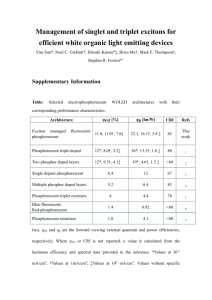
E L E C T R O P H O...
advertisement

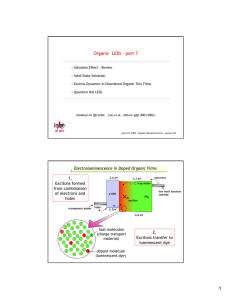
Organic LEDs – part 4 ELECTROPHOSPHORESCENCE 1. OLED efficiency 2. Spin 3. Energy transfer 4. Organic phosphors 5. Singlet/triplet ratios 6. Phosphor sensitized fluorescence 7. Endothermic triplet energy transfer Lecture by Prof. Marc Baldo April 10, 2003 – Organic Optoelectronics - Lecture 17 1 The unit of perceived optical power is defined as the lumen (lm). Power efficiency of LED’s is therefore measured in lumens/Watt. The maximum photopic response is: Φ ~ 20 lm (blue) Φ ~ 680 lm (green) Φ ~ 100 lm (red) Photopic response (lm/W) POWER EFFICIENCY The eye’s response is wavelength dependent. 800 600 400 200 0 400 500 600 700 Wavelength (nm) For an OLED of given color, we must maximize: - quantum efficiency (ηQ = photons out/electrons in) - electrical efficiency (Vλ/V = photon energy/operating voltage) Vλ L ηP = = ΦηQ VI V Power efficiency = photopic response of eye x quantum efficiency x electrical efficiency 2 Quantum efficiency is the measure of a luminescent dye’s performance. ηQ= the number of photons emitted per electron injected. ηQ = χηoutφ PL φPL is fundamental luminescence efficiency of organic material. ηout is output coupling fraction reduced by absorption losses and wave guiding within the device and its substrate. χ is fraction of luminescent molecular excitations (defined by spin selection rules) typically ~ 25% remaining energy is wasted OLED glass Imposes a fundamental limit to OLED efficiencies 3 EXCITON SPIN AND SYMMETRY Molecular excited states (or excitons) are typically mobile, with binding energy ~ 1eV, and spin S = 0,1 Molecular ground state (spatially symmetric under exchange of electrons) E Lowest unoccupied (LUMO) molecular orbital S=0, singlet Highest occupied (HOMO) molecular orbital 1st excited state (spatially anti-symmetric (triplet) or spatially symmetric (singlet)) E LUMO ψ ψ ψ HOMO ψ } S=1, triplet S=0, singlet 4 Fluorescence and Phosphorescence S1 Fluorescence S0 Triplet has lower Intersystem energy because it is spatially crossing (ISC) antisymmetric under T1 exchange of electrons. (e--e- repulsion Phosphorescence lower). Molecular ground state Fluorescence: Decay from singlet allowed by symmetry: fast (109 s-1) and often efficient. Phosphorescence: Decay from triplet disallowed by symmetry: slow ( > 1 s-1) and inefficient. 5 Efficient phosphorescence Need to mix singlet and triplet states: - make both singlet and triplet decay allowed. Use metal-organic complexes with heavy transition metals: Pt, Ir, etc... Ligand molecular orbital Spin orbit coupling mixes states: proportional to atomic number Z4 Type I phosphor Type II phosphor Exciton localized on organic ligand exciton +- Metal-ligand charge transfer exciton MLCT exciton N - N Pt N + N N Ir 3 metal ligand PtOEP less mixing ~ 100µs triplet lifetime metal Ir(ppy)3 ligand most mixing ~ 1µs triplet lifetime 6 Structure and operation of OLEDs - must transfer energy from host material to luminescent dopant. This determines the quantum efficiency of luminescence. A heterostructure OLED holes electrons Hole transport layer Electron transport layer Light Cathode Luminescent dopant molecules Transparent Anode Assume balanced currents: every hole combines with an electron Excitons form in host at interface. Ideally energy is transferred to luminescent molecules 7 Energy transfer (a) Förster energy transfer Long range (up to ~ 100Å) Acceptor (dye ) Donor* Acceptor Donor Acceptor* Donor Exciton non-radiatively transferred by dipole-dipole coupling if transitions are allowed (usually singlet-singlet). (b) Dexter energy transfer Donor Donor* Acceptor Donor Acceptor* Exciton hops from donor to acceptor. Acceptor (dye ) Short range ~ 10Å 8 A red phosphor: Emission spectrum Platinum octaethylporphyrin N Intensity (a.u.) 1 N Pt N • Triplet lifetime ~100ms • Peak external quantum efficiency in Alq3 ~ 4% N 0 PtOEP 400 500 600 700 800 900 Wavelength (nm) Luminance of 6% device (cd/m2) 2 Mg:Ag cathode PtOEP in Alq3 a-NPD Indium tin oxide 10 30 50 100 200 300 10 Quantum Efficiency (%) Device structure 4 1% PtOEP 6% PtOEP 20% PtOEP efficiency roll-off due to triplet-triplet annihilation triplet singlet triplet ground state 1 0.1 Loss proportional to square of triplet lifetime 0.1 1 10 100 Current Density (mA/cm2) 9 DOES PTOEP CAPTURE ALQ3 TRIPLETS? Put fluorescent dye DCM2 in exciton formation zone. 1.8 1.6 PtOEP Ag Ag Mg/Ag Mg/Ag Intensity (arb. units) 500Å Alq3 1.4 1.2 700Å Alq3 100Å PtOEP/Alq3 DCM2 100Å Alq3 100Å DCM2/Alq3 100Å DCM2/Alq3 1.0 350Å a-NPD 350Å a-NPD 0.8 60Å CuPc 60Å CuPc ITO ITO 0.6 Glass Glass 0.4 Device 1 Device 2 0.2 Alq3 0.0 500 600 700 800 Wavelength (nm) 10 The singlet/triplet ratio in Alq3 Device 1 emits from singlets only Device 2 emits from singlets and triplets Ag Ag Mg/Ag Mg/Ag 200Å Alq3 200Å Alq3 400Å DCM2/Alq3 400Å PtOEP/Alq3 450Å a-NPD 450Å a-NPD 60Å CuPc 60Å CuPc ITO ITO Glass Glass Compare ratio of EL emission from devices to get singlet fraction. After correction for PL efficiency of DCM2 and PtOEP, get: χ=(22±3)% 11 IMPROVED PHOSPHORS Must reduce triplet lifetime to minimize T-T annihilation. Increase MLCT component of excited state. 30 20 105 104 10 103 102 4 10 2 1 1 0.1 0 2 4 6 8 Voltage (V) 10 Power efficiency (lm/W) Luminance (cd/m2) The first high efficiency green phosphor was iridium tris(2-phenylpyridine) Ir(ppy)3 MLCT exciton + N Ir 3 metal ligand Broad green emission at λ ~ 515 nm. Phosphorescent lifetime, τ ~ 1 ms. 12 TRIPLET-SINGLET ENERGY There areTRANSFER many more fluorescent than phosphorescent materials. Can we get high efficiency from a fluorescent dye? host fluorescent dye exciton formation S ? exciton formation S T T Need to prevent exciting triplet state in fluorescent dye. Want mechanism for triplet-singlet energy transfer. 13 Phosphor sensitized fluorescence 1. Triplet-singlet hopping transfer is disallowed 2. Triplet-singlet Förster transfer permitted if triplet relaxation on donor is allowed i.e. triplet-singlet transfer is possible from a phosphorescent donor Predicted by Förster in 1959 (†) Observed by Ermolaev and Sveshnikova in 1963 (§) e.g. for triphenylamine as the donor and chrysoidine as the acceptor, in rigid media at 77K or 90K the interaction length is 52Å (†) Förster, Th. Dicussions of the Faraday Society 27, 7-17 (1959). (§) Ermolaev, V.L. & Sveshnikova, E.B. Doklady Akademii Nauk SSSR 149, 1295-1298 (1963). 14 IMPLEMENTATION OF TRIPLET-SINGLET ENERGY TRANSFER host phosphorescent sensitizer S fluorescent dye S S ISC T T T N N N N DCM2 absorbs in the green Ir O CH3 CBP 3 Ir(ppy)3 NC CN 15 10 8 7 6 5 0.2% DCM2 and 8% Ir(ppy)3 in CBP 4 3 Note: factor of 4 difference 2 1 0.9 0.8 0.7 0.6 0.5 0.01 0.2% DCM2 in CBP 0.1 1 10 100 1000 Current density (mA/cm2) Roll-off in efficiency is due to charge trapping on DCM2 molecules EL Intensity (arbitrary units) External quantum efficiency (%) DCM2 fluorescence sensitized by Ir(ppy)3 1.0 0.9 0.8 0.7 0.6 3 0.5 0.4 0.3 0.2 0.1 0.0 400 450 500 550 600 650 700 750 800 DCM2 Ir(ppy) Wavelength (nm) Nearly complete energy transfer from Ir(ppy)3 to DCM2 16 OLED TRANSIENT RESPONSE Intensity (arbitrary units) 100 DCM2 Examine transient response of different spectral components of OLED luminescence. Natural lifetime of DCM2 only ~5ns. 10 Ir(ppy)3 1 0 100 200 300 400 500 600 700 800 900 1000 Time (ns) Delayed DCM2 fluorescence confirms sensitizing action of Ir(ppy)3 17 BLUE Ir F N O F O 2 FIrpic (blue phosphor) (2.56±0.10) eV CBP kH 0.6 0.4 0.2 (2.62±0.10) eV FIrpic kR 6% FIrpic in CBP FIrpic in CHCl3 FIrpic at 460nm 0.8 0.0 Peak wavelength 470nm Triplet lifetime 10 µs kF 1.0 450 500 550 Wavelength (nm) CIE y coordinate N Inentsity (arb. units) Problem: the most energetic charge transport hosts currently available have blue-green triplets. So to transfer energy to a blue phosphor, exciton transfer must be endothermic. 0.4 0.3 0.2 0.1 0.0 6% FIrpic in CBP FIrpic in CHCl3 FIrpic at 460nm 0.0 0.1 0.2 0.3 CIE x coordinate Possible to get efficient endothermic transfer if decay of host triplet disallowed. CBP kG N N Host CBP triplet lifetime ~ 1s. 18 Transient response of endothermic transfer 104 103 50 100 150 200 250 300 Temperature (K) 102 (b) (c) (a) (d) (a)T=300K (b)T=200K (c)T=100K (d)T= 50K 0 10 20 Ir(ppy)3 in TPD 1000 0.5 0.0 PL Intensity (arb. units) EL Intensity (arb. units) PL intensity (arbitrary units) PL intensity (arbitrary units) Firpic in CBP 1.0 30 40 50 Time (µs) Efficiency of luminescence decreases below T=200K as energy transfer to phosphor is frozen out. Transient response is also consistent with endothermic transfer: At T=200K, apparent transient lifetime of FIrpic (includes endothermic transfer) >> natural lifetime (~10µs) 100 τ = (15 ±2) µs 10 6% Ir(ppy)3:TPD 1 0 10 20 Time (µs) 1000 T=292K τ = (80±10)µs 100 T=200K 10 1 τ = (15 ±2) µs 6% Ir(ppy)3:TPD 0 20 40 60 80 100 Time (µs) 19 CIE y coordinate COLOR PURITY 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0.0 ppy2Ir(acac) btp2Ir(acac) PtOEP FIrpic 0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 CIE x coordinate Organic materials have broad luminescent spectra. Overcome in red and blue by shifting toward non-visible wavelengths. 20 OLED SUMMARY Peak power efficiency in green is 60 lm/W Peak quantum efficiency is 19% (corresponds to ~100% internal) Largest remaining problem is operating voltage J = 1 µA/cm2 phosphor host Φ (lm/W) ηP (lm/W) ηQ ext ηQ int Vλ V J = 1 mA/cm2 ηP (lm/W) ηQ ext ηQ int Vλ V ppy2Ir(acac) TAZ 530 60 0.19 0.87 0.60 20 0.15 0.68 0.25 btpIr(acac) CBP 170 4 0.07 0.32 0.34 2.2 0.06 0.27 0.22 FIrpic CBP 260 1.3 0.006 0.027 0.83 5.0 0.057 0.23 0.34 PtOEP CBP 60 0.3 0.056 0.23 0.09 0.2 0.042 0.19 0.08 Table of phosphorescent device operating parameters 21 1.0 0.8 0.6 0.4 0.2 0.0 600 650 700 Intensity (arb. units) Intensity (arb. units) Intensity (arb. units) PHOSPHORESCENT DEVICE PERFORMANCE RED GREEN BLUE 1.0 0.8 0.6 0.4 0.2 0.0 Wavelength (nm) 500 550 600 1.0 0.8 0.6 0.4 0.2 0.0 Wavelength (nm) 450 500 550 Wavelength (nm) CH3 O CH3 Ir O Ir N O 2 O F 1 btp2Ir(acac) in CBP 101 Current density 102 0.1 103 (mA/cm2) FIrpic ppy2Ir(acac) 100 100 10 10 1 1 10-3 10-2 10-1 100 101 102 103 104 Current density (mA/cm2) 10 10 1 1 0.1 10-3 FIrpic in CBP 10-2 10-1 100 101 102 Power efficiency (lm/W) 1 Quantum efficiency (%) 10 Power efficiency (lm/W) Quantum efficiency (%) 10 100 O 2 btp2Ir(acac) 10-1 N CH3 CH3 2 0.1 10-2 F Quantum efficiency (%) N Ir O Power efficiency (lm/W) S N 0.1 Current density (mA/cm222 ) PHOSPHORESCENT STABILITY 1.2 1.0 50 cd/m2 0.8 100 cd/m2 0.6 0.4 0.2 0.0 500 cd/m2 1 10 100 200 cd/m2 1000 10000 Time (hours) Red: BtpIr(acac) in CBP Normalized optical power Normalized optical power Courtesy Ray Kwong and UDC 1.0 200 cd/m2 0.8 500 cd/m2 1000 cd/m2 0.6 0.4 2000 cd/m2 0.2 0.0 1 10 100 1000 10000 Time (hours) Green: (ppy)2Ir(acac) in CBP Phosphors should improve OLED stability by rapidly removing triplet excitons and lowering drive current required. 23