MATERIALS AND METHODS toxic congeners with their corresponding toxic equivalency fac-
advertisement

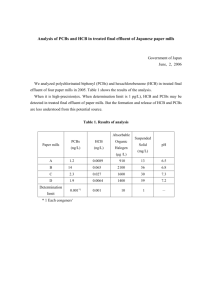
Report Kurunthachalam Kannan, Simonetta Corsolini, Takashi Imagawa, Silvano Focardi and John P. Giesy Polychlorinated -Naphthalenes, -Biphenyls, -Dibenzo-p-dioxins, -Dibenzofurans and p,p’-DDE in Bluefin Tuna, Swordfish, Cormorants and Barn Swallows from Italy Concentrations of p,p’-DDE, polychlorinated biphenyl congeners (PCBs), polychlorinated-dibenzo-p-dioxins (PCDDs), -dibenzofurans (PCDFs) and -naphthalenes (PCNs) were measured in bluefin tuna, swordfish, common cormorants, and barn swallows collected from Italy. Average concentrations of PCBs in livers of tuna, swordfish, cormorant, and swallows were 930, 745, 1420 and 1230 ng PCBs g–1, w.w. respectively. p,p’-DDE was found in tuna, swordfish, cormorant, and swallow livers at mean concentrations of 82, 135, 166 and 95 ng DDE g–1, w.w. respectively. PCNs were found in all the samples analyzed, although at concentrations less than those reported for biota from the Baltic Sea. PCBs, particularly, non-ortho coplanar PCBs accounted for 80–90% of the total TEQs in tuna and swordfish. Relative contribution of PCDDs/DFs to TEQs was greater in cormorants and swallows compared to that in fishes. PCDD/DF congeners accounted for up to 80 and 45% of the total TEQs in cormorants and swallows, respectively. INTRODUCTION Notable concentrations of polychlorinated biphenyls (PCBs) and DDT have been reported in tissues of dolphins found stranded along the Italian coast of the Mediterranean Sea (1-3). Concentrations of PCBs reported in the blubber of cetaceans from the Italian coast of the Mediterranean Sea were greater than a threshold value, to elicit physiological effects in aquatic mammals of 11 µg g–1, w.w. (4). While studies have measured concentrations of PCBs and DDTs in cetaceans, exposure to these compounds in other coastal species, particularly tuna, swordfish, and cormorants is less known. Monitoring of contaminants is essential in view of the fact that the tuna fishery is a major commercial industry in coastal Italy. Consumption of tuna or swordfish containing high concentrations of PCBs or other toxic compounds may have implications for human health. Apart from PCBs and DDTs, no studies have reported concentrations of polychlorinated-dibenzo-p-dioxins (PCDDs), -dibenzofurans (PCDFs) or naphthalenes (PCNs) in fishes from the Italian coast. Furthermore, bird species such as cormorants and swallows have been used as indicators to monitor aquatic pollution by chlorinated organics (5, 6). The objectives of this study were to determine concentrations and congener profiles of organochlorines that elicit dioxin-like toxicity, and their corresponding toxic equivalents in fishes and birds from the Italian coast of the Mediterranean Sea using high resolution gas chromatography–high resolution mass spectrometry. Tissues of bluefin tuna (Thunnus thynnus thynnus), swordfish (Xiphias gladius), and common cormorants (Phalacrocorax carbo) collected from the Italian coasts were analyzed for the presence of p,p’-DDE, PCBs, PCDDs, PCDFs and PCNs. Barn swallows (Hirundo rustica) collected from agricultural areas in Pianura Padana near Milan, northern Italy, were analyzed for the presence of the target organochlorines. 2,3,7,8-tetrachlorodibenzo-p-dioxin equivalents (TEQs) were calculated by multiplying the concentrations of Ambio Vol. 31 No. 3, May 2002 toxic congeners with their corresponding toxic equivalency factors (TEFs) derived from H4IIE in vitro bioassays (7–9). MATERIALS AND METHODS Samples of liver, muscle and fat from sexually mature (fork length > 110 cm) bluefin tuna were collected in Palizzi, southern coast of Italy, during October–November 1999. Total length and weight of tuna ranged from 149 to 171 cm and 46 to 61 kg, respectively. Liver and muscle were taken from mature swordfish, which were harpooned in Stretto di Messina in the Ionian— Tyrrhenian Seas during July 1999. Heavy ship traffic is a major source of pollution in the Stretto di Messina. Total length and weight of swordfish ranged from 107 to 190 cm and 15 to 83 kg, respectively. Tissues from several individuals were pooled to obtain representative samples. Livers of cormorants were collected from the birds that were originally sacrificed in 1997 by the Department of Sanitation, Division of Rearing and Zootechnical Resources. Cormorants were collected near Oristano (West coasts of Sardinia Island) in the vicinity of Cabras lagoon in the Sardinian Sea. It is a protected area and is less polluted. Barn swallows were collected during July–September 1995 at farms located in Pianura Padana near Milan. Pianura Padana is a large area in northern Italy characterized by intensive farming. The barn swallows collected at the nests were 1–2-years old and were between 165 and 118 g total weight. Tissues were wrapped in clean aluminum foil or whirlpac bags and stored frozen at –20°C until analysis. Sampling locations of fishes and birds are shown in Figure 1. The sampling locations, in general, are background sites with no known point sources of pollution. CHEMICAL ANALYSIS Chloronaphthalene (CN), chlorobiphenyl (CB) and 2,3,7,8-substituted congeners of PCDDs and PCDFs were analyzed followFigure 1. Map of Italy showing sampling locations of fishes and birds. © Royal Swedish Academy of Sciences 2002 http://www.ambio.kva.se 207 ing the method described elsewhere (10) with some modifica- PCN, PCDD and PCDF congeners through the analytical protions. Samples were homogenized with sodium sulfate and cedure were between 90 and 100%. Procedural blanks were Soxhlet extracted with methylene chloride and hexane (3:1, 400 analyzed throughout the whole analytical procedure to check for ml) for 16 hrs. The extract was rotary evaporated at 40°C and interferences. The detection limits of individual PCN and PCB an aliquot was used for the determination of fat content by congeners varied depending on the sample masses, response facgravimetry. The remaining extract was spiked with 13C-TCDD, tors and interferences. Generally, detection limits for individual 13 C-TCDF, 13C-OCDD and 13C-OCDF as internal standards and congeners were 1 to 75 pg g–1, on a wet weight basis. Quality interferences removed by fractionation with multilayer silica gel control criteria for positive identification of target compounds column. The multilayer silica gel column was prepared by pack- included signal to noise ratio above 3, isotope ratios of the 2 ing a glass column (20 mm i.d.) with a series of layers of silica monitored ions for each compound within 15% of the theoretigel in the following order: 2 g silica, 6 g 40% acidic-silica, 2 g cal chlorine values, and that the compound should elute at the silica and a thin layer of sodium sulfate at the top. The column same GC retention time as the standards. CN and CB congeners was cleaned with 150 ml of hexane prior to the transfer of sam- are represented by using their IUPAC numbers throughout this ple extracts. Samples were then eluted with 200 ml of hexane report (12, 13). and rotary evaporated to 5 ml. A portion of the aliquot was taken for the analysis of PCB congeners other than non-ortho coplanar RESULTS AND DISCUSSION PCBs. The remaining samples were passed through a glass column (10 mm i.d.) packed with 1 g silica gel impregnated car- Bluefin Tuna bon (Wako Pure Chemical Industries, Tokyo, Japan) for the sepa- Concentrations of total PCBs in livers of bluefin tuna ranged ration of ortho-substituted PCBs from PCNs and PCDDs/DFs. from 224 to 1660 ng g–1 (mean: 934; n = 3) on a wet weight The first fraction, which was eluted with 150 ml of hexane con- basis (w.w.) (Table 1). The 6-fold variation in the concentrations tained major PCB congeners, which interfere with the analysis of total PCBs in tuna livers was related to lipid content. Lipidof PCNs and PCDDs/DFs. The second fraction, which was eluted normalized concentrations of total PCBs in tuna livers were bewith 200 ml of toluene contained non-ortho substituted PCB con- tween 5670 and 14␣ 400 ng g–1. Mean concentrations of total geners—77, 126 and 169—, PCNs and PCDDs and PCDFs. PCBs in tuna muscle and fat were 280 (n = 2) and 817 ng g–1, PCB congeners were identified and quantified using a gas w.w., respectively. Concentrations of total PCBs in bluefin tuna chromatograph (Perkin Elmer series 600) equipped with 63Ni collected from the Ionian and Tyrrhenian Seas in the late 1970s electron capture detector (GC-ECD). A fused silica capillary col- ranged from 4.7 to 5000 ng g–1, w.w. (14). Similarly, muscle of umn coated with DB-5MS ((5%-phenyl)-methylpolysiloxane, 30 bluefin tuna collected in 1993 contained total PCB concentram x 0.25 mm i.d.; J&W Scientific, Folsom, CA, USA) having a tions in the range of 170 to 2200 ng g–1, w.w. (1). Concentrafilm thickness of 0.25 µm was used. PCB congeners were iden- tions of total PCBs in tuna muscle analyzed in this study were tified against a standard mixture containing 100 congeners of less than those reported earlier. However, it should be noted that known composition and content. Further details of PCB analy- tuna analyzed in this study were smaller (46 to 61 kg) than those sis are reported elsewhere (10, 11). Identification and quantifi- analyzed in earlier studies (70 to 400 kg) (1), which could have cation of individual PCN and PCDD/DF congeners were accom- contributed to the observed temporal differences in total PCB plished with a Hewlett Packard 6890 series high resolution gas concentrations. chromatograph (HRGC) coupled to a JEOL JMS-700 high resoHexachlorobiphenyl no. 153 (2,2',4,4',5,5'-HxCB) was the lution mass spectrometer (HRMS). PCN congeners, hepta- and most predominant congener found in tuna tissues (Table 2). This octa-chlorodibenzo-p-dioxins and furans and non-ortho coplanar congener accounted for, on average, 20% of the total PCB conPCBs were separated using a DB-1701 column. Tetra- through centrations in tuna tissues. In a sample of tuna liver, hexa-chlorodibenzo-p-dioxins and furans were separated by a heptachlorobiphenyl no. 180 (2,2',3,4,4',5,5'-HpCB) accounted capillary column coated with SP-2331. The column oven tem- for the greatest proportion of total PCB concentrations (Table perature was programmed from 80 to 160°C at a rate of 40°C 2). A few earlier studies have reported the occurrence of elevated min–1 and then to 170°C at 10°C min–1, to 250°C at 4°C min–1 proportions of congener 180 in livers and blubbers of striped and then to 296°C at 8°C min–1 with a final hold time of 10 min. dolphins from the Italian coast of the Tyrrhenian Sea (2, 3). CB Injector and transfer line temperatures were held at 260 and congener 180 accounts for approximately 10% of the total PCBs 250°C, respectively. Helium was used as the carrier gas. The by weight in Aroclor 1260 (15). This congener accounted for, mass spectrometer was operated at an electron impact (EI) en- on average, 10% of the total PCBs in the tissues of tuna. This ergy of 70 eV. CN and dioxin congeners were determined by selected ion monitoring (SIM) at the 2 most intensive ions of the molecular ion Table 1. Concentrations (wet weight) of p,p’-DDE (ng g–1), total PCBs (ng g–1) and total cluster. A mixture of Halowaxes PCNs (pg g–1) in fishes and birds from Italy. 1001, 1014, and 1051 containing all Species Tissue Fat (%) Collection date Location N p,p’-DDE PCBs PCNs the tri- through octa-chloronaphthaTunafish Liver 11.5 Oct–Nov 1999 Palizzi 1 75 1660 NA lenes was used as a standard for the Tunafish Liver 3.95 Oct–Nov 1999 Palizzi 1 61 224 53.8 quantification of PCNs. PCDD and Tunafish Liver 10 Oct–Nov 1999 Palizzi 5 110 920 NA Tunafish Muscle 5.3 Oct–Nov 1999 Palizzi 3 67 363 22.6 PCDF congeners were quantified Tunafish Muscle 2.92 Oct–Nov 1999 Palizzi 2 30 197 6.97 by comparing individually resolved Tunafish Fat 85 Oct–Nov 1999 Palizzi 3 135 817 552 Swordfish Liver 16.2 July 1999 Stretto Messina 4 135 745 62.9 peak areas to the corresponding Swordfish Muscle 8.78 July 1999 Stretto Messina 4 45 258 14.8 peak areas of the standards. RecovSwordfish Muscle 9.5 July 1999 Stretto Messina 6 69 399 14.6 13 Cormorant Liver 7.6 1997 Cabras lagoon 3 164 722 130 eries of C-labelled PCDDs/DFs, Cormorant Liver 7.76 1997 Cabras lagoon 1 190 2300 795 Cormorant Liver 4.55 1997 Cabras lagoon 1 144 1230 347 which elute in the second fraction Swallow Liver 4.5 1995 Milan 5 84 954 215 containing PCNs and non-ortho Swallow Liver 6.2 1995 Milan 4 105 1510 126 Swallow Muscle 4.7 1995 Milan 4 67 733 116 coplanar PCBs, were 77–95%. ReSwallow Muscle 5.1 1995 Milan 5 82 700 142 ported concentrations were not corN= number of samples pooled. rected for the recoveries of the inNA= not analyzed. ternal standard. Recoveries of PCB, 208 © Royal Swedish Academy of Sciences 2002 http://www.ambio.kva.se Ambio Vol. 31 No. 3, May 2002 Concentrations of 2,3,7,8-substituted congeners of PCDDs/ DFs were generally less than the limits of detection, which varied from 1 to 75 pg g–1, w.w., depending on the congeners and tissue (Table 3). PCDD/DF congeners that were detected in tuna tissues include, 2,3,4,7,8-PeCDF and 1,2,3,4,7,8-HxCDF, which were found in a sample of tuna fat at concentrations of 19–20 pg g–1, w.w. Previous studies have not reported concentrations of PCDDs/DFs in tuna from the Italian coast of the Mediterranean Sea. Nevertheless, concentrations of PCDDs/DFs in livers of dolphins from the Mediterranean Sea have been reported to range from 13 to 107 pg g–1, w.w. (19). suggests exposure of tuna to highly chlorinated PCB mixtures and/or selective retention of congener 180 in fish tissues. Congener 180 has been shown to be resistant to metabolism by marine animals (16). Overall, hexa- and hepta-chlorobiphenyl congeners 153, 138, 180, and 170, collectively accounted for 35 to 47% of the total PCB concentrations in tuna tissues. Mean concentrations of p,p’-DDE in liver, muscle and fat of tuna were 82, 49 and 135 ng g–1, w.w., respectively (Table 1). The observed concentrations of p,p’-DDE in the muscle of tuna were less than those reported for relatively larger size tuna collected in 1993 (56–270 ng g–1, w.w.) (1). Polychlorinated naphthalenes were found in all the analyzed tissues of bluefin tuna at concentrations ranging from 7 (muscle) to 550 pg g–1, w.w. (fat) (Table 1). The observed concentrations of PCNs were 3 to 5 orders of magnitude less than total PCB concentrations. Tuna fat contained the highest concentration of PCNs. In this tissue, 17 of the 75 PCN congeners were found. CN congeners 54 (1,2,3,6,7-PeCN), 56 (1,2,3,7,8-PeCN), 66/67 (1,2,3,4,6,7-/1,2,3,5,6,7-HxCNs), 61 (1,2,4,6,8-PeCN) and 52/60 (1,2,3,5,7-/1,2,4,6,7-PeCNs) were the predominant congeners collectively accounting for 84% of the total PCN concentrations in tuna fat. Concentrations of PCNs (1360 pg g–1, lipid weight (l.w.) in tuna livers were greater than those reported for cod livers (388 pg g–1, l.w.) collected in 1998 from the Arctic Ocean (17). However, the observed concentrations of PCNs in livers of tuna from Italy were less (1360 pg g–1, l.w.) than those found in the livers of cod from the Baltic Sea (9800 pg g–1, l.w.) (18). Swordfish Concentrations of total PCBs in pooled samples of liver and muscle of swordfish were 745 and 329 ng g–1, w.w., respectively (Table 1). Similarly, concentrations of p,p’-DDE in swordfish liver and muscle were 135 and 57 ng g–1, w.w., respectively. The observed concentrations of PCBs and p,p’-DDE in swordfish tissues were similar to those found in bluefin tuna. Similarly, profiles of PCB congeners in swordfish were similar to those observed for bluefin tuna. Hexachlorobiphenyl congener 153 accounted for the greatest proportion of total PCBs (20%) followed in order by 180 (12%) and 138 (7%) (Table 2). PCNs were also found in swordfish tissues at concentrations of 15 pg g–1, w.w., in muscle and 63 pg g–1, w.w., in liver. Tri-CN congeners 14/16 (1,2,4-/1,4,6-TrCNs) were the most abundant in swordfish. These congeners collectively accounted for 50% of the total PCN concentrations. PCDD/DF congeners were not found in swordfish Table 2. Concentrations of PCB congeners (w.w.) including non- (pg g–1), mono- (ng g–1) and di-ortho PCBs (ng g–1) in tuna, swordfish, cormorant and swallow tissues from Italy. Sample Tissue Tunafish Tunafish Tunafish Tunafish Tunafish Tunafish Swordfish Swordfish Swordfish Cormorant Cormorant Cormorant Swallow Swallow Swallow Swallow Non-ortho PCBs (pg g–1) N Fat (%) (pool) Liver Liver Liver Muscle Muscle Fat Liver Muscle Muscle Liver Liver Liver Liver Liver Muscle Muscle 1 1 5 3 2 3 4 4 6 3 1 1 5 4 4 5 11.5 3.95 10 5.3 2.92 85 16.2 8.78 9.5 7.6 7.76 4.55 45 6.2 4.7 5.1 Mono-ortho PCBs (ng g–1) Di-ortho PCBs (ng g-1) Total PCBs 77 126 169 105 118 156 137 138* 153 170 180 194 153 31.5 NA 71.7 23.8 518 82 48.8 490 44.5 98 NA 2940 8790 6330 2470 212 458 293 82.2 40.4 1010 134 60.8 68.6 212 428 NA 567 554 433 480 <23 <7.2 NA 9.2 5.7 62 <26 11 11 20 31 NA 62 39 53 66 25 3.27 7.2 5 2.7 9.5 8.4 2.5 4.5 19 44 29 26 15 14 19 87 7.4 32 14 6.2 16 18 5.3 11 50 13 5.4 6.8 7.5 2.8 5.1 32 <1 4.6 2.6 1.4 5.4 9.5 1.9 1.8 16 36 16 25 24 18 16 12 1.9 <1 2.9 <1 2.7 3.4 <1 <1 <1 1.5 <1 4.3 6.3 1.7 5 59 19 74 34 18 77 58 15 34 72 379 264 83 174 118 129 127 46 173 83 47 194 150 40 92 173 326 260 172 75 129 130 117 5.3 25 10 7.5 29 22 4.8 16 28 123 34 55 99 57 34 288 14 72 26 21 81 57 47 40 38 181 81 104 287 88 77 42 6.3 10 3 1.9 17 5.4 7.1 5.7 12 27 9.6 23 56 34 19 1660 224 920 363 197 817 745 258 399 722 2300 1230 954 1510 733 700 N= number of samples pooled. NA= not analyzed. *138 plus 158. Table 3. Concentrations of 2,3,7,8-substituted PCDDs/DFs (pg g–1, w.w.) in fishes and birds from Italy. PCDD congeners Species Tissue 2378 12378 Tuna Tuna Tuna Tuna Swordfish Swordfish Swordfish Cormorant Cormorant Cormorant Swallow Swallow Swallow Swallow Liver Muscle Muscle Fat Muscle Muscle Liver Liver Liver Liver Liver Liver Muscle Muscle <2 <1 <3 < 40 <1 <3 < 10 < 15 <3 < 10 < 10 < 10 1.41 1.81 <2 <1 <1 < 20 <4 <2 < 10 < 75 <5 5.56 5.77 < 10 2.62 2.36 PCDF congeners 123478 123678 123789 1234678 OCDD 2378 < 10 <2 <3 < 15 <6 < 10 < 20 < 10 <8 <3 < 0.01 < 15 <3 <3 <6 <2 <3 < 18 <5 <6 < 20 <4 <8 <3 < 12 <2 13.1 <3 < 10 <3 <4 < 25 <6 < 10 < 20 < 10 <8 5.07 12.6 9.37 <3 5.7 < 10 <2 <3 < 25 <2 < 10 < 20 < 10 <8 <3 < 13 < 15 <3 <3 <5 <3 <3 < 10 < 10 < 10 < 30 <5 < 30 < 10 < 50 <6 <8 < 10 < 50 <2 <2 <7 <4 <4 < 30 <2 < 10 <6 < 25 <3 <6 <5 12378 23478 123478 123678 123789 234678 1234678 1234789 OCDF NA <2 <5 < 10 <4 <6 < 20 <2 <3 <2 9.11 <1 4.48 <5 NA <2 <6 19 <4 <6 < 20 < 10 <3 7.7 < 12 <2 10.4 15 NA 3.21 <2 19.8 <5 <5 < 10 <4 < 10 <5 < 15 <6 <4 <4 NA < 1.5 <2 <5 <4 <5 < 60 6.36 < 10 <5 < 15 <6 <3 <4 NA <2 < 10 <5 <4 <5 < 10 <4 < 10 <5 < 15 <6 <4 <3 NA <3 <2 < 10 <4 <5 < 10 <4 < 10 <6 < 15 <6 <4 <3 <6 <4 <4 < 12 < 12 < 10 < 25 <6 < 30 < 14 < 40 < 20 <4 < 10 <6 <3 <4 < 25 < 14 <8 < 25 <8 < 30 < 12 < 40 < 15 <4 < 20 < 10 <2 <3 <5 <3 <6 < 20 <4 < 16 <8 < 25 <3 <6 <6 NA = not analyzed. Ambio Vol. 31 No. 3, May 2002 © Royal Swedish Academy of Sciences 2002 http://www.ambio.kva.se 209 above the limits of detection (Table 3). To our knowledge, reports of organochlorine concentrations in swordfish are not available for comparison. This study provides baseline measurements of organochlorines in swordfish for the first time. Cormorants Concentrations of total PCBs in livers of cormorants ranged between 722 and 2300 ng g–1, w.w. (Table 1). The measured concentrations of PCBs in cormorant livers were comparable to those reported in Germany (2400 ng g–1, w.w.) during 1985/86 (6). However, concentrations of total PCBs in cormorant livers from Italy were less than those reported from Lake Biwa, Japan (6800 ng g–1, w.w.) (20). Hexa-CB congener 153 accounted for 20% of the total PCB concentrations followed by congeners 138 (16%) and 180 (7%). Concentrations of p,p’-DDE in cormorant livers ranged from 144 to 190 ng g–1, w.w. (mean: 166 ng g–1, w.w.). Concentrations of PCNs in cormorant livers ranged from 130 to 795 pg g–1, w.w. (mean: 424 pg g–1, w.w.). Mean PCN concentrations in cormorant livers were 7-times greater than those found in the livers of tuna and swordfish. Estimated concentration of total PCNs in livers of cormorants collected from the Baltic Sea was 14␣ 000 pg g–1, w.w. (21). Concentrations of total PCNs measured in cormorant livers from Cabras lagoon in the Sardinian Sea were approximately 30-fold less than those reported in Baltic Sea cormorants. CN congeners 66/67, 42 (1,3,5,7-TeCN), 61 and 52/60 accounted for 90% of the total PCN concentrations in cormorant livers. As mentioned earlier, CN congeners 66/67, 61 and 52/60 were also abundant in bluefin tuna livers. These congeners have been shown to be strongly bioaccumulative in wildlife (21). PCDD congeners, 1,2,3,7,8PeCDD and 1,2,3,6,7,8-HxCDD and PCDF congeners 2,3,4,7,8PeCDF and 1,2,3,6,7,8-HxCDF were detected in livers of certain individual cormorants. Barn Swallows Swallows, particularly, tree swallows, are being widely used as indicators of local contamination because they feed near their nest of emergent aquatic insects (5, 22, 23). Barn swallows are similar to tree swallows in their feeding habits and migratory behavior. Barn swallows collected from agricultural areas near Milan contained remarkable concentrations of PCBs. Mean concentrations of PCBs in liver and muscle of barn swallows were 1230 and 716 ng g–1, w.w., respectively (n = 2). Profiles of relative concentrations of PCB congeners in swallows were similar Table 4. Estimated concentrations (pg g–1, w.w.) of total TEQs* in fishes and birds from Italy. Species Tissue Tunafish Tunafish Tunafish Tunafish Tunafish Tunafish Swordfish Swordfish Swordfish Cormorant Cormorant Cormorant Swallow Swallow Swallow Swallow Liver Liver Liver Muscle Muscle Fat Liver Muscle Muscle Liver Liver Liver Liver Liver Muscle Muscle Toxic Equivalents To our knowledge, no previous studies have reported on the occurrence of PCNs in biota from the Mediterranean Sea. The results of this study permitted the analysis of the relative contribution of PCNs to total TEQs (sum of TEQs of coplanar PCBs, PCDDs/DFs and PCNs). All PCN congeners are planar and several of them exhibit AhR mediated cytochrome P450 induction, analogous to TCDD (8, 9). Toxic equivalency factors (TEFs) or relative potencies reported for several PCN congeners based on in vitro bioassays using H4IIE rat hepatoma cells were used for calculating TEQs (8, 9, 24). For comparison of TEQs contributed by PCNs, PCBs, PCDDs, and PCDFs, it would be appropriate to use TEFs derived using similar bioassays. For comparison of TEQs contributed by PCNs (PCN-TEQs) with those contributed by PCBs (PCB-TEQs), PCDDs (PCDD-TEQs) and PCDFs (PCDF-TEQs), concentrations of 2,3,7,8-substituted congeners of PCDD and PCDF and non- and mono-ortho-substituted PCBs were multiplied by their corresponding TEFs derived from H4IIE bioassays (7). Di-ortho PCBs were not included in PCB-TEQ estimation. It should be emphasized that the estimation of TEQs is for the examination of relative contribution of PCNs, PCBs and PCDDs/DFs to total TEQ concentrations and not for risk assessment, which would require species-specific TEFs. Calculated concentrations of total TEQs (sum of TEQs of PCBs, PCNs and PCDDs/DFs) in the tissues of tuna ranged from 0.99 to 28 pg g–1, w.w. (Table 4). Total TEQs in swordfish tissues were in the range of 1.47 to 3.55 pg g–1, w.w.. Total TEQs in swallow tissues were greater (14.1–19.7 pg g–1, w.w.) than those estimated for cormorant livers (5.75–11.9 pg g–1, w.w.). Non- and monoortho PCBs were the major contributors to total TEQ concentrations in fishes (Fig. 2). In particular, non-ortho coplanar PCB Figure 2. Relative contribution (%) of PCBs, PCNs and PCDDs/DFs to total TEQ concentrations in tissues of tuna, swordfish, cormorants and swallows. PCBs PCNs Total TEQs 6.65 10.1 6.76 2.07 0.99 28.5 3.55 1.47 1.66 9.54 11.9 5.75 18.7 14.1 19.7 18.8 * Sum of TEQs of PCBs, PCNs and PCDDs/DFs. * For those congeners below the limits of detection, detection limits were used to calculate the TEQs. 210 to those observed for cormorants with PCB congener 153 accounting for the greatest proportion of total PCB concentrations followed in decreasing order by CB congener 138 ≥ congener 180. Concentrations of p,p’-DDE in the liver and muscle of swallows were 95 and 75 ng g–1, w.w., respectively (Table 1). PCNs were also found in swallows at concentrations ranging from 116 to 215 pg g–1, w.w.. PCN congeners 52/60 and 66/67 were the major congeners in swallow tissues. Among PCDDs/DFs, 2,3,7,8-TCDD, 1,2,3,7,8-PeCDD, 1,2,3,6,7,8-HxCDD, 2,3,7,8TeCDF, 1,2,3,7,8-PeCDF and 2,3,4,7,8-PeCDF were detected in barn swallows (Table 3). Tuna liver PCDDs/DFs Tuna muscle Tuna fat Swordfish liver Swordfish muscle Cormorant liver Swallow liver Swallow muscle 0 20 © Royal Swedish Academy of Sciences 2002 http://www.ambio.kva.se 40 60 80 Contribution (%) 100 Ambio Vol. 31 No. 3, May 2002 congener 126 accounted for 80 to 90% of the total TEQs in fishes. The contribution of this congener to total TEQs was relatively less in birds accounting for 52 to 76% of the total TEQs. In the livers of cormorants and muscle tissues of swallows, PCDDs/DFs contributed up to 80 and 45%, respectively, of the total TEQs. PCDF congeners 2,3,7,8-TeCDF and 2,3,4,7,8PeCDF accounted for 60% of the PCDD/DF-TEQs in swallow muscle. In cormorant livers, 1,2,3,7,8-PeCDD, 2,3,4,7,8-PeCDF and 1,2,3,6,7,8-HxCDF accounted for 95% of the PCDD/DFTEQs. The contribution of PCNs to total TEQs in fishes and birds from the Mediterranean Sea was less than 2%. The greatest PCN-TEQ concentration of 0.15 pg g–1 was 1.3% of the total TEQs calculated in the liver of a cormorant. In fishes, PCNs accounted for up to 0.3% of the total TEQs. This is less than what has been reported to occur in earlier studies in which PCNs contributed up to 50% of the total TEQs in fishes from the Detroit River (25). PCN congeners 66/67 accounted for 0.3 to 13% of the total TEQs in fishes from the Baltic Sea (26). Relatively less contribution of PCNs to TEQs in Mediterranean fishes and cormorants suggests less exposure in off-shore species analyzed in this study. Although this calculation gives the relative importance of each organochlorine group to the total contribution of dioxin-like effects, caution should be exercised in assessing the risks since non-detects were assigned a concentration of zero for estimating the TEQs. The limits of detection of certain PCDD/ DF congeners were high. References and Notes 1. Corsolini, S., Focardi, S., Kannan, K., Borrell, A., Tanabe, S. and Tatsukawa, R. 1995. Congener profile and toxicity assessment of polychlorinated biphenyls in dolphins, shark and tuna collected from Italian coastal waters. Mar. Environ. Res. 40, 33–53. 2. Marsili, L. and Focardi, S. 1997. Chlorinated hydrocarbon (HCB, DDTs and PCBs) levels in cetaceans stranded along the Italian coasts: An overview. Environ. Monit. Assess. 45, 129–180. 3. Reich, S., Jimenez, B., Marsili, L., Hernández, L.M., Schurig, V. and González, M.J. 1999. Congener specific determination and enantiomeric ratios of chiral polychlorinated biphenyls in striped dolphins (Stenella coeruleoalba) from the Mediterranean Sea. Environ. Sci. Technol. 33, 1787–1793. 4. Kannan, K., Blankenship, A.L., Jones, P.D. and Giesy, J.P. 2000. Toxicity reference values for the toxic effects of polychlorinated biphenyls to aquatic mammals. Hum. Ecol. Risk Assess. 6, 181–201. 5. Ankley, G.T., Niemi, G.J., Lodge, K.B., Harris, H.J., Beaver, D.L., Tillitt, D.E., Schwartz, T.R., Giesy, J.P., Jones, P.D. and Hagley, C. 1993. Uptake of planar polychlorinated biphenyls and 2,3,7,8-substituted polychlorinated dibenzofurans and dibenzo-p-dioxins by birds nesting in the lower Fox River and Green Bay, Wisconsin, USA. Arch. Environ. Contam. Toxicol. 24, 332–344. 6. Scharenberg, W. 1991. Cormorants (Phalacrocorax carbo sinensis) as bioindicators of polychlorinated biphenyls. Arch. Environ. Contam. Toxicol. 21, 536–540. 7. Giesy, J.P., Jude, D.J., Tillitt, D.E., Gale, R.W., Meadows, J.C., Zajieck, J.L., Peterman, P.H., Verbrugge, D.A., Sanderson, J.T., Schwartz, T.R. and Tuchman, M.L. 1997. Polychlorinated dibenzo-p-dioxins, dibenzofurans, biphenyls and 2,3,7,8-tetrachlorodibenzo-p-dioxin equivalents in fishes from Saginaw Bay, Michigan. Environ. Toxicol. Chem. 16, 713–724. 8. Villeneuve, D.L., Kannan, K., Khim, J.S., Falandysz, J., Blankenship, A.L. and Giesy, J.P. 2000. Relative potencies of individual polychlorinated naphthalenes to induce dioxin-like responses in fish and mammalian in vitro bioassays. Arch. Environ. Contam. Toxicol. 39, 273–281. 9. Blankenship, A., Kannan, K., Villalobos, S., Villeneuve, D.L., Falandysz, J., Imagawa, T., Jakobsson, E. and Giesy, J.P. 2000. Relative potencies of individual polychlorinated naphthalenes and Halowax mixtures to induce Ah receptor-mediated responses. Environ. Sci.Technol. 34, 3153–3158. 10. Kannan, K., Hilscherova, K., Imagawa, T., Yamashita, N., Williams, L.L. and Giesy, J.P. 2001. Polychlorinated naphthalenes, -biphenyls, -dibenzo-p-dioxins, and -dibenzofurans in double crested cormorants and herring gulls from Michigan waters of the Great Lakes. Environ. Sci. Technol. 35, 441–447. 11. Khim, J.S., Villeneuve, D.L., Kannan, K., Hu, W.Y., Giesy, J.P., Kang, S.G., Song, K.J. and Koh, C.H. 2000. Instrumental and bioanalytical measures of persistent organochlorines in blue mussel (Mytilus edulis) from Korean coastal waters. Arch. Environ. Contam. Toxicol. 39, 360–368. 12. Ballschmiter, K. and Zell, M. 1980. Analysis of polychlorinated biphenyls (PCB) by glass capillary gas chromatography. Fres. Z. Anal. Chem. 302, 20–31. 13. Kannan, K., Imagawa, T., Blankenship, A.L. and Giesy, J.P. 1998. Isomer-specific analysis and toxic evaluation of polychlorinated naphthalenes in soil, sediment and biota collected near the site of a former chlor-alkali plant. Environ. Sci. Technol. 32, 2507– 2514. 14. Impellizzeri, G., Tringali, C., Chillemi, R. and Piattelli, M. 1982. Observations on the levels of DDTs and PCBs in the central Mediterranean. Sci. Total Environ. 25, 169– 179. 15. Frame, G.M., Wagner, R.E., Carnahan, J.C., Brown, J.F. Jr., May, R.J., Smullen, L.A. and Bedard, D.L. 1996. Comprehensive, quantitative, congener-specific analysis of eight Aroclors and complete PCB congener assignments on DB-1 capillary GC columns. Chemosphere 33, 603–623. 16. Boon, J.P., Arnheim, E.V., Jansen, S., Kannan, N., Petrick, G., Schulz, D., Duinker, J.C., Reijnders, P.J.H. and Goksøyr, A. 1992. The toxicokinetics of PCBs in marine mammals with special reference to possible interactions of individual congeners with the cytochrome P450-dependent monoxygenase system: an overview. In: Peristent Pollutants in Marine Ecosystems. Walker, C.H. and Livingstone, D.R. (eds). Pergamon Press, New York, pp. 119–159. 17. Sinkkonen, S. and Paasivirta, J. 2000. Polychlorinated organic compounds in the Arctic cod liver: trends and profiles. Chemosphere 40, 619–626. 18. Järnberg, U., Asplund, L., de Wit, C., Egebäck, A-L., Wideqvist, U. and Jakobsson, E. 1997. Distribution of polychlorinated naphthalene congeners in environmental and source-related samples. Arch. Environ. Contam. Toxicol. 32, 232–245. 19. Jimenez, B., Gonzalez, M.J., Jimenez, O., Reich, S., Eljarrat, E. and Rivera, J. 2000. Evaluation of 2,3,7,8 specific congener and toxic potency of persistent polychlorinated dibenzo-p-dioxins and polychlorinated dibenzofurans in cetaceans from the Mediterranean Sea, Italy. Environ. Sci. Technol. 34, 756–763. 20. Guruge, K.S., Tanabe, S., Fukuda, M., Yamagishi, S. and Tatsukawa, R. 1997. Accumulation pattern of persistent organochlorine residues in common cormorants (Phalacrocorax carbo) from Japan. Mar. Pollut. Bull. 34, 186–193. 21. Falandysz, J. 1998. Polychlorinated naphthalenes: an environmental update. Environ. Pollut. 101, 77–90. 22. Froese, K.L., Verbrugge, D.A., Ankley, G.T., Niemi, G.J., Larsen, C.P. and Giesy, J.P. 1998. Bioaccumulation of polychlorinated biphenyls from sediments to aquatic insects and tree swallow eggs and nestlings in Saginaw Bay, Michigan, USA. Environ. Toxicol. Chem. 17, 484–492. Ambio Vol. 31 No. 3, May 2002 23. Harris, M.L. and Elliott, J.E. 2000. Reproductive success and chlorinated hydrocarbon contamination in tree swallows (Tachycineta bicolor) nesting along rivers receiving pulp and paper mill effluent discharges. Environ. Pollut. 110, 307–320. 24. Hanberg, A., Ståhlberg, M., Georgellis, A., de Wit, C. and Ahlborg, U.G. 1991. Swedish dioxin survey: evaluation of H4IIE bioassay for screening environmental samples for dioxin-like enzyme induction. Pharmacol. Toxicol. 69, 442–449. 25. Kannan, K., Yamashita, N., Imagawa, T., Decoen, W., Khim, J.S., Day, R.M., Summer, C.L. and Giesy, J.P. 2000. Polychlorinated naphthalenes and polychlorinated biphenyls in fishes from Michigan waters including the Great Lakes. Environ. Sci. Technol. 34, 566–572. 26. Järnberg, U., Asplund, L., de Wit, C., Grafström, A-K., Haglund, P., Jansson, B., Lexén, K., Strandell, M., Olsson, M. and Jonsson, B. 1993. Polychlorinated biphenyls and polychlorinated naphthalenes in Swedish sediment and biota: Levels, patterns and time trends. Environ. Sci. Technol. 27, 1364–1374. 27. We thank Istituto Centrale per la Ricerca Scientifica e Tecnologica Applicata al Mare (ICRAM) for providing samples of tuna fish and sword fish; Prof. Claudio Leonzio (University of Siena) and Istituto Zooprofilattico Sperimentale per la Sardegna, Dipartimento Igiene degli Allevamenti e delle Produzioni Zootecniche for providing tissues of cormorants. We thank Prof. Nicola Saino, University of Milano for providing tissues of barn swallows. 28. First submitted 19 March 2001. Accepted for publication after revision August 2001. Kurunthachalam Kannan, PhD, is a Visiting Associate Professor at National Food Safety and Toxicology Center in Michigan State University, East Lansing, Michigan, USA. His address: National Food Safety and Toxicology Center, Department of Zoology, Institute for Environmental Toxicology, Michigan State University, East Lansing, MI 48824, USA. E-mail: kuruntha@msu.edu Simonetta Corsolini, PhD, is a research scientist at Department of Environmental Biology, University of Siena, Italy. Her address: Dipartimento di Scienze Ambientali, Università di Siena, I-53100 Siena, Italy. E-mail: corsolini@unisi.it Takashi Imagawa, PhD, is a senior research scientist at National Institute for Resources and Environment, Tsukuba, Japan. His address: National Institute for Resources and Environment, 16-3 Onogawa, Tsukuba, Ibaraki 305, Japan. Silvano Focardi, PhD, is a professor at Department of Environmental Biology, University of Siena, Italy. His address: Dipartimento di Scienze Ambientali, Università di Siena, I-53100 Siena, Italy. John P. Giesy, PhD, is a distinguished professor of Department of Zoology, National Food Safety and Toxicology Center and Institute for Environmental Toxicology at Michigan State University. His address: National Food Safety and Toxicology Center, Department of Zoology, Institute for Environmental Toxicology, Michigan State University, East Lansing, MI 48824, USA. E-mail: jgiesy@aol.com The research interests of the authors include understanding of environmental distribution, dynamics, fate and effects of trace organic pollutants. © Royal Swedish Academy of Sciences 2002 http://www.ambio.kva.se 211