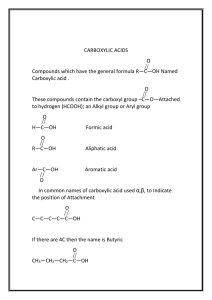
AMINES A STUDENT WHO HAS MASTERED THE MATERIAL IN THIS SECTION... ABLE TO: 1.
advertisement

AMINES A STUDENT WHO HAS MASTERED THE MATERIAL IN THIS SECTION SHOULD BE ABLE TO: 1. Name given the structure, and draw the structure given the name, of amines. Also, give the classification of compounds in any of the following groups, and give examples: 1˚, 2˚, and 3˚ amines; quaternary ammonium salts. 2. Predict the product of reactions giving rise to amines and the reactions of amines, when given the starting materials and reaction conditions. Important reactions include: Acid-base reactions Nucleophilic substitution (including the Gabriel synthesis) Reduction of nitro compounds, azides, amides, and nitriles Reductive amination Reactions of 1˚ aromatic amines with nitrous acid Reactions of diazonium salts Reaction of amines and acid chlorides 3. From your knowledge of the effects involved, predict or explain experimental results. Important areas include: Physical properties (boiling point, solubility) Relative acidity and basicity Nucleophilic substitution (primarily SN2) 4. Use the reactions of #2 above, plus others learned earlier, to propose syntheses of amines, ammonium salts, and related compounds. 5. Predict and interpret IR and NMR spectra, solubility test results, and simple chemical test results to identify unknown amines. Remember that amines, unlike any other common family of organic compounds, are basic. 231 A STUDENT WHO HAS MASTERED THE OBJECTIVES ON THE PREVIOUS PAGE SHOULD BE ABLE TO SOLVE THE FOLLOWING PROBLEMS AND RELATED ONES: 1.1 1.2 Draw the structure of each of the following. a) tert-butylamine b) diisopropylamine Name each of the following compounds. a) (CH3 CH2 )2 CHNH 2 2. c) 3-amino-1-propanol b) c) CH3 CH2 NHCH 2 CH2 CH(CH 3 )2 NH 2 Predict the product(s) (if any) of each of the following reactions. a) b) c) NH 2 + H 2 SO 4 ----> O + NH 2 Pt + H 2 -----> O NH 3 LiAlH4 || C-Cl -------> ----------> ether LiAlH4 d) (CH3 )2 CHCH 2 Cl + NaN3 ----> -----------> ether O C e) CH2 N C O f) 3.1 H3 O + -------> H2 O heat + N N: BF4 - ------> Which of the following compounds is the strongest base? NH 2 NH 2 NH 2 A. B. C. D. CH3 232 OH 3.2 Which of the following species is the strongest acid? NH 3+ A. NH 3+ A. NH 2 B. Cl C. Cl D. Which of the following compounds reacts most rapidly with the N3 – ion in an Sn2 reaction? Cl CH2 Cl A. 4. CO2 CH3 CF3 Which of the following has the greatest solubility in water? NH 2 3.4 D. F CH2 CH3 3.3 C. B. NH 3+ NH 3+ B. H3 C Cl Cl C. D. Propose a synthesis of each of the compounds indicated, from the given starting material and any other needed reagent. O || a) CH2 NH 2 from C-OH b) CH3 CH2 NH 2 from CH3 CH2 OH c) O || CNHCH 2 CH3 from d) NH 2 from O || C-H 233 4. Synthesis, continued. e) f) Br 5.1 OH from C N from For these questions, choose the answers from the list of possible compounds at the end of the section. a) Which of the following compounds gives an aqueous solution that turns red litmus blue? b) Which of them is insoluble in water and soluble in 5% HCl (aq)? c) Which of them is insoluble in water, HCl (aq), and NaHCO3 (aq), but soluble in NaOH (aq)? The possible answers are: CH2 CH3 A. CH2 NH 2 B. E. CH3 CH2 NH 2 5.2 CO2 H C. F. CH 3 CH2 OH OH D. G. CH3 CO2 H An unknown compound had the molecular formula C4 H11 N. Its IR spectrum includes a peak at 3400 cm–1 . Its proton magnetic resonance spetrum follows. doublet, δ 0.9, 6H singlet, δ 1.1, 2H multiplet, δ 1.6, 1H doublet, δ 2.5, 2H What is the structure of this unknown? 234 SOLUTIONS TO SAMPLE PROBLEMS: 1.1 a) tert-butylamine b) diisopropylamine (CH3 )3 CNH 2 c) 3-amino-1-propanol (CH3 )2 CHNHCH(CH3 )2 HOCH 2 CH2 CH2 NH 2 1.2 a) 3-pentanamine b) cyclopentanamine (IUPAC) or cyclopentylamine (common) c) N-ethyl-3-methyl-1-butanamine 2. Predict the product: a) b) c) NH 3 + + HSO 4 - NH 2 + H 2 SO 4 ----> Pt NH 2 + H 2 -----> H2 O + O + O NH 3 LiAlH4 || -------> ----------> C-Cl ether NH CH2 NH 2 LiAlH4 d) (CH3 )2 CHCH 2 Cl + NaN3 ----> -----------> (CH3 )2 CHCH 2 NH 2 ether O C e) CH2 N C O f) 3.1 3.2 3.3 3.4 A C A A 4. Synthesis: a) + + H3 O -------> H2 O heat N N: BF4 - ------> CH2 NH 2 + CO2 H CO2 H F O SOCl2 NH 3 || C-OH --------->-------> O LiAlH4 || CNH 2 ---------> CH2 NH 2 HBr NH 3 , excess base b) CH3 CH2 OH ------> CH3 CH2 Br -----------------> -------> CH3 CH2 NH 2 235 4. Synthesis, continued. O H2 CrO4 || c) ----------> C-H O SOCl2 CH3 CH2 NH 2 || COH ---------> O || CNHCH 2 CH3 d) HNO3 --------> H2 SO 4 e) HNO3 H2 -------> ----> H2 SO 4 Pt H2 NO 2 ----> Pt NH 2 NaNO 2 NH 2 ----------> HCl N2 + Cu2 O 2+ Cu H2 O OH f) Br2 -------> FeBr3 HNO3 H2 NaNO 2 Br --------> -----> ---------> Br H2 SO 4 Pt HCl N2 + CuCN Br 5.1 5.2 a) E b) B c) D This unknown is (CH3 )2 CHCH 2 NH 2 236 C N Name __________________________________________________ Eighth Drill Test (Sample) Organic Chemistry 2220 Answer All Questions 1) Draw: isobutylamine 3) Which of the following is the strongest base? NH 2 NH 2 NH 2 A. B. NO 2 OH B. C. CO2 H NH 2 D. CH3 Predict the product of each of the following reactions (if any). O || Pt a) CH2 CCH3 + NH 3 + H2 -----> b) N: + HCl -----> Br c) 6) D. Which of the following is insoluble in water and 5% aq. NaOH, but soluble in 5% aq. HCl? A. 5) NH 2 C. OCH 3 CH3 4) 2) Name: (CH3 )2 CHCHCH 2 CH3 | NH 2 NaN3 LiAlH4 --------> ----------> Propose a synthesis of each, from the given starting material and any other needed reagents. a) CH2 NH 2 from CH2 OH b) CH3 CH2 CH2 NH 2 from CH3 CH2 OH c) N C OCH 3 from OCH 3 237 Name __________________________________________________ Eighth Drill Test (Sample) Organic Chemistry 2220 Answer All Questions 1) Draw the structure of N-phenyl-2-heptanamine. 2) Name the compound shown. NH 2 3) Which of the following is MOST soluble in water? A. 4) N B. D. Complete the following reactions. a) N2 + Cl - H3 PO 2 → CuBr → b) c) (CH3 )2 CHC≡N → d) CH3 CH2 N3 LiAlH4 → f) (CH3 )2 CHCH 2 Br g) Br (CH3 )2 CHCH 2 NH 2 NaNO 2 → HCl e) 5) NH 2 C. NH 2 + N2 + → Cl - (CH3 )2 CHCH 2 NH 2 SO 3 H -----> Propose a synthesis of the following compound from the given starting material. O || from N Cl | H 238 Br