CH9080 UNIVERSITY OF WARWICK MASTER OF SCIENCE EXAMINATIONS: MARCH 2010
advertisement

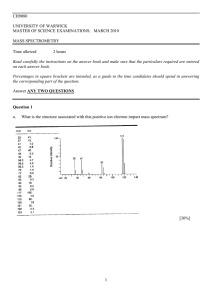
CH9080 UNIVERSITY OF WARWICK MASTER OF SCIENCE EXAMINATIONS: MARCH 2010 MASS SPECTROMETRY Time allowed 2 hours Read carefully the instructions on the answer book and make sure that the particulars required are entered on each answer book. Percentages in square brackets are intende, as a guide to the time candidates should spend in answering the corresponding part of the question. ANSWER ANY TWO QUESTIONS 1 Question 1 [50%, 15% part a, 35% part b] a. (15 pts) What is the structure associated with this positive ion electron impact mass spectrum? Notes: isotope pattern indicates chlorine. 35 is just Cl+. 47 has one chlorine, so it must be CCl+. 82 has two chlorines, must be CCl2+. 117 has three chlorines, must be CCl3+. 118 intensity indicates 1 carbon. 117 cannot be the molecular ion – it’s an even electron ion. So, it’s probably carbon tetrachloride. It cannot be HCCl3 – which would have a molecular ion at 118. It cannot be BrCCl3 (or any other halogen substitution), which would generate a mixed spectrum. Answer: carbon tetrachloride. 2 B. (35 pts) What is the structure associated with this positive ion electron impact spectrum? Assign structures to each peak whose mass is noted on the spectrum. Draw 'fishhook' diagrams for each structure to explain its formation. Notes: alkane series suggests a 4-carbon alkyl group – it goes up to 56 (butyl) and no higher – probably linear n-butyl because no particularly intense peak and lack of a -15 Da loss from the precursor (though in reality, isomer determination is hard here). 77 indicates a benzyl group. Complete lack of 91 indicates that benzyl group has no alkyl derivatives. 105 suggests a carbonyl attached to the benzyl (+28 Da). 123 is 55 below 178, suggesting loss of the whole butyl group (as •C4H7) – this is a pseudo complementary ion to 56. 123-105 = 18, suggesting a bridging ether. Answer: n-butyl benzoate 3 Question 2 A. (15 pts) Draw a diagram of a MALDI-TOF mass spectrometer, label the appropriate parts and describe their function. What is the difference between "linear" mode and "reflector" mode? In linear mode, ions hit the first detector. Resolution is determined by the kinetic energy distribution in the source, and by the flight tube length. In reflector mode, ions are bounced off an electrostatic mirror to the second detector. Resolution is higher because the mirror partially corrects the kinetic energy distribution by making faster (more energetic) ions penetrate the mirror slightly farther - giving them a longer path length to the detector. 4 B. (10 pts) In a linear mode MALDI-TOF experiment, calculate the time-of-flight of singly charged ions using the following parameters: Ion source potential, 20 kV; drift tube grounded. Drift tube length: 1.5 meters. Mass of the ion: 1348.6 Da. 1 2 1 d2 K mv m 2 ; solve for t. 2 2 t thus: t md 2 (1348.6)(1.67e 27 /1.6e 19)(1.5)2 28.1 s 2K 2* 20000 5 C. (15 pts) Draw a diagram of an electrospray quadrupole ion trap mass spectrometer. Explain how the ion trap works, and explain how ions can be "scanned" out into a detector. The ion trap works by trapping ions in an oscillating electric field between the endcaps and ring electrodes. If the amplitude and frequency are correct, ion trajectories inside the trap are stable. Ions can be scanned out to the detector in two ways. First, the RF amplitude can be ramped, causing lighter ions to become successively unstable in the trap and fall out. Second, a resonant RF pulse can be added on top of the main trapping field and scanned to dump the ions out of the trap. Either way, a plot of amplitude or pulse frequency versus ion current is the resulting mass spectrum. 6 D. (10 pts) In a quadrupole ion trap, using the equation below calculate the low mass cut off (LMCO) for the trap if it is run at 500 Vrf amplitude, 850 kHz, a pole-to-pole (z - axis) minimum inner distance of 12 mm, and a z/r aspect ratio of 1.1. LMCO is given by qz 0.908 8eV m (r 2 2 z 2 ) 2 Low mass cut off (LMCO) is given by qz 0.908 8eV ,so m (r 2 2 z 2 ) 2 8V , with V = 500 Volts, ω = 0.908 (r 2 1.12 r 2 ) 2*pi*850 kHz, and r = 12/2 mm, z = 1.1r. So: LMCO = 186 Da. rearranging, we get m / z 2 7 Question 3 [50%, 25% part a, 25% part b] a. (25 pts) Given the sequence of the peptide (VFDKDGDGYISAAELR), assign all the peaks in the spectrum which are labeled with a mass (if possible). This is a CID/CAD MS/MS spectrum of the 2+ ion, at m/z 878.5. Hint: this is the same sequence as in part b, so the results should be consistent. 8 Notes: students will need to calculate this table by hand, and then the assignment (marked above) is trivial. 9 b. (25 pts) Given the sequence of the peptide (VFDKDGDGYISAAELR), assign all the peaks in the spectrum which are labeled with a mass (if possible). This is an ETD MS/MS spectrum of the 2+ ion, at m/z 878.5. Hint: this is the same sequence as in part a, so the results should be consistent 10 Notes: students will need to calculate this table by hand, and then the assignment (marked above) is trivial. 11