Introduction Baran Group Meeting 10/5/05 Aphidicolin
advertisement
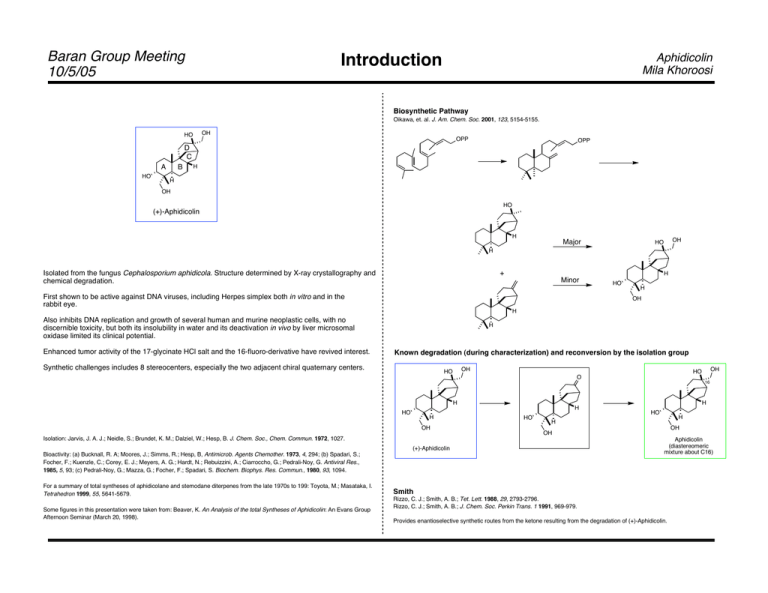
Baran Group Meeting 10/5/05 Introduction Aphidicolin Mila Khoroosi Biosynthetic Pathway Oikawa, et. al. J. Am. Chem. Soc. 2001, 123, 5154-5155. OH HO OPP OPP D C A HO B H H OH HO (+)-Aphidicolin H Major OH HO H Isolated from the fungus Cephalosporium aphidicola. Structure determined by X-ray crystallography and chemical degradation. + H Minor First shown to be active against DNA viruses, including Herpes simplex both in vitro and in the rabbit eye. HO H OH H Also inhibits DNA replication and growth of several human and murine neoplastic cells, with no discernible toxicity, but both its insolubility in water and its deactivation in vivo by liver microsomal oxidase limited its clinical potential. Enhanced tumor activity of the 17-glycinate HCl salt and the 16-fluoro-derivative have revived interest. H Known degradation (during characterization) and reconversion by the isolation group Synthetic challenges includes 8 stereocenters, especially the two adjacent chiral quaternary centers. OH HO H HO H OH Isolation: Jarvis, J. A. J.; Neidle, S.; Brundet, K. M.; Dalziel, W.; Hesp, B. J. Chem. Soc., Chem. Commun. 1972, 1027. H HO H HO H OH OH Bioactivity: (a) Bucknall, R. A; Moores, J.; Simms, R.; Hesp, B, Antimicrob. Agents Chemother. 1973, 4, 294; (b) Spadari, S.; Focher, F.; Kuenzle, C.; Corey, E. J.; Meyers, A. G.; Hardt, N.; Rebuizzini, A.; Ciarroccho, G.; Pedrali-Noy, G. Antiviral Res., 1985, 5, 93; (c) Pedrali-Noy, G.; Mazza, G.; Focher, F.; Spadari, S. Biochem. Biophys. Res. Commun., 1980, 93, 1094. Some figures in this presentation were taken from: Beaver, K. An Analysis of the total Syntheses of Aphidicolin: An Evans Group Afternoon Seminar (March 20, 1998). 16 H (+)-Aphidicolin For a summary of total syntheses of aphidicolane and stemodane diterpenes from the late 1970s to 199: Toyota, M.; Masataka, I. Tetrahedron 1999, 55, 5641-5679. OH HO O Aphidicolin (diastereomeric mixture about C16) Smith Rizzo, C. J.; Smith, A. B.; Tet. Lett. 1988, 29, 2793-2796. Rizzo, C. J.; Smith, A. B.; J. Chem. Soc. Perkin Trans. 1 1991, 969-979. Provides enantioselective synthetic routes from the ketone resulting from the degradation of (+)-Aphidicolin. Baran Group Meeting 10/5/05 Aphidicolin Mila Khoroosi First Syntheses: Trost and McMurry Trost: A vinyl-cyclopropane rearrangement McMurry: A Robinson Annulation Trost, B. M.; Nishimura, Y.; Yamamoto, K.; McElvain, S. S. J. Am. Chem. Soc. 1979, 101, 1328-1330. McMurry, J. E.; Andrus, A.; Ksander, G. M.; Musser, J. H.; Johnson, M. A. J. Am. Chem. Soc. 1979, 101, 1330-1332. O O O 1. HOCH2CH2OH, TsOH 1. LiAli-Bu2-t-BuH, -78 °C 2. Li/NH3; TMSCl 3. MeLi; CH2O O 2. 3 N HCl 3. Me2CO, TsOH O O O HO LDA; 1. OsO4; NaIO4 I O O O O OTMS O O O Trost intermediate 1. PhSeSePh NaBH4 SPh2 O 2. NaH O O 2. MeC(OTMS)=NTMS Et3N O O O 1. LAH TMSO H 3. Li/NH3 4. MeC(OTMS)=NTMS 1. n-BuLi, I O O 360 °C 2. EtOCH=CH2 O 1. 610 °C 2. Pd(OAc)2 CHO O O O 2 H O O O BH O O 2. H2O2, NaBH4 O O CH2OTs 1. LAH OH OH O O H O H O Na2Fe(CO)4 H 2. p-TsCl, py O 1. DHP, TsOH 2. NH2NH2, KOH PCC; KOH O O O 3. Me2CO, TsOH 4. PCC O O H O O O Baran Group Meeting 10/5/05 Aphidicolin Mila Khoroosi Corey Corey Corey, E. J.; Tius, M. A.; Das, J. J. Am. Chem. Soc. 1980, 102, 1742-1744. S S S S Me O- CO2Me O- Br O NaH, (EtO)2POCl TBSO 1. TMSCN, ZnI2 O t-Bu Me 2. DIBAl-H, 0 °C Me O OMe S Me O H t-Bu O CHO OTMS S 1. LiSiMe3 Me 2. LDA; H+ Me O H t-Bu O H O TBSO O CO2Me OP(OEt)2 1. NaBH4 2. TBSCl 3. deprotection CO2Me Me O 1. Hg(OCOCF3), MeNO2, 0 °C Me ClHg 2. NaCl, H2O TBSO CO2Me Me O 1. HOCH2CH2OH, TsOH 2. NaBH4 Me 4. H2, Pd/C 5. (n-Bu)4NF 2. TsCl H 3. PDC 4. (n-Bu)4NF 5. L-Selectride O t-Bu CHO Me H O O 1. O t-Bu H O O H t-Bu O TMSS STMS Li 1. ketalization 2. separation OEt H 2. HOAc, MeOH/H2O HO 3. deprotection H O t-Bu HO H O HO Me HO O H H H (±)-Stemodinone H (+)-Aphidicolin Applications to other natural products ZnI2 H HO OH Me NH • HOAc OH HO (±)-Aphidicolin 2. O OH O Me H Me OH 1. MVK, DBU, K2CO3 Me LiN(t-Bu)2, -120 °C O HO Me 3. PCC 4. HClO4 H2O/acetone OTs O O t-Bu 1. t-BuCHO, TsOH 2. LAH O Me Me TBSO O H (±)-Stemodin Me H CHO Baran Group Meeting 10/5/05 Aphidicolin Mila Khoroosi van Tamelen and Bettolo Van Tamelen: A biogenetic approach Bettolo: [2+2] cycloaddition van Tamelen, E. E.; Zawacky, S. R.; Russell, R. K.; Carlson, J. G. J. Am. Chem. Soc. 1983, 105, 142-143. Bettolo, R. M.; Tagliatesta, P.; Lupi, A.; Bravetti, D. Helv. Chim. Acta 1983, 66, 1922-1928. OMe O OMe 1. NBS; K2CO3 2. LiN(Et)2 O O 1. NaH; BnBr; H3O+ 1. FeCl3 3. VO(acac)2 t-BuOOH 4. NaH, BnI 2. Li/NH3 3. Me2CO, TsOH O OBn 2. HCO2Et, NaH 3. MVK HO H O HO H O O O O O HO O O O O H H H O O H BnO H 2. O3 3. NaBH4 BnO BnO BnO O O H 1. HOCH2CH2OH, TsOH • H 1. Trisylhydrazine 2. n-BuLi O H BnO BnO O 1. m-CPBA 1. H2, PtO2 2. Pb(OAc)4 H O OH O conditions unclear 1. NaH, MeI, CS2 2. Na 3. MsCl, py H OCSSCH3 OH H BnO BnO OMs OH Me2CO, H2O H O ! H O H O H O O 1. CrO3• py2 H BnO BnO O OH Me2CO, H2O CrO3• py2 O BnO OMs O 2. MsCl, Et3N H BnO O 1. ! 2. NaBH4 H BnO BnO H 2. Li/NH3 O O Baran Group Meeting 10/5/05 Aphidicolin Mila Khoroosi Ireland and Iwata Iwata Ireland Tanaka, T.; Okuda, O.; Murakami, K.; Yoshino, H.; Mikamiyama, H.; Kanda, A.; Kim, S-W.; Iwata, C. Chem. Pharm. Bull. 1995, 43 1407-1411. Ireland, R. E.; Dow, W. C.; Godfrey, J. D.; Thaisrivongs, S. J. Org. Chem. 1984, 49, 1001-1013. MeO CH2 1. O CH2TMS CO2Me H BnO O OBn OBn O ! O + TMSOTf, -78 °C 3 steps CH2SiMe3 O O OMe CO2Et BnO OMs OMs BnO O CH2SiMe3 O CH2SiMe3 1. H2, Pd/C t-BuOK O 1. n-BuLi, i-AmONO 2. NH2Cl O OMs O 2. DIBAl-H 3. Ph3P=CH2 O BnO + unwanted diastereomer 2. Separation N2 O O BnO hv O CH2 O O O SiO2 O O O O HO O TMS 2. Li, O O O EO O 1. LDA, PhSeBr; H2O2, pyr O O Br 1. PCC HO 2. Me2CuLi O O 8 steps Provides a route to another natural product O O O CH3 H HO CH2 H O H (previous work) p-TsOH O HO H OH O OH O O (±)-Aphidicolin "-Chamigrene (±)-Aphidicolin OH HO OH HO Baran Group Meeting 10/5/05 Aphidicolin Mila Khoroosi Enantioselective Syntheses Holton: First enantioselective synthesis Tanis: Cationic Cyclization Holton, R. A.; Kennedy, R. M.; Kim, H.-B.; Krafft, M. E.; J. Am. Chem. Soc. 1987, 109, 1597-1600. Tanis, S. P.; Chuang, Y.-H.; Head, D. B. J. Org. Chem. 1988, 53, 4929-4938. 1. PhCOCl 2. SeO2, TBHP O + TBSO OLi S(O)(Tol) O O -78 °C TBSO O 2. HF MgCl O O S(O)(Tol) S(O)(Tol) O O NaOMe O 3. L-DIPT, Ti(Oi-Pr)4 TBHP 4. PhCH2Br, NaH S(O)(Tol) O 1. CH2=CHLi OH 4. LAH 5. TBSCl, DMAP, Et3N 6. CrO3, Py Li2CuCl4 O BnO O 2. MgBr2; L-Selectride 3. LAH 3. HOCH2CH2OH, TsOH 4. Li/NH3; CH2O O 1. Me2CO, H+ 2. NBS, DMF 3. nBuLi; MeI HO O HO O O O O O OH HO O Me OTBS O O t-Bu Corey 2. H2, Pd/C H H HO O H OH O H O 10.7 % overall from geraniol HO H OH (+)-Aphidicolin (+)-Aphidicolin OH HO McMurry 1. m-CPBA Me 3 steps HO HO Et3N, -78 °C O 1. (COCl)2, Et3N, DMSO 1. KOt-Bu 2. H2, Pd/C O O BnO O OTBS 2. n-BuLi, p-TsOH LiCl BF3·Et2O O O CHO O BnO O Note: These first three steps can be carried out in one pot in 45 % overall yield. 1. Zn, NH4Cl 2. HOCH2CH2OH, TsOH 3. O3; DMS 1. NaOMe, MeOH n-Bu4NI OCOPh Baran Group Meeting 10/5/05 Aphidicolin Mila Khoroosi Enantioselective Syntheses Toyota/Fukumoto: The Heck Reaction Toyota: Palladium-catalyzed cycloalkenylation Fukumoto, et. al. Tetrahedron 1994, 11153. Fukumoto, et. al. Tetrahedron 1994, 10183. Fukumoto, et. al. Tet. Lett. 1994, 6495. Toyota, M.; Sasaki, M.; Ihara, M. Org. Lett. 2003, 5, 1193-1195. O O O O O LDA, THF Bromoacrolein 1. Pd(OAc)2, P-(o-Tolyl)3 K2CO3, MeCN, ! (90%) Br (89%) MOMO OMOM OMOM O 2. HOCH2CH2OH, PPTS (89%) O OH O Diastereoselection 3:1 O O O O 1. O3 230 °C 15 steps O 2. LDA, I H H MOMO TBSO TBSO O O O 1. CH2=CHOEt, Hg(TFA)2 O O 1. PCC, NaOAc (92%) 2. Ph2P(O)CH2CH2=CH2 n-BuLi, HMPA 1. O2, PdCl2, CuCl (70%) 2. H2, Pd-C (82%) 3. Ph3P=CH2 (81%) 2. Toluene, !, then NaBH4 (82%) OH HO O Ireland Bu4NF, CH2O H H HO HO O HO H H OH HO (+)-Aphidicolin Summary O 1. methylene blue, 230 °C O O 2. O2, hv, porphyrin, pyr 3. NaI, AcOH 4. MnO2 (82%) Iwata Me H O H HO H OH Longest Linear Sequence Overall Yield McMurry 18 steps 0.9% yield Trost 23 steps 0.6% yield van Tamelen 25 steps 0.03% yield Bettolo/Lupi 24 steps 2.9% yield Ireland 28 steps 0.93% yield 37 steps 0.96% yield 27 steps 2% yield 40 steps 0.19% yield 25 steps 2.3% yield OH HO O O 3. KOH 4. Me2CO, PPTS 2. Pd(OAc)2 OH O 1. L-Selectride 2. Me2S+ (O)CH2- 1. LDA; TBSCl Corey Tanis (+)-Aphidicolin Iwata Fukumoto Holton 28 steps 1.65% yield H