Monthly Seminar Program
advertisement

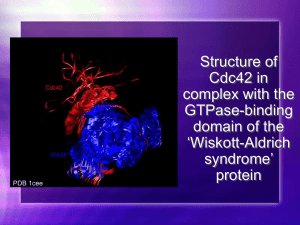
Monthly Seminar Program Guest Speakers: Dr. John Ward Research Fellow Speaker: Dr. Wayne Croft Phd Speakers: Eva Caamaño Kathryn Richardson Paddy Slator 1st March 2013 Seminar program Time 13:00-14:00 Session Lunch Location Common room 14:00-15:00 John Ward MOAC Seminar room 15:00-15:15 Tea and co↵ee break Common room 15:15-16:15 3 Phd Presentations Phd presentations consist 15 minute talks (including questions) audience rotates between three rooms Eva Caamaño Kathryn Richardson Paddy Slator MOAC Seminar Room WSB 32 WSB 336 16:15-16:20 Break Common room 16:20-16:45 Wayne Croft MOAC Seminar room 16:45 onwards Wine and Cheese Common room 1 Presentation Description Guest Speaker Session JOHN WARD Quorum sensing Dr. John Ward research interest lie the application of mathematical modelling to problems in biology and medicine. This talk will focus on quorum sensing. More information about Dr Ward’s research interests including a list of publications can be found at: http://homepages.lboro.ac.uk/ majpw2/ Research Fellow Session WAYNE CROFT Oscillatory dynamics of Cdc42 for polarised cell growth. A fundamental problem in cell biology is how cells determine discrete sites for growth. The capability to polarise (form asymmetric shape and/or asymmetrically distribute proteins/functions) is an absolute requirement for most cell-types. Cell polarity is essential for development and maintenance of tissue integrity, loss of polarity is considered a hallmark and precondition for human cancer, and its dysregulation can cause tumour cell invasion into neighbouring tissues. Uncovering the molecular mechanisms that enable cells to polarise, define specific growth sites and to migrate is vitally important for the development of innovative treatments to cell polarity related diseases. The highly conserved Rho-like GTPase Cdc42 is a central player coordinating the actin cytoskeleton for polarised cell growth. We utilise time-lapse fluorescence microscopy, quantitative image analysis and kinetic computational modelling to investigate mechanisms regulating the spatio-temporal activity of Cdc42 in determining cell growth sites. Recycling of a Ras protein upstream of Cdc42 is shown to be required for Cdc42 oscillations and subsequent bipolar cell growth in fission yeast. Phd Session 1: EVA CAAMAÑO Systems biology, metabolic modelling and metabolomics of the malaria parasite. Malaria is a vector-borne infectious disease whose pathology and symptoms are caused by a protozoan parasite of the genus Plasmodium to whose vector is the female mosquito of the genus Anopheles. Malaria is one of the biggest killers in the world. In 2009, 225 million cases were reported, 2 resulting in over half a million deaths. Unfortunately, no e↵ective vaccine is available and, although there are e↵ective medications to both treat and prevent malaria, resistance has developed to several of them. This resistance is an increasing concern and emphasize the need for an economically viable long-term treatment not susceptible to reduce its e↵ect. Plasmodium has a complex life cycle with di↵erent stages. Despite all the published knowledge about the parasite such as the genome sequence in 2002 or the metabolic models by Plata et al. and Huthmacher et al., there are still big questions about the metabolic make up of the parasite. Understanding how the metabolism of Plasmodium works, where the drugs act and how likely are these targets, to be replaced in the metabolism creating resistance, is of paramount importance in order to find new unique targets with low probability of being tricked by the parasite. The aim of my PhD research is to take a systems approach integrated with metabolomics and metabolic modelling to create a metabolic network model for P. falciparum (the most deadly among the genus). The novelty of this PhD includes the availability of specific quantitative metabolic data that will allow the creation of a biologically qualitative and quantitative accurate model. Metabolome readouts from parasites collected in response to drug perturbations will be included in the modeling to evaluate the model and enhance its predictive power. This approach will allow us to identify key nodes of regulation and determine how metabolism responds to changes in the environment/drug perturbation. Phd Session 2: KATHRYN RICHARDSON Desensitisation mechanisms of GPCR signalling pathways. A chalk talk introducing GPCRs, yeast, yeast genetics and desensitisation mechanisms. Phd Session 3: PADDY SLATOR Characterising cell membrane heterogeneity through analysis of particle trajectories. The organisation of the plasma membrane is complex. It is clear that there is heterogenous structure which mediates functionality and a↵ects the 2D di↵usion of biomolecules. Single particle tracking and STED-FCS microscopy allow direct observation of the behaviour of single proteins and lipids within the membrane (Eggeling et al., Sahl et al.). My project aims to robustly interpret this data. 3