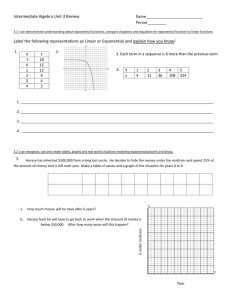
A Comparison of Extensively and Intensively Raised Early-Life Stage Walleye... Minnows Samuel D. Hempel*
advertisement

1 2 3 A Comparison of Extensively and Intensively Raised Early-Life Stage Walleye fed 4 Minnows 5 6 7 8 Samuel D. Hempel*,1 9 10 11 Northern Aquaculture Demonstration Facility 12 University of Wisconsin-Stevens Point 13 2100 Main Street 14 Stevens Point, WI 54481-3897 15 16 * Corresponding author: Samuel.D.Hempel@uwsp.edu 17 1 18 167 TNR Building, 800 Reserve Street, Stevens Point, WI 54481-3897 Present address: Biology Department, University of Wisconsin-Stevens Point, 19 Abstract 20 Two commonly used techniques for propagating Walleye are intensive and 21 extensive rearing treatments; fish reared using both methods can be fed commercialized 22 feed or natural feed or a combination of the two. The two treatments have differing 23 control levels with their own advantages. The objective of this research was to compare 24 growth of Walleye reared using intensive and extensive methods. Walleye fry were 25 reared in either an intensive system (commercial feed) or extensive system (zooplankton) 26 for 54 to 70 days. At that time juvenile Walleye were brought indoors, separated by 27 treatment, and evenly distributed among three flow-through tanks at a density of 20 fish 28 per tank. All fish were then fed fathead minnows for the duration of the experiment. Total 29 length (mm) and weight (g) of all fish were recorded every four days. Mean total length 30 and mean weight gained and specific growth rates were calculated and compared between 31 the treatments. The intensive treatment exhibited significantly higher sizes at the 32 beginning and end of the experiment, along with total length and total weight gained over 33 the course of the experiment. However, specific growth rates were higher in the extensive 34 treatment. The results suggest that the extensive treatment was deficient in essential 35 nutrients and when provided ample feed, growth rates will increase. 36 37 Introduction Walleye Sander vitreus are a cool-water species native to a substantial portion of 38 North America and are among the most sought after fishes by recreational anglers 39 (NEED CITATION). Due to their popularity, Walleye have been stocked far outside 40 their original distribution (Summerfelt 2000). They are a large predatory fish that feed 41 exclusively on living organisms, such as zooplankton, insects, and other fish 42 (Kapuscinski 1986), and exhibit certain characteristics (including intractable feeding 43 habits and a tendency toward cannibalism when young), walleye are very difficult to 44 culture (Kapuscinski 1986). The quality and taste is desired by fishermen and consumers 45 throughout the nation. They are described as one of the “best eating of all freshwater 46 fishes” (Carmichael et al. 1991), and “one of the most delicious of fresh-water fishes” 47 (Cameron and Jones 1983). Their popularity as a food fish continues to grow for and it is 48 one of the most targeted species in Wisconsin for recreational fishermen. 49 Walleye has been recognized as a species with substantial aquaculture potential in 50 the National Aquaculture Plan (Joint Subcommitte on Aquaculture 1983). The Wisconsin 51 Walleye Initiative, started in May 2013, has provided funding to produce large fingerling 52 walleye in aquaculture systems for Wisconsin waters. Public hatcheries have spawned, 53 incubated, and hatched walleye for more than 100 years (Summerfelt 2000). 54 Two commonly used techniques for propagating walleye are intensive and 55 extensive rearing treatments. Intensively reared walleye are hatched and raised in a 56 controlled indoor environment, or tank, that has less vulnerability to biohazards and 57 diseases, and permits-control of environmental factors such as temperature, light, 58 turbidity, and effluents. Intensive culture has many factors in its favor: It is not subjected 59 to variable environmental conditions, so cultural conditions are fairly stable, temperature 60 can be controlled to lengthen growth and the growing season lengthened, nuisance 61 aquatic organisms are eliminated, and the quality and quantity of the feed is controlled 62 (Summerfelt 1996). Extensively reared walleye are hatched indoors and transferred to 63 outdoor ponds with less control over their environmental factors. Variability in water 64 quality and uncontrollable parameters, such as the weather, cause problems when trying 65 to maintain adequate zooplankton populations (Summerfelt 1996). Feed is naturally 66 available in the ponds (e.g., zooplankton and macroinvertebrates) but the fish must search 67 out the prey to eat. When zooplankton populations are lacking, fish may starve (McIntyre 68 et al. 1987). 69 Walleye are predators and feed primarily on other fishes and feeding them 70 commercial pellets in a cultured system can be difficult. Two of the primary techniques 71 used to feed walleye in cultured settings are commercialized feed or natural feed such as 72 minnows. Walleyes start to feed on fish when they reach 30-50 mm total length (Smith 73 and Pycha 1960; Forney 1966; Bulkley et al. 1976). More specifically, fathead minnows 74 Pimephales promelas, because they are highly cultured as baitfish and are relatively easy 75 to propagate, making them economically practical. Development of formulated feeds and 76 modified rearing techniques for the intensive production of walleyes would bring several 77 advantages over the use of live feed: labor costs associated with the collection or culture 78 of live feeds would be eliminated; and introductions of disease and contaminants would 79 be much less likely (Barrows et al. 1988). Commercialized or formulated feed (pellets) 80 can come from all over the world and in comparison, commercialized feeds are cheaper 81 because the resources are easier to obtain, packaging is simplified, and transportation is 82 less complicated. 83 Knowledge of the two feeding techniques varies between treatments. Strategies 84 consist of using either commercialized or natural feed for the entire rearing process, or a 85 mix of both techniques, depending on what works best for a given producers’ situation. 86 Feeding a predator something natural (e.g. minnows) can have setbacks, reports on 87 converting (habituating) pond-reared fingerlings to commercial feeds (Cheshire and 88 Steele 1972), confirm that walleye can be domesticated on commercial feed to reduce or 89 eliminate setbacks such as late arrival of minnows or gape limitations of Walleye. 90 91 The objective of this study was to determine if intensively reared early life-stage walleye exhibited faster growth than extensively reared walleye fed minnows. 92 93 94 Methods Experimental Design and Study Environments Eyed-eggs were obtained from the Genoa National Fish Hatchery on April 29th, 95 2014 (Mississippi River Strain) and hatched on May 5th, 2014. Three days after hatch, fry 96 were transferred to a 240 L fiberglass tank and given a commercially available feed. They 97 remained on commercial feed until the start of the experiment but were transferred to 98 1000 L tanks on May 25th, 2014. These fish will be referred to as the intensive treatment. 99 Eyed-eggs were also obtained from Hayward Bait (Upper Chippewa River Strain) and 100 hatched on May 21th, 2014. These eggs were collected by the Wisconsin Department of 101 Natural Resources Governor Thompson Fish Hatchery. Three days after hatch, 80,000 fry 102 were transferred to a 0.16 ha (0.4 acre), clay-lined pond and were allowed to feed on 103 zooplankton and aquatic invertebrates until July 14th, 2014, the start of the experiment. 104 These fish will be referred to as the extensive treatment. 105 On July 14th, 2014(day 1 of the experiment), 60 intensive age-0 walleye were 106 randomly selected and transferred from a 1000 L holding tank into three-240L round 107 fiberglass tanks at a density of 20 fish per tank (Figure 1). At the same time, 60 extensive 108 age-0 walleye were randomly selected and transferred indoors into three-240 L round 109 fiberglass tanks at a density of 20 fish per tank. Weight (g), and total length (TL; mm), 110 was recorded for all individual fish. 111 All tanks were on a flow-through system with flow rates at 4L/min. In-flow water 112 was degassed, aerated, and held at a temperature of 20°C. Low light conditions were 113 accomplished by enclosing the experimental area under black tarps and overhead lights at 114 a low setting. Dissolved oxygen, temperature, and pH were recorded daily. Turbidity of 115 the water was zero. 116 Sampling Design 117 Total fish biomass (g) was calculated for each tank and 20% of total biomass was 118 given as feed in fathead minnows the next day. Fathead minnows were purchased as feed 119 for both intensive and extensive treatments. They were kept in a separate holding tank 120 away from the experimental tanks. Once tempered to approximately 20°C, minnows were 121 weighed to 20% total fish biomass per tank and added to the tanks in the afternoon. 122 Feeding activities of walleye were recorded for observational data. Left over minnows 123 were removed every morning and wet weight of the total minnow biomass left in the 124 tanks were recorded daily. 125 Every four days Walleye were sedated with MS-222 (Tricaine Methanesulfonate) 126 and removed from the tanks for cleaning. At this time we measured and recorded total 127 length and wet weight (g) for all fish. Mortalities were measured, recorded, and removed. 128 Total fish biomass (g) was calculated for each tank after each sampling day and 20% of 129 that was given as feed. The experiment lasted for six weeks and all fish were sampled for 130 final measurements (i.e. weight and length). 131 Statistical Analysis 132 133 134 Weight gain, length gain, and specific growth rate (SGR) of weight and length were calculated using the following formulas Weight Gain = wt2-wt1 135 Length Gain = TL2-TL1 136 SGR = [loge(xf) – loge(xi)]/ET ×100 137 where, wt1 = mean weight (g) at initial time point, wt2 = mean weight (g) at the final time 138 point, TL1 = total length (mm) at initial time point, TL2 = total length (mm) at final time 139 point, ET = elapsed time (d) between sample days, xi = initial weight or length, and xf = 140 final weight or length. 141 A two sample t-test: assuming unequal variances was used to determine if length and 142 weight differed between treatments at the beginning of the experiment. Analysis of 143 covariance (ANCOVA) was used to determine if growth (weight and length gained) 144 145 differed between treatments over time (∝= 0.05). Linear regressions were used to 146 level. Average weight and total length were plotted against days post hatch. determine if specific growth rates differed between treatments at the 95% confidence 147 148 149 Results Mean Total Length Comparisons Mean total length was significantly different between the two treatments at the 150 beginning of the experiment and there was no overlap of the upper and lower 95% 151 confidence intervals (t Stat=34.753, df=80) (Table 1). Standardized data showed Walleye 152 in the intensive treatment grew at a rate of 1.30 mm, while Walleye from the extensive 153 treatment grew at a rate of 0.86 mm (Figure 2). Mean total length gain was significantly 154 faster in the intensive treatment than the extensive treatment (F=8.659, df=5, P=0.0604). 155 The intensive treatment grew at a rate of -0.09 mm/d, while Walleye from the extensive 156 treatment grew at a rate of 0.15 mm/d (Figure 3). When the data from sample day 86 was 157 removed, the intensive treatment grew at a rate of 0.03 mm/d, while Walleye from the 158 extensive treatment grew at a rate of 0.31 mm/d (Figure 4). Mean total length and mean 159 total length gain exhibited faster growth for the intensive treatment than the extensive 160 treatment. 161 Specific growth rate increased significantly faster in the extensive treatment than 162 the intensive treatment. The extensive treatment had a slope that grew at a rate of 0.04 163 mm/mm/d, while Walleye from the intensive treatment slope grew at a rate of -0.03 164 mm/mm/d (Figure 5). 165 Mean Weight Comparisons 166 Mean weight was significantly different between the two treatments at the 167 beginning of the experiment and there was no overlap of the upper and lower 95% 168 confidence intervals (t Stat=24.653, df=61). The intensive treatment slope grew at a rate 169 of 0.3987 g which was over three times greater than the extensive rate of 0.1325 g 170 (Figure 6). Mean weight gain was significantly faster in the intensive treatment than the 171 extensive treatment (F=15.9246, df=7, P=0.0104). The intensive treatment grew at a rate 172 of 0.0954 g/d, while Walleye from the extensive treatment grew at a rate of 0.0388 g/d 173 (Figure 7). Mean weight and mean weight gain exhibited faster growth for the intensive 174 treatment than the extensive treatment. 175 Specific growth rate increased significantly faster in the extensive treatment than 176 the intensive treatment. The extensive treatment grew at a rate of 0.125 g/g/d, while 177 Walleye from the intensive treatment grew at a rate of 0.1027 g/g/d (Figure 8). 178 179 Discussion We found that, on average, Walleye from the extensive treatment exhibited higher 180 specific growth than Walleye from the intensive treatment. A similar relationship has 181 been seen between intensive and wild populations as the growth rate of intensively 182 cultured walleyes is usually less than that of fish in the wild (Malison et al. 1990). 183 Specific growth rate of both length and weight of the extensive treatment increased at a 184 higher rate than the intensive treatment. However, total weight gained and total length 185 gained were both higher for the intensive treatment compared to the extensive treatment. 186 Total weight and length gained comparisons may not be appropriate because the two 187 treatments differed significantly in both length and weight at the beginning of the 188 experiment; the intensive treatment was overall larger in size than the extensive treatment 189 and remained higher throughout the experiment. In intensive culture, growth rates can be 190 increased or slowed by temperature manipulations (Summerfelt 2000,) which could have 191 contributed to the overall larger size of the intensive treatment throughout the experiment 192 because they were reared at higher temperatures before the experiment began. 193 Two different strains were used in the experiment and were derived from two 194 different broodstocks. One was from Genoa National Fish Hatchery and consisted of the 195 Mississippi strain (Intensive Treatment), while the other broodstock were collected by 196 Wisconsin Department of Natural Resources Governor Thompson Fish Hatchery and 197 consisted of the Hayward Bait strain (Extensive Treatment). Hatch dates differed between 198 the two strains and the data was standardized to make accurate comparisons between the 199 two treatments. Three replicates helped to eliminate error and determine significance. 200 Our SGR results suggest that the extensive treatment outperformed the intensive 201 treatment. A factor that could have influenced this was the extensive treatment moving 202 from a less nutrient rich pond to a higher nutrient environment of minnows being 203 provided in limited space. The more nutrient rich environment of the experiment is 204 supported by the SGR results and suggests greater assimilation of feed than the intensive 205 treatment. Due to their fast growth rate, fish larvae have a high requirement for amino 206 acids, the building blocks for the protein synthesis (Ronnestad et al. 2000). These nutrient 207 requirements may have been limiting for the extensive treatment compared to the 208 complete early-life stage diet that the intensive treatment received. The decreasing rate of 209 SGR (total length) for the intensive treatment may be correlated with observational data 210 that fin nipping was occurring and in fact had an effect on growth in length. This could 211 not be determined as 24 hour surveillance did not occur but we suggest future studies to 212 focus on such behaviors. Although the intensive treatment was larger in size overall, we 213 refute our hypothesis that intensively reared walleye grew at a faster rate than extensively 214 reared walleye. 215 The results of weight and length gain suggest that the intensive treatment grew at 216 a faster rate. This was possibly because the extensive treatment needed more than the 217 20% total fish biomass provided in minnow feed to grow at the same rate of the intensive 218 treatment. The number of minnows provided per walleye for the extensive treatment was 219 less than a 2:1 ratio while the intensive treatment had a ratio above 11:1 minnows to 220 walleye. This occurred at the start of the experiment and may have had an additive effect 221 throughout the experiment as there was not sufficient forage for optimal growth of the 222 extensive treatment but as the number of minnows provided increased, so did their 223 growth rates. Walleye studies show similarities to several studies that have been 224 conducted on the effects of environmental temperature on growth, digestion, and 225 metabolism in channel catfish (Shrable et al., 1969; West, 1966;Asadi, 1967; Kilambi et 226 al., 1970), but few quantitative data are available on the interactions of feeding rates and 227 temperature and their effects on growth, food conversion, and body composition of 228 catfish (Andrews and Stickney 1972). 229 A complete early-life stage diet may have also contributed to the overall higher 230 growth of the intensive treatment as we propose that this treatment assimilated feed better 231 than the extensive treatment. Stress may have limited feeding and assimilation of the 232 extensive treatment when they were moved from a natural pond to indoor tanks with 233 increased activities in and around the tanks. Wild broodstock were used; these walleye 234 are skittish and more easily stressed by overhead movement and hatchery activities than a 235 domesticated aquaculture species such as rainbow trout (Summerfelt 2000). 236 Conclusion 237 Rearing techniques and feed types will continue to change to optimize Walleye 238 growth for both conservation and aquaculture purposes. To improve the experiment, 239 Walleye strains should be the same as well as hatch dates to make a more accurate 240 comparison of growth between the intensively and extensively reared treatments. The 241 amount of spending a culturist is allowed can have a limiting effect on feed types to 242 provide the best chances of optimal growth. 243 Acknowledgments 244 This research was funded by the University of Wisconsin Stevens Point, College 245 of Letters and Sciences. I would like to thank everyone involved at the UWSP-Northern 246 Aquaculture Demonstration Facility and the University of Wisconsin-Stevens Point. I 247 would also like to thank Dr. Hartleb and Greg Fischer for a great learning experience. I 248 would also like to thank the Genoa National Fish Hatchery, Wisconsin DNR Governor 249 Thompson Fish Hatchery, and Hayward Bait & Bottle Shoppe. 250 Table 1. Summary of significance for mean total length and weight and mean total length 251 and weight gained of Walleye between treatments from the UWSP-Northern Aquaculture 252 Demonstration Facility 2014 intern project. df t-Stat F-Value P-Value 80 34.753 - - 61 24.653 - - 5 - 8.659 0.0604 7 - 15.9246 0.0104 Mean Total Length Mean Weight Mean Total Length Gained Mean Weight Gained 253 254 255 References Andrews J.W. and Stickney, R.R. 1972. Interactions of feeding rates and environmental 256 temperature on growth, food conversion, and body composition of Channel 257 Catfish. Transactions of the American Fisheries Society. 101:94-99. 258 Asadi, S. 1967. Effect of temperature on the digestive enzymes of channel catfish, 259 Ictalurus punctatus (Rafinesque). M.S. Thesis, Kansas State University, 260 Manhattan, Kansas. (Unpublished). 261 Barrows, F.T., W.A. Lellis, and J.G. Nickum. 1988. Intensive culture of larval Walleyes 262 with dry or formulated feed: Note on swim bladder inflation. The Progressive 263 Fish-Culturist, 50:160-166. 264 Bulkley, R.V., V.L. Spykermann, and L.E. Inmon. 1976. Food of the pelagic young of 265 walleyes and five cohabiting fish species in Clear Lake, Iowa. Transactions of the 266 American Fisheries Society 105:77-83. 267 268 269 270 271 272 273 274 Cameron, A., and J. Jones. 1983. The L.L. Bean game and fish cookbook. Random House, New York. Carmichael, G., M. Ring, and J. McCraren. 1991. Sea fare cookbook. Volume 1. American Fisheries Society, Bethesda, Maryland. Cheshire, W.F., and Steele, K.L. 1972. Hatchery rearing of walleye using artificial food. The Progressive Fish-Culturist 34:96-99. Forney, J.L. 1966. Factors affecting first-year growth of walleyes in Oneida Lake, New York. New York Fish and Game Journal 13:146-167. 275 Joint Subcommitte on Aquaculture. 1983. National aquaculture development plan, 276 volumes 1 and 2. Joint Subcommittee on Aquaculture, Washington, D.C. 277 Kapuscinski, A., and J. Mittelmark. 1986. Walleye culture in Minnesota. Minnesota Sea 278 Grant. Available: http://www.seagrant.umn.edu/aquaculture/mn_walleye_culture. 279 (October 2014). 280 Kilambi, R.V., J. Noble, and C.E. Hoffman. 1970. Influence of temperature and 281 photoperiod on growth, food consumption and food conversion efficiency of 282 channel catfish. Presented at the Southern Division American Fisheries Society 283 meeting, Atlanta, Georgia and to be published in the Proc. Of the 24th Ann. Conf. 284 Southeast. Assoc. Game and Fish Commrs. 285 Malison, J.A., T.B. Kayes, J.A. Held, and C.H. Amundson. 1990. Comparative survival, 286 growth, and reproductive development of juvenile Walleye and Sauger and their 287 hybrids reared under intensive culture conditions. The Progressive Fish-Culturist. 288 52:73-82. 289 290 291 McIntyre, D.B., F.J. Ward, and G.M. Swanson. 1987. Factors affecting cannibalism by pond-reared juvenile walleyes. Progressive Fish-Culturist 49:264-269. Ronnestad, I., L.E.C. Conceicao, C. Aragao, and M.T. Dinis. 2000. Free amino acids are 292 absorbed faster and assimilated more efficiently than protein in postlarval Senegal 293 Sole (Solea senegalensis). The Journal of Nutrition. American Society for 294 Nutritional Sciences. 295 Shrable, J.B., O.W. Tiemeier, and C.W. Deyoe. 1969. Effects of temperature on rate of 296 digestion by channel catfish. Progressive Fish-Culturist. 31:131-138. 297 Smith, L.L., Jr., and R.L. Pycha. 1960. First-year growth of the walleye, Stizostedion 298 vitreum vitreum (Mitchill), and associated factors in the Red Lakes, Minnesota. 299 Limnology and Oceanography 5:281-290. 300 Summerfelt, R.C. 1996. Walleye culture manual. NCRAC culture series 101. North 301 Central Regional Aquaculture Center Publications Office, Iowa State University, 302 Ames. 303 Summerfelt, R. C. 2000. A white paper on the status and needs of walleye aquaculture in 304 the North central region. North Central Regional Aquaculture Center. Iowa State 305 University. 306 West, B.W. 1966. Growth, food conversion, food consumption, and survival at various 307 temperatures of the channel catfish, Ictalurus punctatus (Rafinesque). M.S. 308 Thesis, University of Arkansas, Fayetteville, Arkansas. (Unpublished). 309 Figure Legends 310 Figure 1. Schematic outlining the study design of the intensive and extensive walleye 311 reared at the Northern Aquaculture Demonstration Facility during the summer of 2014. 312 Walleye started out in large circular tanks (intensive) and 0.4 acre outdoor rearing ponds 313 (extensive) prior to the experiment starting. At this time the fish were transferred to 240 L 314 indoor tanks at a density of 20 fish per tank. 315 Figure 2. Mean total length for days post hatch of walleye from the 2014 UWSP- 316 Northern Aquaculture Demonstration Facility intern project. Black diamonds and dashed 317 line represent the intensive treatment. White squares and a solid line represent the 318 extensive treatment. 319 Figure 3. Mean total length gained for days post hatch of walleye from the 2014 UWSP- 320 Northern Aquaculture Demonstration Facility intern project. Black diamonds and dashed 321 line represent the intensive treatment. White squares and a solid line represent the 322 extensive treatment. 323 Figure 4. Mean total length gained for days post hatch of walleye, excluding day 86, 2014 324 UWSP-Northern Aquaculture Demonstration Facility intern project. Black diamonds and 325 dashed line represent the intensive treatment. White squares and a solid line represent the 326 extensive treatment. 327 Figure 5. Specific growth rate (SGR) of total length for days post hatch of walleye from 328 the 2014 UWSP-Northern Aquaculture Demonstration Facility intern project. Black 329 diamonds and dashed line represent the intensive treatment. White squares and a solid 330 line represent the extensive treatment. 331 Figure 6. Mean weight for days post hatch of walleye from the 2014 UWSP-Northern 332 Aquaculture Demonstration Facility intern project. Black diamonds and dashed line 333 represent the intensive treatment. White squares and a solid line represent the extensive 334 treatment. 335 Figure 7. Mean weight gained for days post hatch of walleye from the 2014 UWSP- 336 Northern Aquaculture Demonstration Facility intern project. Black diamonds and dashed 337 line represent the intensive treatment. White squares and a solid line represent the 338 extensive treatment. 339 Figure 8. Specific growth rate (SGR) of weight for days post hatch of walleye from the 340 2014 UWSP-Northern Aquaculture Demonstration Facility intern project. Black 341 diamonds and dashed line represent the intensive treatment. White squares and a solid 342 line represent the extensive treatment. 343 344 Figure 1. 345 346 347 Figure 2. 348 349 350 351 Figure 3. 352 353 354 355 Figure 4. 356 357 358 359 Figure 5. 360 361 362 363 Figure 6. 364 365 366 367 Figure 7. 368 369 370 Figure 8.