Biocatalysis
advertisement

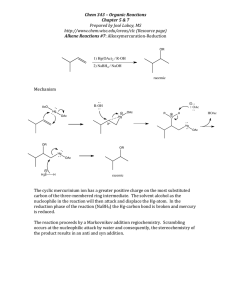
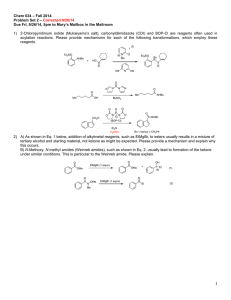
Biocatalysis Ta nj a G ul de r Enzymes are catalysts evolved in nature to achieve the speed and coordination of a multitude of chemical reaction necessary to develop and maintain life. B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Induced fit Model for the enzyme-substrate interaction introduced by Koshland Enzymes are globular proteins which range from 62 (monomer of 4-oxalocrotonate tautomerase) to over 2 500 amino acid residues (animal fatty acid synthase), but only a small portion (~ 3-4 amino acids are directly involved in catalysis) Classification Oxidor eductases catalyze oxidation/reduction reactions T r ansfer ases transfer a functional group (e.g. methyl group) Hyd r olases catalyze the hydrolysis of various bonds Lyases cleave various bonds by means other than hydrolysis and oxidation Mechanism of trans itions stat e st abilization Isomer ases catalyze isomerization changes within a single molecule Catalysis by bond strain Ligases join two molecules with covalent bonds Enzyme reaction affinity of the enzyme to the transition state is greater than to the substrate itself ground state destabilization effect Catalysis by proxim ity and orientation enzyme-substrate interactions align reactive groups and hold them close together reduces the overall loss of entropy Catalysis involving pr oton donor s or acceptors (acid/base catalysis) stabilization of developing charges in the transition state activation of nuceophiles and electrophiles or stabilization of leaving groups 1 B a ra n Gr ou p Me eti ng 07 /1 1/2 0 09 Biocatalysis Ta nj a G ul de r pyruvate decarboxylase mechanism initial step of the serine protease catalytic mechanism: R' A sp As p H is O H O O N His H O N Me N N Se r H R' H O H N Se r N R' H R O R Electrostatic catalysis stabilization of charged transition states by forming ionic bonds with residues of the active site initial step of the carboxypeptidase catalytic mechanism: O Glu R O O R Glu HN R' H O H C O2 C O2 O HN Zn R' O O H S N Me Me R th ia mi n e py ro ph o sp h ate (TP P, vita mi n e B 1) R' S Me N R OH O Me OH OH O C O2 O R' S H Me N R Me H OH H R' S Me Me N R OH Advantages of biocatalysts and enzymes - very high enantioselectivity - very high regioselectivity - transformation under mild conditions - 'green chemistry' e.g. solvent often water Disadvantages of biocatalysts and enzymes H O 2+ Me N R TPP yl id e H O O H R' O S Zn 2+ Covalent catalysis substrate is forming a transient covalent bond with a residue in the active site in order to reduce energy of later transitions states of the reaction - often low specific activity - instability at extreme temperatures and pH values - availability for selected reactions only - long development time for new enzymes advances in genomics, directed evolution, gene and genome shuffling and the exploration of Earth´s biodiversity aided by bioinformatics and high-throughput screening facilitate the discovery and optimization of enzymes It is estimated that biocatalysis and biotransformations account for 30% of the chemical business by the year 2050 2 B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r Literature Biotransformations on an Industrial Scale t/a product > 1 000 000 high-fructose corn syrup > 100 000 lactose-free milk > 10 000 acrylamide cocoa butter > 1 000 nicotinamide D-pantothenic acid (S)-chloropropionic acid 6-aminopenillanic acid 7-aminocephalosporanic acid aspartame L-aspartate D-phenylglycine D-p-OH-phenylglycine > 100 ampicillin L-methionine, L-valine L-carnitine L-DOPA L-malic acid (S)-methoxyisopropylamine (R)-mandelic acid L-alanine enzyme glucose isomerase lactase nitrilase lipase nitrilase aldonolactonase lipase penicillin amidase glutaryl amidase thermolysin aspartase hydantoinase hydantoinase penicillin amidase aminoacylase dehydrase/ hydroxylase !-tyrosinase fumarase lipase nitrilase L-aspartate-!-decarboxylase further applications: baby foods, brewing industry, fruit juice, dairy industry, starch, paper, biofuels, detergents, rubber,.... - K. Drauz, H. Waldmann, Enzy me Cataly sis in Or ganic Synthesis, Wiley-VCH, 2002 - V. Gotor, I. Alfonso, E. Garcia-Urdiales, Asy mmetric Organic Synthesis w ith Enzy mes, Wiley-VCH, 2008 - E. Garcia-Junceda, Multi -Step Enzy me Catalysis, WileyVCH, 2008 - D. Enders, K.-E. Jaeger, Asymmetric Synthesi s with Chemi cal and Bioblogical Methods, Wiley-VCH, 2007 - A.S. Bommarius, B. R. Riebel, Biocataly sis, Wiley-VCH, 2004 - G. Carrea, S. Riva, Organic Sy nthesis with Enzymes in NonAqueous Media, Wiley-VCH, 2008 Desy mmetriza tions Me O2 C Pi g l iv er e ste ra se Me O2 C H 2 O/ ac eto n e O O O H O2 C R m es o Me O2 C Pi g l iv er e ste ra se MeO C O2 Me C O2 Me p H 8, 7 d pr oc hi ra l O O O by sso c hl am ic ac id Wh ite et a l., JAC S . 2 0 00 , 86 6 5 9 8 % , >9 9 % e e HO 2C Me O CO 2H N H S CO 2Me 89%, 95% ee Cl OMe ( -)-v ira n tmy cin Wu lff e t al ., A C IE 2 0 04 , 64 9 3 enantiotopos-differentiating hydrolysis 3 B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r Dynamic k inetic resolut ions C AL B , NH2 enzyme-metal combination Ph Me Ph 4 , Na 2 C O 3, to lu en e , 9 0 °C , 3 d lipase R-sec-alcohol Ac OAc NH R 90% 9 8 % ee Me R-ester Ac yl- OR Ru-catalysts: metal subtilisin S-sec-alcohol S-ester O Ph A cyl -OR Ph Ph OAc Ph OH Ph Me Cl 1, to lu e ne , 70 °C, 2 4- 72 h Ph R Me 7 8- 92 % > 9 9% e e Ph Me OAc CO [R u ], K OtB u, N a 2C O 3, to lu e ne , rt Ph R Me OC Ph Ph Ph CO Ru Ph Cl OC 2 CO S ho v' s ca tal ys t 1 Ph Ru OC Ph Cl O R Ph Ph 7 8 -9 2% >9 9 % e e OAc Ph Ru Ph C A LB , Ph H Ru OC OAc used for the production of R-phenylethanol by DSM OH H N Ph Ph Ruthenium-catalyzed reactions C A LB , O H R R 3 CO R O O H OC R R R H Ru Ru CO OC R CO 4 [R u ] = 2 , 31 h [R u ] = 3 , 3h Ph R = p -M eO -C 6H 4 O Ph Ph O OH Ph Me su b tili si n, P rC O 2 C H 2C F 3 2 , TH F, r t OC O Pr Ph S Me 95% 92 % ee Ph Ru Cl CO 5 Cl Ru Cl 6 2 4 B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r Tandem-DRK-Diels-Alder reaction OH O C AL B, EtO Meerwein-Ponndorf-Verley-Oppenauer reaction CO 2Et O O OH C O2 Et O Ph OAc C AL B, Al Me 3 Me Ph OAc 9 6% , 9 6% e e Me BIN OL 6 , NE t 3 , M S, Me C N , 3 5 °C , 3 d 8 1% , 97 % e e O CO 2Et to l ue n e, rt, 3 h O DRK with enzyme-base combination R Hydantoinase-carbamylase system R Palladium-catalyzed reactions OAc Me OAc Ph Me - P d( 0) + Pd (0 ) OH TH F, 25 °C , 1 .5 d Me R S Ph 7 1% , 9 8% e e i P rOAc AcO H2 N EtO OH Me or ra ce ma se HN D -hy da n toi n as e C O2 H NH R C AL B, V O(OS iPh 3 )3 , a ce ton e , 2 5 °C , 4 .5 d 9 1% , 9 9% e e N H2 O D -ca rb o my ol as e 10 0 kg sca le p il ot p ro ce ss fo r te rt -Le u ci n a t De g us sa OAc Me R CO 2H HN L -ca r bo m yo la se OAc NH R O O Vanadium-catalyzed reactions Me R L OH O O bo ra te b u ffe r, R p H 9, 4 0o C L- hy da n toi na s e Ph Pd NH HN C A LB , i P rOH Ph O ( S) R CO 2H NH2 R CO 2H NH2 5 B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r Cyanohydrine-mediated DRK Ox idations Cl Cl O M an d el o ni tril e ly as e KC N , i Pr OH /H2 O OH Cl R CN OH HCl R C O2 H solutions: - closed-loop systems with an additional enzyme for co-factor regeneration - electrochemical co-factor recycling - application of metals for regeneration - living whole cells qu a n t., 8 3% e e produced by DSM Chemie Linz, Nippon Shokubai, Clariant Cl CO 2Me N a n ti p la tel et cl o pi d og re l (P la vi x) drawback: co-factor dependence of oxidases/reductases S de h yd ro g en a se su b stra te (re d .) N AD (P ) OH OH O H CN CN n itr il as e pH 7 .2 , 4 0 oC Ps eu d o mo n a s ce p ac ia li p as e R CO 2H la cta te d eh yd ro g e na se re g e ne ra tio n o f e n zym e co su b stra te e .g. p yru va te (o x.) Oxidations of alcohols and amines 1 2 -H SD H OAc S CN N A D( P)H >9 5 %, >9 9% e e applied by Lonza, BASF, and Mitsubishi Rayon on a multiton scale OAc by pr od u ct e .g. la cta te (r e d.) p ro d uc t (o x.) OH Me R regioselective oxidations of bile acid depending on hydroxysteroid dehydrogenase used >9 6 %, >8 4% e e nonselective nitrile hydratase: Rhodococcus r hodochr ous J1 - acrylamide production (Nitto process, > 20 000 t/a) - nicotinamide synthesis (Lonza, 3000 t/a) Me HO OH OH 7 -H S DH 3 -H SD H 6 Oxygenation of nonactivated carbon centers Deracemization of secondary alcohols OH Me R2 L -l a ctate o xid a se L -L ac tate O OH Me N a BH 4 C O2 H R1 p y ru va te CO 2H Me Deracemization of tertiary amines NaO 2 C H S- am in e ox id a se va ri an t Na B H4 N HMe pH 7 R N Me N u n na tu ra l en a nti om e r N Me O H O CO 2H H NH HO HO N St re p to my ce s Me c a rb o ph i l us Me N e p ib a ti d in e: a na l ge si c O Me H Me HO p ra va stati n (P ra va ch o l) Epoxidation O N HO produced by BMS and Sankyo Pharma 3.6 billion US Dollar annual market value ni co tin e P se u d om o na s s p. D SM 86 5 3 S N Me OH N aO 2C M L 236B fro m P en i ci l l i um ci tr i nu m N S Me N HO O pr od u ctio n of co rti co ster o ne OH O Me H 95 % , >9 5 % ee A r thr o b ac te r o x yd a n s O 70% N N H HO O Me H H R 1 = O , OH R 2 = H , Me D -L a cta te H al mo s t a ll C a to ms a t th e ste ro id n uc le u s ca n b e hy dr ox yl ate d ste re os pe ci fic al ly OH O Me HO 3 Me R N aB H 4 CO 2H Me B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r R R h o d oc oc cu s equi 70 % O O R R OH R = CH 2 C H 2OM e O m eto pr o lo l Lo p re so r/To p ro l-X L h y pe rte ns io n R HN 7 O Baeyer-Villiger-Oxidations O O O c ycl op e nta n on e m on o ox yg en a se Co m am o na s N C IMB 98 7 2 O O HO O S NH HO B PD O O OH OH ( +)-sh o wd o my ci n S 7 0% , 9 5% e e Ph O OH H HO R ud ro ff e t a l . C h em . C om mu n . 20 0 6, 3 21 4 HO Br H H O O2 O RS (+)-tr a n s-ku m au sy ne g on i ofu fu ro n e a n al og s OAc Br R cyto ch ro m e-ty pe mo n oo xy ge n a se RL model for predicting the regio- and stereochemical course for the cis selective dihydroxylation reaction O Aryl dihydroxylations O B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r R R b a cte ria l d io x yg en a se e uk ar yo ts p ro ka ryo ts o r tho a nd me ta h yd ro xyl ati on o ccu rs us in g to lu e ne (TD O, Ps e ud o m on a s pu ti d a F3 9 /D ), n a p hth al e ne (N D O, P . pu ti d a 11 9 ), or bi ph e n yl d io xy ge n as e s (BP D O, Os el tam iv ir NH2 H3 PO4 (Tam if lu ) OH Fa n g e t al . A C IE 20 0 8 , 5 7 88 OH Me Me OH TD O 1. D MP 2. O 3 /D MS S ph i n go m on a s y an o i ku y a e B8 /3 6) OH R CO 2 Et AcHN OH 77%, 99% ee OH O Br TD O Me O O O O R OH TD O OH R = H , Cl , B r, I, M e , CN , C O 2 Et, etc Al 2O 3 O CO 2H OH NDO Me OH HO OH PGE 2 ! O O O H u dl ic ky e t al . J AC S 1 9 8 8, 4 73 5 8 B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r ipso and ortho dioxygenations possible with Ralstonia and Pseudomonas mutant strains CO 2H A lc a li g e ne s eu tr o ph y u s HO 2C str ai n B 9.4 .5 OH HO 2C OH c ar ro t C O2 R O O R CO 2R X X X = H , o -C l , p -C l, p -M e; R = M e , E t 9 2 -99 % e e OH >95 % ee OH O OH Formation of the 3,5-dihydroxy side chain in statins P ar ke r e t a l. S yn l e tt 20 0 4, 2 0 95 O X = O: to p ira m ate an ti- ep i le ps y, an ti- mi gr an e H2 NO 2SO O X O N H O Re ductions O Reduction of aldehydes N O Me HO h o rse li ve r a lc oh o l d e hy dr og e n as e Me Me OH + HO 2C N AD , E tOH (OC )3Cr (OC )3Cr S 33 % , 91 % e e Reduction of ketones OMe S a to rv a stati n (l ip ito r) produced by Pfizer 12.4 billion US Dollar 2008 OH 3,5-dihydroxy side chain common in all statins R 5 1 % , 8 1 % ee OMe R N H (OC )3Cr d il tia ze m h yp er te n si on , an g in a p e cto ri s, a n d s om e typ e s o f ar rh yth mi a H F O ke to r ed u cta se O Cl CO 2 Et OH Cl S h a lo h yd rin de h yd ro g en a se CO 2Et OH NC R CO 2 Et OMe O O C h a da et a l. J . M o l . C a ta l. B 2 0 0 4, 1 03 H S S N H O O baker's yeast 80 % , >9 9% e e H S S S N H OH O O a tor va sta ti n H 2N O CO 2tBu 9 Ca rbon-Ca rbon coupling reac tions dihydroxyacetone phosphate (DHAP)-dependend aldolase stereodivergent product generation possible using stereocomplementary enzymes Aldolreactions -> generation of 2 stereocenters 1,6-bisphosphate aldolase (FruA) H 1 R R2 R R2 O O O 1 A ld o la se (L ya se c la ss ) OH HO R HO OH OMe OBn OMe OBn 1 . 1 ,6- bi sp h os ph a te a ld o la se (Fru A ) O R2 H O B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r O R1 R2 1 OH O A ld ol a se / Tra n sa ld o la se (Tr an sfe r as e cl a ss) O HO OPO 3 2. p ho sp h ata se 42 % OMe OH OH OH OH OH HO OH R1 R2 O O HO O O O HO Ke tol a se (L ya se cl as s) R1 1 R2 OH R1 HO O O OMe Me Me Sh i ma g ak i e t a l. C h em . P ha r m B ul l . 19 9 3, 2 82 OH p en ta myc in O R OH OH C5 H11 Thiamine diphosphate dependent conversions R2 OMe O HO OH O H HO O Ke tol a se / Tra ns ke tol a se (Tra n sfe ra se cl as s) R2 Enzyme classification dependent on the nucleophile: 1. pyruvate-dependent aldolase 2. dihydroxyacetone phosphate (DHAP)-dependend aldolase 3. acetaldehyde-dependent aldolase 4. glycine-dependent aldolase acetaldehyde-dependent aldolase 2-deoxyribose-5-phosphate aldolase (DERA) -> generation of 1 stereocenters O Cl O Me OH O D ER A Cl 70 % , >9 9.5 % e e, 9 6.6 % d e 4 s te p s OH O ato rv as tati n NC O CO 2tBu 10 O O HO B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r O D ER A OH O Tot al Synthesis of Natural Products O i n v itro reconstitution of complete biosynthetic pathways Me 46% Me Me C O2 H Me OH OH L-tr yp to p h an : ph e ny lp yr uv ate a mi n otra n sfe ra se Td iD NH2 O H HO Me Me OH S Me N Me O Me Me S Me CO 2H C O2 tBu e p oth il o ne A HO O O Me S Me N O O O HN OH O S NH HO HO O O OH Me Th D P S Me q u in o ne re du cta se Td iC OH HN HO N AD P H OH NH OH (-) -e ph e dr in e Me HO Me N N O6 P2O HN OH D id em e thy lO a ste rri qu i no n e D NH NHMe O CO 2 H Me R - ph e ny la ce tyl c ar bi n ol > 9 8% e e -C O 2 O OH O Me P DC , Th D P H2 N Me , H2 , Pt H o ffm ei ste r C e l l 2 0 0 7, 635 HO NH NH 21 % PD C , Th D P OH O O Me 3 5% Pyruvate decarboxylase (PDC, thiamin diphosphate depended) Me Td iA O OAc O bi si nd o ly lqu i no n e s yn the ta se Tdi A AT P NH 2 O Td iA O D ER A OH CO 2 H OTBS Me Me N H Me Wo ng e t a l. A CI E 2 0 02 , 14 0 4 S O N H PMPO O Me O Me CO 2H Me N H2 N O HN p r en yl tra ns fer as es Tdi B/Td iE HN Wa l sh e t a l. N a t. C h em . B i ol . 2 00 7 , 5 84 O OPP te rr eq u in o ne 11 O a cyl ca rri e r p ro te in En cC li ga se En cN A TP OH 7x ke to sy nth a se E nc A-E nc B, k eto re du c ta se En cD , tra n sa cy la se Fab D O O O HO S EncC HO O Ph O O O O C OSEn cC O O O fe rre d o xin , fe rr ed o xin -N A DP + r ed u cta se En cR OH Ph O O O O HO 9 Ph O HO wa i lu pe m yci n F O O COSEncC O O de sm e th yl -5 d e ox ye nte ro ci n ~ 2 5 % o ve ra l l yi el d ; for ma tio n o f 1 0 C -C , 5 C -O, a n d 7 ste re o c en te rs i n o n e p ot O OH Ph OH HO O HO NA D P O H O Ph OH OH HO O 5 -d eo xy en te ro ci n " fav o rsk iia se " fl av op ro te in En cM HO 5 SA M O MeO O me thy ltra ns fer as e En cK HO 9 OH O O H O O OH Ph OH 5 HO Ph HO O H O EncC S O w a il up e myc in G Ph O O O HO OH O HO OH HO 9 O EncC O OH Fa v or ski i re ar ra ng m en t O S O O NA D PH 7x SCoA O B ar an Gro u p M ee tin g 0 7 /11 /2 00 9 Biocatalysis Ta nj a G ul de r HO O MeO e nte ro ci n O M oo re e t a l. N a t. C he m. B io l . 20 0 7, 5 57 12