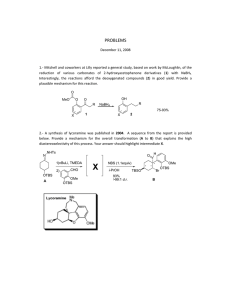
University of Warwick Department of Chemistry
advertisement

University of Warwick Department of Chemistry Advice for successful arrow pushing (suggestions from MW). 1) Don’t push arrows from protons – they have no electrons. CH3 O Avoid: H CH3 Instead push arrow to proton: O H H wrong - no electrons on a proton. H correct 2) Substitution at unhindered saturated (sp3) C atoms is via SN2 mechanism (and inversion when there is a chiral centre): OMe d- OMe OMe H Et Me via transition state: H Et H Me Et Me Br d- Br Br Note that the reaction proceeds via a transition state, as illustrated. 3) Substitution at tertiary sp3 atom is via SN1 mechanism and a cation intermediate. MeOH H OMe OMe Me Me Ph -H Me Me Et Ph Me -Br Br Ph Me Me Me cation intermediate (i.e. it is not a transition state). 4) Substitution at a sp2 C atom is via a tetrahedral intermediate: MeO Ph Cl OMe Cl Ph Ph -Cl O O OMe O Tetrahedral intermediate 5) The two steps above can be condensed into one: 1 MeO MeO Ph OK Cl Ph OMe Ph Cl OR -Cl Ph OMe -Cl O O OK O O Me O Ph But you MUST show the arrows on the C=O bond, i.e. this is INCORRECT: Cl WRONG -Cl Ph OMe O O 6) Bromine addition goes via the bromonium ion: Br Br Br Br Br Br (although not illustrated, note that the bromonium ion opening proceeds via an SN2 mechanism, and the bromide atoms in the product end up trans to each other). 7) The addition of ‘HX’ goes via a cation (the most stable one if there is a choice): Br Br H Br H H Note – there is no cyclic intermediate in this case because the proton is too small to bridge the bond (in contrast the Br+ is big enough). 8) Elimination – the E2 reaction is a concerted reaction: Br Base: alkoxide, LDA etc. (do you know what these are?). H :base 9) Protons at the a-position to a carbonyl group are acidic because an enolate is formed, with the charge on O, not C. This is how it is formed: You may have seen an enolate drawn like this. But remember that is doesn't really have a charge on the C atom: O O H O :base 2 10) The aldol reaction is a remarkably important and valuable synthetic reaction. Learn the mechanism: First deprotonate: O O + H-base :base H (could be from the protonated base, or another source) Then react with an aldehyde or ketone and finish by protonating: O O O H OH O O R R R In some cases an elimination can take place: O O OH H R R :base 11) Grignard Reagents have partial ionic and partial covalent character. Draw one form or the other in mechanisms: MeMgBr = Me MgBr or O Me MgBr O but AVOID: Me MgBr So if you draw the ionic version, the arrow comes from the bond. If you draw the ionic one then the arrow comes from the negative group. 12) Lithium aluminium hydride is a more powerful reducing agent than sodium borohydride. LiAlH4 will reduce virtually all C=O containing substrate, whereas NaBH4 will generally reduce only aldehydes and ketones (although there are some exceptions). LiAlH4 will instantly ignite into flames if you splash water on it whereas NaBH4 can be used as a reagent in a mixture of methanol and water! 3