Neisseria meningitidis Andrew Schofield, Jamie Wood and James Moir
advertisement

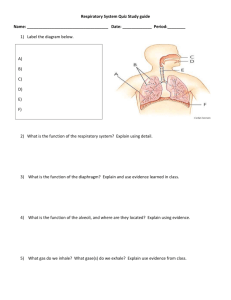
Respiratory pathway choice in Neisseria meningitidis Andrew Schofield, Jamie Wood and James Moir Overview Introduction: Neisseria and respiration Organisation of respiratory chain components in N. meningitidis Regulation: (i) Genetic control, (ii) Metabolic control Biology of a microbial respiratory system Early stages of mathematical model building: perspectives and approaches Neisseria meningitidis natural habitat – the human nasopharynx (5-20 % of population colonised) Occasionally, invasive disease occurs … Multiplication in blood septicaemia Multiplication in cerebrospinal fluid meningitis meninges Neisseria meningitidis 0.01 C. violaceum N. weaveri N. m eningitidisand N. gonorrhoeae459 N. flavescens are more closely related to one 410 another than other, non-pathogenic N. subflava Neisseria. Phylogenetic 998 N. meningitidis tree based on 16S rRNA N. gonorrhoeae 810 sequenc e. N. lactamica 610 N. pharyngis 1000 N. mucosa 494 N. flava 956 N. sicca N. elongata N. bacilliformans 985 762 Only these two species are pathogenic, the others are strictly commensal. (Why?) Colonisation of human nasopharynx necessary precursor to virulence Survival in natural habitat and blood/CSF relies on virulence determinants such as adhesion / immune evasion etc. but also on respiratory metabolism Respiration Oxidation of cellular reductants to generate biochemical energy reduction potential + ½O2 +830 mV H 2O electrochemical (H+) gradient across biological membrane 2e- -340 mV - NADH NAD+ drives synthesis of ATP In the mitochondrion Respiratory electron transport NADH Q cyt. bc1 cyt. c aa3 O2 succinate H+ H+ H+ H+ Q ½O2 NADH NAD+ H2O ADP ATP In bacterial plasma membrane Respiratory electron transport cbb3 NADH succinate hydrogen formate Q cyt. bc1 nitrate reductase ba3 O2 methanol methylamine sulfur dehydrogenase cyt. c aa3 O2 O2 nitrite reductase nitric oxide reductase nitrous oxide reductase *selected respiratory pathways of Paracoccus denitrificans, a close relative of the mitochondrion In bacterial plasma membrane Respiratory electron transport predicted for N. meningitidis cbb3 NADH Q cyt. bc1 O2 cyt. c succinate Cytochrome cbb3 oxidase, identified in: Microaerophilic pathogens: Helicobacter, Campylobacter Necessary for maintaining oxygen sensitive nitrogenase in root nodule bacteroids of rhizobia Expressed under microaerobic conditions in versatile bacteria such as Paracoccus, Pseudomonas Bradyrhizobium japonicum cbb3 has high affinity (Km = 7 nM) for O2 Oxygen and Neisseria environment air-flow O2 colonising the nasopharyngeal mucosa Common bacterial cohabitants with Neisseria species: Streptococcus, Staphylococcus Veillonella, Prevotella, Fusobacterium (“strict” anaerobes) Respiratory pathways in N. meningitidis Predictions from genomes of Neisseria meningitidis O2 NO3- cbb3 NO2- H2O AniA NO microaerophilic oxidase NorB N2O N2 partial denitrification …function of denitrification genes verified genetically: Denitrification of nitrite supports growth under microaerobic conditions and requires AniA and NorB Respiratory substrates in the environment of N. meningitidis NO3- / NO2- saliva macrophages NO air-flow O2 colonising the nasopharyngeal mucosa Meningococcus exposed to variety of potential respiratory electron acceptors Must deal with NO generated in the host by NO synthase Questions How is the respiratory pathway of N. meningitidis organised? How does the bacterium regulate respiratory flux? Is mathematical modelling necessary to appreciate a system that consists of just three respiratory reactions? Does analysis of respiration in N. meningitidis tell us anything about its nature as a pathogen? Can analysis of respiration in Neisseria tell us anything more general about the nature of respiratory chain structure and function in other bacteria? Part 1 Organisation of Neisseria respiratory chains: architecture and function of respiratory chain components Focus on: electron transport pathways and topological arrangements Respiratory pathways in N. meningitidis Predictions from genomes of Neisseria meningitidis cbb3 NADH Q cyt. bc1 succinate H2O cyt. c NO O2 AniA NorB N2O *confirmation of bc1 complex dependent pathway to oxygen and nitrite using inhibitor sensitivity with myxothiazol NO2NO Respiratory pathways in N. meningitidis Predictions from genomes of Neisseria meningitidis outer membrane 4H+ periplasm 4H+ 2H+ inner NorB membrane ? oxidase NADH dehydrogenase 2NO +2H+ NAD+ NADH N2O -0.2 NADH/NAD+ 2H+ 4H+ Thermodynamic considerations: -0.4 0 0.2 0.4 NO2-/NO 0.6 2H+ bc1 Q Q AniA NO2+2H+ NO 0.8 ½O2/H2O potential (V) 1.0 1.2 NO/N2O 2H+ ½O 2 +2H+ H2O NADH/O2 NADH/NO2NADH/NO H+:e5 3 3 ? Potential pathways of electrons to O2 and nitrite OM (* like cytochrome-NIR fusion in Bdellovibrio) $ * Periplasm $ like extra domain in NG cbb3 oxidase IM ? Cytochrome cx NMB0717 monohaem, Cytochrome c4 NMB1805 di-haem, predicted periplasmic Cytochrome c5 NMB1677 di-haem, predicted predicted periplasmic membrane-associated similar to NGO0292 similar to NGO0101 (near (different C termini) identical) identical to NGO1328 To cut a long story short… Organisation of respiratory chain in N. meningitidis outer membrane NO2- periplasm NO inner membrane Q NADH Q NAD+ 2NO N2O H2O ½ O2 electron flow between membranes in N. meningitidis outer membrane ~50 Å AniA ~50 Å e~60 Å e- cyt. c 5 e- inner membrane N. meningitidis versus all the other Neisseria sp. Same respiratory components except… …different domain structure of cbb3 oxidase N. meningitidis N. gonorrhoeae, N. lactamica, etc. CcoP CcoP CcoO CcoO H2O H2O ½ O2 CcoN ½ O2 CcoN >99% sequence conservation between conserved domains, but loss of domain between other Neisseria and evolution of N. meningitidis –strong selection pressure –for what? Nitrite reductase activity –dependency on c5 Strain Nitrite reductase activity N. meningitidis wild type + + + N. meningitidis c5- N. gonorrhoeae wild type N. gonorrhoeae c5- Why are multiple pathways necessary for oxygen / nitrite reduction? Experimental approaches are vital, but can modelling provide additional insight? Once we have predicted qualitative behaviour we should be able to test this with whole cell UV/visible spectroscopy … Redox difference spectra of N. meningitidis intact cells 0.16 absorbance 0.12 c-type cytochrome 0.08 b-type cytochrome 0.04 0 350 -0.04 400 450 500 550 600 650 700 Wavelength (nm) -0.08 Reduced cells oxidised by oxygen Reduced cells oxidised by nitrite Reduced cells oxidised by nitric oxide Part 2 Regulation: Genetic and metabolic Respiratory pathways in N. meningitidis O2 cbb3 NO2- H2O AniA NO microaerophilic oxidase NorB N2O partial denitrification this doesn’t look too complicated... The “switch” from aerobic to anaerobic is a complex one 1000 60 NO 800 50 O2 40 30 600 400 [NO] (nM) [O2] (% air saturation) 70 20 200 10 0 0 0 0.5 50 1 1.5 2 2.5 3 3.5 4 4.5 100 150 200 250 300 350 Time 400 (h) 450 1. Oxygen becomes depleted 0 NO2- AniA NO NorB N2O 2. Nitrite reductase induced, NO accumulates 3. NO inhibits oxidase, O2 rises O2 cbb3 H2O 4. NO induces NO reductase, NO falls 5. Oxygen falls, steady state respiration under oxygen limitation ensues The “switch” from aerobic to anaerobic is a complex one transition from aerobic to denitrifying growth: high failure rate in N. meningitidis in batch growth 1.6 1.4 1.4 1.2 1.2 1.6 1.4 1.4 1.4 1.4 1.2 1.2 1.2 1 1 1 0.8 0.8 0.8 0.6 0.6 0.6 0.4 0.4 0.4 0.2 0.2 0.2 0 0 1.2 1 1 1 0.8 0.8 0.8 0.6 0.6 0.6 0.4 0.4 0.4 0.2 0.2 0 0 0.2 0 2 4 6 8 time(h) 10 0 2 4 8 time(h) 6 10 0 0 2 4 6 8 10 time(h) 0 2 4 6 8 time(h) 10 0 0 2 4 6 8 time(h) 10 0 2 4 6 8 10 time(h) [nitrite] cell density N. meningitidis is under stress during the transition and does not always achieve transition to denitrification readily This effect can be independent of culture history / genotype (different behaviour of duplicates) 1.6 1.8 1.4 1.6 1.4 1.2 1.2 1 1 0.8 0.8 0.6 0.6 0.4 0.4 0.2 0.2 0 0 0 Unpredictability disappears in absence of active nitrite reductase pathway 2 4 6 8 time(h) 10 0 2 4 In aniA- strains 6 8 time(h) 10 Metabolic control of respiration NO2- AniA NO - NorB - Thermodynamic considerations: - -0.4 - O2 N2O Oxygen, nitrite and nitric oxide compete for the same pool of electrons cbb3 -0.2 NADH/NAD+ 0 0.2 0.4 NO2-/NO 0.6 0.8 ½O2/H2O potential (V) 1.0 1.2 NO/N2O H2O NO is a competitive inhibitor of the oxidase (O2 and NO have similar molecular structures) and thus denitrification intermediate accumulation can inhibit oxidase activity. Genetic control of respiration Synthesis of denitrification reductases is governed by availability of respiratory substrates oxygen, nitrite and nitric oxide O2 inhibits aniA expression NO2- activates aniA expression NO activates norB expression NO2- AniA NO NorB + + - O2 cbb3 H2O N2O Genetic control of respiration Synthesis of denitrification reductases is governed by availability of respiratory substrates via transcriptional regulators FNR, NarQP and NsrR 384 base promoter region nitric oxide reductase norB - NarP NsrR - + - nitrite reductase aniA NsrR FNR - + NO2 - NO + O2 NO NO Effect of NO on mRNA measured using RT-PCR 12 14 norB 12 aniA 10 10 8 1.4 FNR +ve control 1.2 1.0 8 6 6 10 8 6 0.6 4 2 2 aniA in nsrR- 12 0.8 4 4 14 2 0.4 0 10 20 30 40 [NO] 50 0 10 20 30 40 50 [NO] 0 10 20 30 40 50 [NO] 0 10 20 30 40 50 [NO] Genetic control of respiration Synthesis of denitrification reductases is governed by availability of respiratory substrates via transcriptional regulators FNR, NarQP and NsrR 384 base promoter region nitric oxide reductase norB - NsrR NO NarP + + NO2 - - + nitrite reductase aniA NsrR FNR NO O2 NO Positive and negative regulation of aniA by NO should ensure aniA synthesis can be initiated before NO accumulation, can be optimal during denitrification, and can be switched off if NO approaches toxic concentration. Complex regulation of a system with only three reactions: respiration in N. meningitidis NsrR NsrR NarP + NO2- + + AniA NO NorB N2O - FNR- FNR - O2 cbb3 - H2O And... [Disproportionation of nitrite to NO under acidic conditions] [Reaction of NO with oxygen] [Irreversible inhibition of oxidase by peroxynitrite] keeping it “simple” by ignoring genetic regulation using this… …to attempt to explain this d [O2 ] [O2 ] k1[O2 ][Ca ] (1 sat ) dt K O2 d [ NO] l1[ NO][ Ba ] m1[ NO2 ][ Aa ] [ NO] dt d [ NO2 ] m1[ NO2 ][ Aa ] dt d [Qa ] g[Qi ] f [Qa ][ L] l3[Qa ][ Bi ] dt i k3 K NO [ E ][Ci ] d[E] f [Qa ][ L] m3[ E ][ Ai ] i dt [ NO] K NO E L X, Qi Qa Q d [ Aa ] m3 [ E ][ Ai ] m1[ NO2 ][ Aa ] dt d [ Ba ] l3 [ E ][ Bi ] l1[ NO][ Ba ] dt i d [Ca ] k3 K NO [ E ][Ci ] k1[O2 ][Ca ] i dt [ NO] K NO Ai Aa A, Bi Ba B, Ci Ca C NO2O2 NO NO2NO e- - O2 H2O - Transition from oxygen to denitrification appears unstable under laboratory conditions. What about in vivo? Some patients recovering from meningococcal disease do make antibodies against AniA But, some clinical isolates show pseudogenization of aniA* i.e. Nitrite reduction can be a useful strategy for N. meningitidis in vivo, but it can also be selected against, presumably because the short term adaptation has a high failure rate [*norB always conserved and plays a role in protecting bacterium from host immune system in tissue/organ culture models] aniA is *very highly conserved* in N. gonorrhoeae and other commensal Neisseria and aerobic/anaerobic switch consistently proceeds predictably in N. gonorrhoeae -why the difference? Transition from oxygen to denitrification appears unstable under laboratory conditions What factors affect this transition? N. meningitidis N. gonorrhoeae, N. lactamica, etc. CcoP cbb3 Structure of respiratory chain (components)? -differences between N. meningitidis and the rest of Neisseria Transcriptional regulatory network? -differences between N. gonorrhoeae and the rest of Neisseria Population structure / density? CcoP cbb3 Regulation and gonococcus versus meningococcus Same components, same result, different mechanism FNR binding to N. gonorrhoeae aniA promoter weak c.f. that of N. meningitidis Compensated for by cis effect on narP binding site AND trans effect on nitrite sensitivity of NarQ N. gonorrhoeae N. meningitidis NO2- NO O2 NO2- O2 NO2- NO O2 NsrR FNR NarQ NarQ NsrR FNR + NarP NarP PaniA PaniA PaniA Summary Respiratory pathway choice in N. meningitidis is controlled in a complex manner metabolically and genetically An ODE model for respiratory pathway choice is providing good qualitative fits to explain metabolic regulation but requires further parameterisation This approach should be able to inform us regarding the key control features in this system, and help us appreciate issues relating to different adaptations between closely related Neisseria species Neisseria uses multiple respiratory electron carriers to shuttle electrons from bc1 complex to terminal electron acceptor reductases Adapting TASEP for application to branched respiratory chains may provide insight regarding organisational principles with relevance to other bacteria /synthetic biology Acknowledgements University of York Jamie Wood Andrew Schofield Elizabeth Thomas Yujiang Wang Manu Deeudom Melanie Thomson Karin Heurlier Jonathan Rock Diana Quinn University of Oslo Mike Koomey Ashild Vik University of Sheffield Robert Read University of Leeds Chris Needham