X-rays Henri G.J. Moseley 1887 – 1915
advertisement

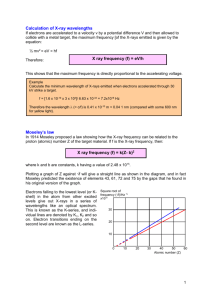
X-rays Henri G.J. Moseley 1887 – 1915 X-Ray Production • While an electron is in the vicinity of a nucleus, it feels its electromagnetic field • Acceleration (v2/R) = Electromagnetic Radiation • Bremsstrahlung (“Braking radiation”) effect Emission of Photons (Xrays: λ=0.01 to 1nm) • Not a continuous process, since a photon can only be created with an energy E = hν X-rays • When Bohr’s model appears (1913), people know about X-rays (Roentgen, 1895): – Bombardment of e- into materials: • Bremstrahlung (Continuum) • Peaks (which are different for each element) Shells • Historically: – States were given letters: • • • • • n=1 n=2 n=3 n=4 etc… K shell, L shell, M shell, N shell, – Transitions ∆n between shells: • ∆n=1 • ∆n=2 • etc… α, β, Many electrons atom • Bohr’s model works for Hydrogen • What about the other elements ? – Z: Atomic number Number of electrons • • • • Z=1 Hydrogen Z=2 Helium Z=6 Carbon Z=92 Uranium … -1.51 Z2 eV -3.40 Z2 eV – Approximation: we ignore the mutual repulsion of the electrons. • The coulomb force between one electron and the nucleus is: F = (Ze2)/(4πε0r2) • Applying the same treatment than before: En = Z2 E1/n2 10.2 Z2 eV -13.6 Z2 eV Characteristic X-ray Wavelengths • 1913: H.G.J.Moseley 1887-1915 – Catalogs the X-ray emission (K) of various elements – Finds an empirical formula: – Volunteered to fight in World war I, killed in action in 1915 (he was 27 years old) 2 (Z-1) ? Why -1 ? • Transitions measured (K) correspond to the transition of the electrons deep in the atom. – Does not depend on Z Always the closest – So why (Z-1) ? – At the time, nobody knows, but there can be 2 electrons in the inner orbit Charge screening effect Moseley Plot • X-ray energy depends on Z, so one can determine Z by measuring the energy of the X-rays • Some elements were missing ! – Z=43 – Z=61 – Z=75 Technecium Promethium (1940) ! Rhenium • Element composition: X-ray fluorescence – Non-destructive