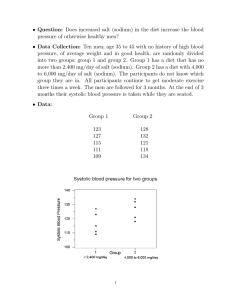
Ammonium oxalate treatment application in the presence of solu-
advertisement

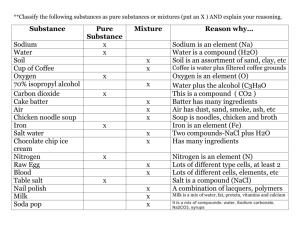
Built Heritage 2013 Monitoring Conservation Management Ammonium oxalate treatment application in the presence of soluble salts: laboratory results on soft limestone Tabitha Dreyfuss; Joann Cassar Department of the Built Heritage, Faculty for the Built Environment, University of Malta, Msida, Malta 1. Aims of study The aims of this research were multiple: to investigate the different possible outcomes when ammonium oxalate treatment is applied to Globigerina Limestone that contains different types of soluble salts; to understand the role played by the stone’s pathology vis-à-vis the treatment; and to assess the effect of treatment contact time so as to consider the impact that this may have on the resulting formation of calcium oxalate. This paper focuses on that part of the research concerning the resistance to salt crystallization of weathered limestone samples treated with ammonium oxalate. 1.1. Introduction The Maltese Islands, which are located in the centre of the Mediterranean Sea, consist of a small archipelago measuring 316 sq. km. The Islands have numerous historic limestone buildings and monuments that span the millennia. These are mostly built in Maltese Globigerina Limestone - a highly porous (total porosity up to 40%) calcareous stone which deteriorates, often catastrophically, in an environment that is exposed to both high moisture levels and elevated amounts of soluble salts. Globigerina Limestone exists as one of two types - the more durable franka and the less durable soll [Cassar, 2002]. Many of the historic stone edifices in Malta and Gozo were built before the insertion of a damp proof course became mandatory, thus allowing water entry in the form of rising damp together with any soluble salts present . Additionally, traditional wall construction generally utilised soil infill, usually salt laden, between two masonry wall leaves. The Island environment further enhances salt contamination through wind driven and aerosol borne salts. The context is therefore a porous limestone which has a continual supply of moisture and soluble salts. Treatment of exposed Globigerina Limestone which has lost cohesion, manifested as powdering/ granular disintegration must therefore take this context into account. 1.2. Ammonium oxalate treatment Ammonium oxalate treatment of calcareous stone has, over the past 30 years, emerged as a conservation treatment with both consolidating and protective properties [Matteini and Moles, 1986; Matteini et al., 1994; Cezar, 1998; Miliani et al., 2007; Matteini, 2007; Bracci et al., 2008; Charola et al., 2010; Conti et al., 2011; Booth et al., 2012]. The surface conversion of calcium carbonate to calcium oxalate is ideal for calcareous stone. Results from studies on Globigerina Limestone have confirmed that the resulting calcium oxalate is harder and more resistant to acid attack, with a lower solubility than calcium carbonate, and with an improvement in surface cohesion occurring after treatment [Mifsud, 2006; Mifsud and Cassar, 2006; Dreyfuss and Cassar, 2012]. Althou1271 Built Heritage 2013 Monitoring Conservation Management gh the treated stone undergoes some reduction in surface porosity (detailed studies in this respect are ongoing), its wetting and hydrophilic properties are retained [Croveri, 2003; Croveri et al., 2004]. Furthermore, treatment in the presence of high levels of sodium chloride does not impede the conversion to calcium oxalate [Mifsud and Cassar, 2006; Pinna et al., 2011]. This is an important factor, especially in a marine environment like the Maltese Islands where marine aerosols are ever present. Other salts that are commonly present in historical masonry include both sulphates and nitrates. This study has centred on Globigerina Limestone of the franka type and includes both these salt types - as sodium sulphate and sodium nitrate - as well as sodium chloride, in a comparative study where ammonium oxalate treatment was applied to samples of this soft limestone under laboratory conditions. 2. Sample selection and preparation In an attempt to recreate site conditions in a controlled laboratory environment, the range of franka samples tested included artificially weathered types as desalinated samples as well as salt contaminated samples. In addition, stone that was treated with different contact times was also included. The selection of samples included in this part of the research programme is summarised in Table 1 below. The sample types outlined in this table were prepared as 50mm x 50mm x 50mm cubes as described in Sections 2.1 to 2.4 below. 2.1. Sample preparation Quarry franka samples in the form of stone blocks (approximately 410mm W x 230mm D x 267mm H) were obtained from the main quarry area of Qrendi from the area known as Ta’ l-Iklin (quarry coordinates: 51500, 66500) at a depth of 12m below ground level. Cubes measuring 50mm x 50mm x 50mm were dry cut from the stone blocks. The samples were then dusted/ brushed with a dry nylon brush to remove superficial surface dust resulting from the Table 1 Range of samples included in the study. Treatment is with a saturated (5%) ammonium oxalate (monohydrate) solution applied as a poultice 1272 Built Heritage 2013 Monitoring Conservation Management cutting procedure. The weathered pathology of the franka samples was then induced through artificial weathering through 2 cycles of salt weathering as per EN12370:2000 using anhydrous sodium sulphate. This resulted in a powdering surface texture with an increased surface area, visible to the naked eye. 2.2. Desalination The desalinating procedure was first carried out on all the samples. These were immersed in distilled water, repeatedly changing the water until its conductivity revealed that soluble salts were no longer present. The conductivity of the distilled water used (≤3μS) was measured by means of a conductivity meter (HI98308 PWT – Hanna instruments). The conductivity of the distilled water containing the immersed samples was measured after each immersion until its conductivity was ≤3μS. All of the samples- including those that were to be salt contaminated - were thus desalinated and then oven dried for 24 hours at a temperature of 105ºC, then cooled in the laboratory to constant mass at 20 ºC room temperature. One fourth of the samples were then retained as is, to represent the desalinated type samples, while the remaining samples were selectively salinated as described in Section 2.3 below. 2.3. Selective salt contamination (chlorides, sulphates, nitrates) Following the desalination procedure, those samples that were designated for salt contamination as per Table 1 were selectively contaminated. The chloride group were immersed in a saturated solution of sodium chloride, the sulphate group in a saturated solution of sodium sulphate and the nitrate group in a saturated solution of sodium nitrate – all for a duration of 2 hours. Following immersion, the samples were air dried to constant weight. 2.4. Ammonium oxalate treatment A saturated (at 5%) ammonium oxalate (monohydrate) treatment was applied in a cellulose pulp poultice for 2 different contact times – 5 hours and 24 hours. Calcium oxalate had already been found to form on franka limestone after a 5 hour contact time of an ammonium oxalate paper pulp poultice (Mifsud 2006). The 24 hour contact time is however considered to be more practical in the field since this allows treatment to be applied up until the end of the working day rather than limiting treatment to take place 5 hours before the end of the working day. For this reason samples with a 24 contact time of treatment were included in this study. Following treatment, the poultice was manually removed and the samples left to air dry at room temperature. The excess pulp was brushed off with a soft and dry nylon brush. Only the top surface of the cube samples was treated. For every treated sample type, an untreated control sample was included. 3. Testing The determination of resistance to salt crystallisation was carried out in accordance with EN 12370:2000 on the treated and untreated samples, to determine whether or not the ammonium oxalate treatment improved the stones’ ability to resist this type of deterioration. The number of cycles carried out was 1273 Built Heritage 2013 Monitoring Conservation Management 4 and the resistance to salt crystallisation was calculated as the percentage mass of material lost against the initial dry mass. The results obtained are given in Figure 1. 4. Results and Discussion Fig.1 - % mass lost after 4 cycles of the sodium sulphate weathering test. (WF = weathered franka; DES = desalinated; CL = contaminated with sodium chloride; SUL = contaminated with sodium sulphate; NIT = contaminated with sodium nitrate; UN = untreated; T1 = treated for 5 hours; T2 = treated for 24 hours) For all sample types – desalinated, contaminated with sodium chloride, contaminated with sodium sulphate and contaminated with sodium nitrate – all of the untreated samples were found to have lost a greater percentage of their initial mass when compared to treated samples of the same type. Although the ammonium oxalate treatment was only applied to the top face of the cube samples, the difference between treated and untreated samples was significant for all cases. Untreated samples in the desalinated group, chloride group, sulphate group and nitrate group lost 47%, 62%, 56% and 59% respectively while treated (5hours) samples lost 22%, 39%, 23% and 35% respectively and treated (24 hours) samples lost 22%, 37%, 25% and 33% respectively. This suggests that ammonium oxalate treatment does in fact provide protection from salt crystallization even when this is formed in the presence of sodium chloride, sodium sulphate or sodium nitrate. This is also evidenced visually - Figure 2 - where the top untreated row is significantly more deteriorated, less sound and with less pronounced corners and edges than the bottom two rows (which were treated). The differences in resistance to salt crystallization between those samples treated for a 5 hour contact time and those treated for a 24 hour contact time were less significant, indicating that the increased contact time is probably neither crucial for nor detrimental to salt crystallization protection. Given the increased salt crystallization resistance in the weathered franka samples following ammonium oxalate treatment, the treated and untreated sample types were studied through water absorption tests, to verify whether the treatment had affected the water transport properties. Samples shown in Table 1 were prepared as per Sections 2.1 to 2.4 above and then tested. The 1274 Built Heritage 2013 Monitoring Conservation Management Fig.2 - photographs of the samples after the 4th cycle of the salt crystallization test samples were evaluated by means of the water absorption test through capillarity in accordance with EN15801:2009. The samples were placed over a 10cm bed of paper pulp, the bottom 8cm of which was saturated with distilled water. The treated samples were placed face down with the treated face in contact with the paper pulp. The reason for using paper pulp as opposed to filter paper as specified in the EN15801:2009 was due to the uneven surface of the naturally weathered stone samples which were seen not to have had full contact when placed on a wad of filter paper. The quantity of water absorbed per unit surface area during a given amount of time was measured. The results are expressed graphically in Figure 3, where the mass of water absorbed per unit surface area (y-axis) is plotted against the square root of time taken (x-axis). The graphs obtained show that, for all cases, they are similar in shape. This confirms that although ammonium oxalate treatment improves the salt crystallization resistance of weathered franka, it does not inhibit the mode or amount of water uptake through capillarity through the treated face. Fig.3 - water absorption graphs for weathered franka samples (WF = weathered franka; DES = desalinated; CL = contaminated with sodium chloride; SU = contaminated with sodium sulphate; 1275 NIT = contaminated with sodium nitrate; UN = untreated; T1 = treated for 5 hours; T2 = treated for 24 hours) Built Heritage 2013 Monitoring Conservation Management 5. Conclusion This stage in the understanding of ammonium oxalate treatment of Globigerina Limestone under different soluble salt content conditions is an important step towards carrying this treatment forwards to real site conditions, where soluble salts are naturally present in the stone and where desalination may prove to be difficult or impossible. The results obtained have shown that even in the presence of sodium chloride, sodium sulphate or sodium nitrate, a degree of protection from salt crystallization is achieved with ammonium oxalate treatment. This protection is achieved both for treatment applied for a 5 hour contact time as well as for treatment applied for a 24 hour contact time. Additionally, the pathology of powdering weathered franka is conducive to the successful consolidation of the stone as seen through the increased resistance to salt crystallization of weathered types. Moreover, this increased salt crystallization resistance does not alter the water transport properties of the stone. This is an important aspect to retain when considering historical limestone buildings and monuments that are exposed to moisture and soluble salts. The progression from laboratory to site is an important step to be considered in the development of this treatment for the conservation of Globigerina Limestone and it is hoped that through this research programme, a reliable conservation procedure may emerge that will assist within the overall conservation of Globigerina Limestone in real restoration projects. References Booth J., Viles, H., Fletcher P., 2012, An experimental assessment of 3 inorganic consolidants for use on museum artefacts in comparison to organosilanes, in 12th International Congress on the Deterioration and Conservation of Stone, New York. Bracci S., Sacchi B., Ferreira Pinto A. P., Delgado Rodrigues J., 2008, Inorganic consolidants on stone artefacts: optimisation of application procedures for marble and limestones, in Proceedings of the International Symposium - Stone consolidation in cultural heritage; research and practice, Lisbon, Portugal, pgs 81-90. Cassar J., 2002, Deterioration of the Globigerina Limestone of the Maltese Islands, in Siegesmund S., Weiss T., and Vollbrecht A. (eds)., Natural Stone, Weathering Phenomena, «Conservation Strategies and Case Studies», 205: 33-49, London: Geological Society London. Cassar J., Vella A. J., 2003, Methodology to identify badly weathering limestone using geochemistry, «Quarterly Journal of Engineering Geology and Hydrogeology», 36(1): 85-96, London, Geological Society London. Cezar T. M., 1998, Calcium Oxalate: A Surface Treatment for Limestone, in «Journal of Conservation and Museum Studies», 4, Institute of Archaeology, University College London, London. Charola A. E., Centeno S. A., Normandin K., 2010, The New York Public Library : Protective Treatment for Sugaring Marble, «Journal of Architectural Conservation», Vol. 16, No. 2, pgs 29-44. Conti C., Colombo C., Dellasega D., Matteini M., Realini M., Zerbi G., 2011, Ammonium oxalate treatment: Evaluation by μ-Raman mapping of the penetration depth in different plasters, «Journal of Cultural Heritage» Vol. 12, pgs 372-379. Croveri P., 2003, Consolidation of globigerina limestone (Malta) by means of inorganic treatments: preliminary results, in 10th International Congress on Deterioration and 1276 Built Heritage 2013 Monitoring Conservation Management Conservation of Stone, Sweden, ICOMOS. Croveri P., 2004, Metodologie di consolidamento di materiali lapidei nell’area Mediterraneo: La Globigerina Limestone maltese – degrado e consolidamento, Dottorato di Ricerca in Scienza per la Conservazione dei Beni Culturali XVII cicolo, Universita degli Studi di Firenze. Dreyfuss T., Cassar J., 2012, The performance of an induced calcium oxalate surface on Globigerina Limestone, in 12th International Congress on the Deterioration and Conservation of Stone, New York. EN 12370:2000: Natural stone test methods – Determination of resistance to salt crystallisation. EN 15801:2009: Conservation of cultural property – Test methods – Determination of water absorption by capillarity. Mallia-Milanes V., 1988, The British Colonial Experience 1800-1964: The Impact on Maltese Society, Mireva Publications. Matteini M., Moles A., 1986, Le patine di ossalato di calico sui manufatti in marmo, OPD Restauro, Restauro del Marmo – Opere e Problemi, Opus Libri, Firenze. Matteini M., Moles A., Giovannoni S., 1994, Calcium oxalate as a protective mineral system for wall paintings: methodology and analyses, in 3rd International Symposium on the Conservation of Monuments in the Mediterranean Basin, Venice, Italy. Matteini M., 2007, Conservation of stone monuments and artefacts: new possibilities offered by the ammonium oxalate based treatment, http://www.eu-artech.org/files/ CUBA/MATTEINI.pdf Mifsud T., 2006, The treatment of weathered Globigerina Limestone: The surface conversion of calcium carbonate to calcium oxalate, A dissertation presented to the Institute for Masonry and Construction Research of the University of Malta in part fulfilment of the requirements for the Degree of Master of Science in Conservation Technology for Masonry Buildings. Mifsud T., Cassar J., 2006, The treatment of weathered Globigerina Limestone: the surface conversion of calcium carbonate to calcium oxalate, Proceedings of Heritage, Weathering and Conservation, Madrid, pgs 727-734. Miliani C., Doherty B., Pamplona M., Selvagg R., Matteini M., Sgamellotti A., Brunetti B., 2007, Efficiency and resistance of the artificial oxalate protection treatment on marble against chemical weathering, «Applied Surface Science», Vol. 253 pgs 4477-4484. Elsevier. Pinna D., Salvadori B., Porcinai S., 2011, Evaluation of the application conditions of artificial protection treatments on salt-laden limestones and marble, «Construction and Building Materials», Vol. 25 pgs 2723-2732. Elsevier. 1277