. The effect of phenolic substances on the growth activity of indoleacetic acid applied to pea root or stem sections. p. 553-561. In: F. Wightman and G. Setterfield [ ed. ] Biochemistry and Physiology of Plant Growth Substances. Proceedings of the 6th International Conference on Plant Growth Substances. The Runge Press, Ottawa. 2. Arnow, L. E. 19 37. Colorimetric determination of the components of 3, 4-dihydroxypheny.lalanine-tyrosine mixtures. Jour. Biol. Chem. 118:5 31-5 37. 3. Feldman, A. W. , and R. W. Hanks. 1966. Identification and quantification of phenolics in the leaves and roots of healthy and exocortis-infected citrus. Florida Agricultural Experiment Station Journal Ser. No. 2423. p. 11. 4. Furuya, M. , and A. W. Galston. 1961. Effect of in vitro preincubation with co-factors on the activity of the indoleacetic acid oxidase of peas. Physiol. Plantarum 14:750-766. 5. Furuya, M. , A. W. Galston, and B. B. Stowe. 1962. Isolation from peas of co-factors and inhibitors of indolyl- 3- acetic acid oxidase. Nature 193:456-457. 6. Galston, A. W. 1967. Regulatory systems in higher plants. Am. Scientist 55:144-160. 7. Goldacre, P. L. 1963. The indole-3-acetic acid oxidase-peroxidase of peas. Plant Growth Regulation. Fourth International Conference on plant growth regulation. The Iowa State University Press, Ames, Iowa, U.S.A. p. 143-147. 8. Goldacre, P. L. , A. W. Galston, and R. L. Weintraub. 195 3. The effect of substituted phenols on the activity of IAA oxidase of peas. Arch. Biochem. Biophys. 43:358-373. 9. Gortner, W. A., and M. Kent. 1953. Indoleacetic acid and, an inhibitor in pineapple tissue. J. Biology Chem. 204:59 3-603. 10. Hillman, W. S. , and A. W. Galston. 1956. Interaction of Mn and 2, 4-D in the enzymatic destruction of IAA. Plant Physiol. 9:230-325. 65 11. Hoepfner, W. 1932. Two new reactions for caffeic acid and chlorogenic acid. Chem-Zeit 56:991-993. 12. Ibrahim, R. K. , and G. H. N. Towers. I960. tion by chromatography of plant phenolic acids. Biochem. Biophys. 87;125-128. 13. Kenten, R. H. 1950. The oxidation of manganese by peroxidase systems. Biochem. J. 46s67-73. 14. Kenten, R. H. 1955. The oxidation of indolyl-3-acetic acid by wax pod bean root sap and peroxidase systems. Biochem. J. 59:110-121. 15. Kenten, R. H. , and P. J. G. Mann. 1952. Oxidation of manganese by enzyme systems. Biochem J. 52:125-130. 16. Li, C. Y. , K. C. Lu, J. M. T.rappe, and W. B. Bo.llen. 1971. Phenolic compounds in roots of Red Alder and Douglas fir. (In Press). 17. Maclachlan, G. A., and E. R. Waygood. 1956. Catalysis of indoleacetic acid oxidation by manganic ions. Physiol. Plantarum 9:321-330. 18. Mi.l.likan, C. R. 1950. Radio-autographs of manganese in plants. Australian J. Sci. Res. B4:28-41. 19. Moore, T. C. 1970. Comparative net biosynthesis of indoleacetic acid from tryptophan in cell-free extracts of different parts of Pi sum sativum plants. Phytochemistry 9:1109-1120. 20. Morgan, P. W. 1964. Distribution of indoleacetic acid oxidase and inhibitors in light grown cotton. Plant Physiol. 39:741-746. 21. Morgan, P. W. , and W. C. Hall. 1963. Indoleacetic acid oxidizing enzyme and inhibitors' from light grown cotton. Plant Physiol. 38:365-370. 22. Morgan, P. W. , H. E. Joham, and J. V. Amin. 1966. Effect of manganese toxicity on the indoleacetic acid oxidase system of cotton. Plant Physiol. 41:718-724. 23. Rabin, R. S. , and R. M. Klein. 1957. Chlorogenic acid as a competitive inhibitor of indoleacetic acid oxidase. Arch. Biochem; Biophys. 70:11-15. The identificaArchives 66 24. Ray, P. M. I960. The destruction of indoleacetic acid^III. Relationships between peroxidase action and indoleacetic acid oxidation. Arch. Biochem. Biophys. 87jl9-30. 25. Ray, P. M. , and K. V. Thimann. 1956. The distribution of indoleacetic acid. I. Action of an enzyme from Omphalia flavida. Arch. Biochem. Biophys. 64:175-'192. 26. Romney, E. M. , and S. J. Toth. 1954. Plant and soil studies with radioactive manganese. Soil Sci, 77:107-117. 27. Rosenblatt, M. , and J. V. Pleuso. 1941. Determination of tannins by photocolorimeter. Jour. Assoc. Off. Agr. Chem, 24:170-180. 28. Sabharwal, S. L. , R. Garren, Jr., and O. C. Compton. 1971. Growth and mineral composition response of strawberry plants to manganese nutrition. (In Prepaf-ation). 29. Sacher, J. A. 1963. Effects of inhibitors on the kinetics of indoleacetic acid oxidation. Amer. J. Bot. 50:116-122. 30. Schaeffer, G. W. , J. G. Buta, and F. Sharpe. 1967. Scopoletin and polyphenol-induced lag in peroxidase catalyzed oxidation of indole-3-acetic acid. Physiol. Plants 20:342-347. 31. Stonier, T. 1969. Studies on auxin protectors. VII. Association of auxin protectors with crown gall development in sunflower stems.. Plant Physiol. 44:1169-1174. 32. Stonier, T. , F. Rodriguez-Tormes, and Y. Yoneda. 1968. Studies on auxin protectors. IV. The effect of manganese on auxin protector of the Japanese morning glory. Plant Physiol. 43:69-72. 33. Stonier, T. , and Y. Yoneda. 1967. Auxin destruction and growth in Japanese morning glory stems. Ann. New York Acad. Sci. 144:129-135. 34. Straus, J. , and R. K. Gerding. 1963. Auxin oxidase and growth control in tissue cultures of Ephedra. Plant Physiol. 38:621-27. 35. Stutz, R. E. 195 7. The indoleacetic acid oxidase of Lupinus a lb us L. Plant Physiol. 32:31-39. 67 36. Tang, Y. W. , and J. Bonner. 1946. The enzymatic inactivation of indoleacetic acid I. Arch. Biochem. Biophys. 13:11-25. 37. Taylor, D. M. , P. W. Morgan, H. E. Joham, and J. V. Amin. 1968. Influence of substrate and tissue manganese on the IAAoxidase system in cotton. Plant Physiol. 43:243-247. 38. Tomaszewski, M. , and K. V. Thimann. 1966. Interactions of phenolic acids, metallic ions, and chelating agents on auxininduced growth. Plant Physiol. 41:1443-1454. 39. Wagenknecht, A. C. , and R. H. Burris. 1950. Indoleacetic acid inactivating enzyme from bean roots and pea seedlings. Arch. Biochem. 25:30-5 3. 40. Zenk, M. H, , and G. Muller. 1963. In vivo destruction of exogenously applied indoly.l-3-acetic acid as influenced by naturally occurring penolic acids. Nature 200:761-763.