Chapter 2 Steroid Biosynthesis and Regulation of Cortisol Production
advertisement

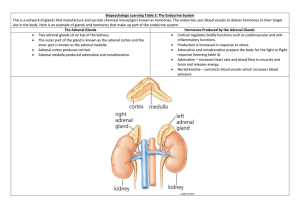
Chapter 2 Steroid Biosynthesis and Regulation of Cortisol Production The adult adrenal cortex produces three classes of steroid hormones: glucocorticoids, mineralocorticoids, and adrenal androgens. The specialized fetal adrenal, in conjunction with the fetal liver, the placenta, and some maternal organs, produces the steroid hormones of pregnancy. Cortisol, the major glucocorticoid in humans, is rapidly synthesized and secreted in response to ACTH; this is part of a response to stress and increases circulating levels of energy-providing compounds: glucose, free fatty acids and free amino acids. Aldosterone, the major human mineralocorticoid, is synthesized and secreted in response to angiotensin II; it helps prevent ECF depletion by promoting sodium reabsorption and fluid retention. The physiological roles of adrenal androgens are poorly understood. Because the actions of these steroid hormones result from effects on gene expression, the effects generally have a slow onset and are long-lived. Many clinically significant examples of adrenocortical hypo-, hyper-, and dys-function are known. Adrenal Morphology As shown schematically in Figure 1, the adrenal is surrounded by the capsule, a tough layer of connective tissue that protects the organ, and helps to maintain its structure and shape. Within the capsule are three layers, or zones, of cells that comprise the steroidogenic structures of the adrenal: the zona glomerulosa, the zona fasciculata and the zona reticularis. Structure Direction of Blood Flow Steroids Produced Capsule Zona Glomerulosa -Aldosterone Zona Fasciculata Cortisol, DHEA, DHEAS Zona Reticularis Cortisol, DHEA, DHEAS Medulla None Figure 1. Cartoon of Adrenal Morphology. The zona glomerulosa, the outermost zone, is the site of aldosterone production. The zona fasciculata is the largest zone, and is the major site of cortisol synthesis; the zona fasciculata also produces the adrenal androgens DHEA and DHEAS. The innermost steroidogenic zone is the zona reticularis, which also produces cortisol, 19 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt DHEA, and DHEAS. The steroid profiles produced in the zonæ fasciculata and reticularis are similar. It is not clear why there are two zonæ with the same products, although it has been suggested that the zona reticularis is more involved in adrenal androgen production. This is supported by the fact that the zona reticularis is poorly organized until adrenarche. The cortex surrounds the adrenal medulla, a non-steroid producing, modified neural tissue. The blood flow in the adrenal is centripetal (i.e. from the outside toward the center). As a result each zone is exposed to increasing levels of adrenal steroids. This is particularly important in the medulla, which requires high cortisol concentration to induce one of the enzymes necessary for epinephrine biosynthesis. The fetal adrenal consists of two zonæ: the relatively thin definitive zone (also called the neocortex) and the fetal zone. The definitive zone develops into the cortex after birth. The fetal zone, which is the site of DHEAS synthesis during fetal life, regresses and differentiates into the fasciculata following birth. The adrenal medulla is not present as a discrete structure during fetal life, and only forms during and after regression of the fetal zone. Note that the zona fasciculata is the middle zone of the adult; the reticularis is not fully organized until adrenarche (at age 8-12), when adrenal androgen production increases markedly. Adrenarche is also characterized by the loss of the connective tissue barrier between the cortex and medulla. The fetal adrenal is large compared to the size expected based on the size of the fetus. At term it usually contains about 8 grams of tissue (comparable in size to the adult organ), shrinking to about 5 grams a month later. The fetal adrenal has low levels of 3β-hydroxysteroid dehydrogenase/∆5-∆4isomerase; this results in DHEA and DHEAS being the major products. The other steroidogenic enzymes appear to be present, although probably largely in the definitive zone, and the fetal adrenal does produce some cortisol and aldosterone, either from cholesterol or from maternal progesterone. Adrenal Cell Structure There are some differences in detail between cells of different zonæ, but the general structure is similar to that of any cell specialized for steroid biosynthesis. The cells contain a number of lipid droplets, which are the storage site for cholesteryl esters, the precursors for all steroid hormone synthesis. The cells also contain large amounts of smooth endoplasmic reticulum and many mitochondria. The steroid synthetic enzymes are associated with the membranes of these organelles. Steroid Biosynthetic Pathways The major synthetic pathways for the adrenal steroid hormones are shown on the next page (Figure 2). The chart begins with cholesterol and goes to the adrenal products aldosterone, cortisol, and DHEA(S), and to the non-adrenal steroid hormones progesterone, testosterone, and estradiol. The steroids within the grey box are not produced by the adrenal. Note that while progesterone is synthesized in the adrenal, it is generally not released in significant quantities; instead it merely serves as a precursor for some of the adrenal hormones. 20 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt O HO Cholesterol * –O SO 3 P450-scc O O DHEA-Sulfate O OH HO HO 17α-OH-Pregnenolone Pregnenolone HO Dehydroepiandrosterone 3β-Hydroxysteroid dehydrogenase/∆5-∆4-isomerase O O O O OH O O 17α-OH-Progesterone Progesterone O Androstenedione HO Estrone 17β-Hydroxysteroid dehydrogenase P450-21 OH OH O O OH OH OH O O Deoxycorticosterone * P450-18 or 11β P450-11β OH O HO O Corticosterone Estradiol * OH O O Aldosterone (aldehyde form) Cortisol Name Cholesterol side chain cleavage enzyme 17α-hydroxylase/17-20-lyase 21-hydroxylase 11β-hydroxylase 18-hydroxylase aromatase Note that P450-17α catalyzes both reactions in the conversion of pregnenolone to DHEA and in the conversion of progesterone to androstenedione. Non-enzymatic OH HO O O O OH Designation * P450scc P450-17α P450-21 * P450-11β * P450-18 P450-arom P450-18 HO HO O O * Testosterone OH HO O O Deoxycortisol * -- Denotes mitochondrial enzyme (all others located in the smooth endoplasmic reticulum. Aldosterone (hemiacetal form) Figure 2. The major steroid biosynthetic pathways of the adrenal. (The steroids within the grey box are not normally produced within the adrenal.) 21 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt The chart appears complex but is logically organized. In each case the difference between one row and the next lower row is the reaction catalyzed by a single enzyme. The reactions linked by the boxes are catalyzed by the same enzyme. Thus, the first row (with the steroids pregnenolone, 17α-hydroxy-pregnenolone, and dehydroepiandrosterone (DHEA) contains 3 -hydroxy- 5 steroids. (It may help to refer to the nomenclature rules for steroids in Chapter 1.) The second row (progesterone, 17α-hydroxy-progesterone, and androstenedione) contains 3-keto- 4 steroids. All are produced from first row steroids by the enzyme 3β-hydroxysteroid dehydrogenase/∆5-∆4-isomerase. The third row contains 21-hydroxysteroids (deoxycorticosterone and deoxycortisol). These are produced from the corresponding second row steroids by the enzyme 21-hydroxylase. The fourth row contains 11 hydroxysteroids (corticosterone and cortisol), products of the enzyme 11βhydroxylase (cortisol, and some corticosterone), or 18-hydroxylase (corticosterone); in the case of corticosterone production, the same reaction can be catalyzed by two different enzymes. The fifth row contains a single steroid, aldosterone, the final product of the 18-hydroxylase. The columns are similarly organized; the first column contains 21-carbon steroids, the second column contains 21-carbon, 17 -hydroxysteroids, and the third column contains 19-carbon steroids. Note that P450-17α catalyzes two separate reactions: 17α-hydroxylation of pregnenolone and progesterone, and the cleavage of the short side chain to produce the 19-carbon steroids. The enzyme can release the 21-carbon 17α-hydroxysteroids, allowing the production of (eventually) cortisol, or can catalyze the second step, and release the 19-carbon final product. The thick arrows in Figure 2 are cytochrome P450 enzymes. (Note that these all have two names, which are given in the table on Figure 2.) These enzymes are members of a large superfamily of heme-containing proteins which catalyze mixed function oxidation reactions, using molecular oxygen and electrons from NADPH to modify the substrate. The mechanisms of these enzymes are very complex, but allow an attack on an unsubstituted carbon atom, a process that is chemically very difficult. They all catalyze a reaction that follows the equation: R-H + O2 + NADPH cytochrome P450 R-OH + H2 O + NADP+ In some cases the process ends there, and in some cases continues with further steps, such as the breaking of C-C bonds. Three of the enzymes, all cytochromes P450 (P450scc, P450-11β, P450-18) are located within the mitochondria; all of the other steroidogenic enzymes are found in the smooth endoplasmic reticulum. The first enzyme in the pathway is cytochrome P450scc, also known as cholesterol side chain cleavage enzyme. P450scc catalyzes the first, slowest, and therefore, rate limiting step for hormone synthesis. As you might expect from the observation that P450scc catalyzes the rate limiting step, P450scc is also the 22 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt major site of physiological regulation. In each steroid synthetic tissue, the control hormone for that endocrine gland increases the activity of P450scc. Pregnenolone, the product of the P450scc reaction, is converted to the final hormone product by sequential steps along the pathway depending on the enzymes that are present in that tissue. Thus, the zona glomerulosa makes aldosterone, because it contains 3β-HSD, P450-21, and P450-18, and lacks P450-17α. Gonadal tissues lack P450-21 and therefore can only make the sex steroids progesterone, testosterone and estradiol. P450-18 is closely related (about 95% sequence identity) to P450-11β, and has both 11β-hydroxylase activity and the ability to catalyze the additional reactions that produce aldosterone. The majority of human corticosterone is produced by the action of 11β-hydroxylase on deoxycorticosterone present in the zona fasciculata; normally, 18-hydroxylase carries out all of the reactions required to convert deoxycorticosterone to aldosterone without releasing the intermediates. This chart is important in understanding the normal biosynthetic processes involved in steroid production. In cases of genetic enzyme deficiencies, however, the patient will make products determined by enzymes that are still present. For example, a patient lacking P450-21 can only make androgens. A patient lacking P450-17α has an entire adrenal that acts like the zona glomerulosa, producing large amounts of deoxycorticosterone, corticosterone, and aldosterone, but nothing else. You will need to understand the chart so that you can make sense of clinical findings. Fetal Steroid Synthesis The steroidogenic pathways discussed above apply primarily to the adult adrenal. During pregnancy, however, the amounts and types of adrenal steroids produced are somewhat different. Production of the major steroids of pregnancy requires a cooperative interaction between the mother, the fetus, and the placenta. It should be noted that human fetal steroid production is not as well studied as that of the adult. In addition to the morphological differences between the fetal and adult adrenal, the fetal adrenal has two enzymatic differences. For the majority of gestation, the fetal adrenal has very low 3β-hydroxysteroid dehydrogenase/∆5-∆4-isomerase activity (with probably none at all in the fetal zone). (The fetal adrenal does produce some cortisol and aldosterone, either from cholesterol via the small amount of 3β-HSD in the definitive zone, or from maternal progesterone.) The fetal adrenal also has a high sulfotransferase activity. As a result of these two properties, the main fetal adrenal reaction is: Cholesterol Pregnenolone DHEA DHEAS The fetal liver contains considerable amounts of an enzyme not shown in Figure 2, 16α-hydroxylase (P450-16α). The reaction catalyzed by P450-16α is shown in Figure 3. The fetus therefore converts cholesterol to two major steroid products: DHEAS 23 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt and 16α-hydroxy-DHEAS. O O P450-16α O3 SO OH O3 SO DHEAS 16α-hydroxy-DHEAS Figure 3. The reaction catalyzed by P450-16α. Placental Steroid Synthesis The placenta has high levels of a few selected steroidogenic enzymes. It has high levels of P450scc and 3β-hydroxysteroid dehydrogenase/∆5-∆4-isomerase activity, but lacks both P450-17α and P450-21. As a result, the placenta synthesizes large amounts of progesterone, but is incapable of producing cortisol or DHEA. The placenta also has large amounts of a sulfatase that removes the sulfate from circulating DHEAS and 16α-hydroxy-DHEAS. Finally, the placenta has high levels of aromatase (P450arom), which converts androgens to estrogens; note that all four enzymes – the sulfatase, 3β-HSD, 17β-HSD, and aromatase – are required to convert DHEAS to estradiol, or 16α-hydroxy-DHEAS to estriol (Figure 4). From Circulation O O Sulfatase O3 SO HO DHEAS O O 3β-HSD O DHEA HO Androstenedione Estrone 17β-Hydroxysteroid dehydrogenase OH O OH OH O3 SO O 16α-hydroxy-DHEAS HO Testosterone Estradiol Sulfatase O OH OH 3β-HSD HO 16α-hydroxy-DHEA OH O OH OH OH 17β-HSD O 16α-hydroxy-Androstenedione O 16α-hydroxy-Testosterone HO Estriol Figure 4. Estrogen production in the human placenta. The steroids in the grey box are obtained from the circulation; the reactions shown are catalyzed by placental enzymes. Note that most of these reactions can also occur in other tissues. Because the placenta lacks P450-17α, it cannot produce DHEA or androstenedione by itself; therefore, the placenta can only make estrogens by converting substrates (e.g., DHEA and 16α-hydroxy-DHEA) synthesized elsewhere. Because the fetus 24 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt makes and secretes large quantities of these steroids, placental estrogen production is quite high. Feto-Placental Unit During the later stages of pregnancy the mother, placenta, and fetus collaborate to make large amounts of steroids. The maternal liver secretes LDL, which transports cholesterol to the maternal adrenal and to the placenta. The placenta produces large amounts of progesterone from cholesterol. The fetal adrenal synthesizes DHEA and DHEAS as major products; the maternal adrenal also produces these steroids. The DHEA(S) produced by both the mother and the fetus are converted to estradiol by the placenta. The fetal liver converts fetal DHEAS to 16α-hydroxyDHEAS (note that maternal DHEAS is not 16α-hydroxylated by the fetus, since, in order to reach the fetal liver, the maternal steroid would have to traverse the placenta, and would be converted to estradiol during that time); the fetal 16αhydroxy-DHEAS is then converted to estriol by the placenta. Finally, the maternal liver 21-hydroxylase activity increases during pregnancy, and the maternal liver therefore converts some of the placental progesterone to deoxycorticosterone (DOC). Table I. Pregnancy Steroid Production Levels at Term Steroid Amount Secreted (mg/day) Progesterone 250 Estradiol 20 Estriol 80 Deoxycorticosterone 2-10 The reason for these high levels of steroids is not fully understood. Progesterone production at term, for example, is 5 to 10 times higher than the peak level of progesterone observed during the menstrual cycle, and the circulating levels of progesterone are far in excess of the amount required to saturate the progesterone receptor. The high level of progesterone is thought to have an immunosuppressive effect that prevents rejection of the fetus by the immune system of the mother. This may be mediated by the glucocorticoid receptor; the levels of progesterone are so high that during pregnancy progesterone may act as a glucocorticoid in spite of the weak affinity of progesterone for the glucocorticoid receptor. Alternatively, the immunosuppression may be mediated by the progesterone receptors that are present in some types of immune system cells. The function of the high levels of estradiol is also incompletely understood. One function is probably (as with progesterone) to induce immunosuppression. Estradiol is known to affect the reninangiotensin system, and therefore blood flow and blood pressure. Estradiol probably regulates the levels of placental 11β-HSD, and therefore the exposure of the fetus to maternal cortisol. Estradiol may stimulate progesterone production both by increasing placental P450scc activity and by 25 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt increasing placental LDL uptake. Estradiol and progesterone are known to affect the uterus, both in maintaining quiescence during pregnancy, and in preparing for labor, and to assist in preparing the mammary gland for lactation. Finally, increased fetal DHEA synthesis (resulting in an increased estradiol/progesterone ratio) may be one signal that the fetus is ready for the onset of labor. Estriol is a weak estrogen, and is only produced in significant quantities during pregnancy. The physiological role of estriol is unknown; however, estriol levels are useful clinically as a monitor of fetal health. In contrast to progesterone production, which only requires the placenta, and can continue for some time after death of the fetus, estriol synthesis requires a functional fetal adrenal, fetal liver, fetal circulation (to transport the steroids between the various organs), in addition to a functioning placenta. Deoxycorticosterone is a weak mineralocorticoid, and as such increases sodium retention and blood pressure. Overproduction of DOC and some of its metabolites has been linked to some types of pregnancy-induced hypertension. Regulation of Cortisol Secretion Adrenal cortex hormone production is primarily regulated by two separate systems corresponding to the two main hormonal products, glucocorticoids and mineralocorticoids. Quantitatively, the major steroid product of the adult adrenal is cortisol, and we will examine the regulation of its production first. Cortisol, like all other steroid hormones, is not stored at its site of synthesis. Instead, the release of steroid hormones is controlled almost entirely by regulating their rate of synthesis. Stress Hypothalamus (–) CRH Anterior pituitary ACTH (–) (+) Adrenal cortex Cortisol Target tissues Figure 5. The hypothalamic - pituitary - adrenal axis. Note the closed control loop among the hypothalamus, pituitary and the endocrine gland. The secretion of cortisol is ultimately controlled by the central nervous system. Stress (e.g., injury, fasting, surgery, final exams) induces the hypothalamus to release corticotrophin releasing hormone (CRH) into the portal vein. CRH binds receptors on corticotrophs in the anterior pituitary, and stimulates the release of adrenocorticotrophic hormone (ACTH). ACTH travels through the blood26 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt stream to the adrenal cortex, and increases synthesis and release of cortisol. Cortisol then travels through the bloodstream back to the hypothalamus and pituitary to exert negative feedback on CRH and ACTH secretion, and to target tissues throughout the body to mediate the stress response. The system is a closed feedback loop (Figure 5). Cortisol suppression of CRH and ACTH prevents overproduction of cortisol (in normal individuals); breakdown of this feedback system causes severe problems due to uncontrolled production of cortisol. ACTH is a 39 amino acid peptide hormone. It is synthesized as part of a larger precursor polypeptide called proopiomelanocortin (POMC); ACTH release requires proteolytic processing of POMC. POMC also contains at least two other hormones, β-endorphin and melanocyte stimulating hormone (MSH). β-endorphin is an endogenous opioid which has a variety of apparent functions in experimental animals and in vitro; its physiological functions are currently uncertain. MSH stimulates melanin production; excess production of POMC can result in hyperpigmentation. POMC has a negative GRE in the upstream untranscribed region of the gene; glucocorticoid receptors, following activation by cortisol binding, bind to the nGRE, and markedly decrease POMC expression, and therefore, decrease production of ACTH. In contrast, one effect of CRH is to increase POMC expression by activating a different, positive, enhancer element of the POMC gene (in addition to stimulating the release of vesicles containing previously synthesized ACTH). CRH is a 41 amino acid peptide hormone, also synthesized as part of a larger precursor. Cortisol decreases hypothalamic transcription of the CRH gene. Estrogens stimulate CRH transcription; the reason for this is not understood. Diurnal Variation of Cortisol Production In addition to the stress response, there is a diurnal variation in cortisol levels: cortisol secretion rises during sleep, and is highest in the morning. This occurs in spite of the negative feedback effects of cortisol on the hypothalamus and pituitary. The reason for the diurnal rhythm of cortisol secretion (in response to increased ACTH, almost certainly as a result of increased CRH) is not understood. Aldosterone levels follow a similar pattern to cortisol, although this may not be a response to ACTH, since the pattern of aldosterone release also occurs under conditions in which release of cortisol is suppressed (e.g., dexamethasone treatment or hypophysectomy). Sleep Noon 4 PM 8 PM Midnight 27 4 AM 8 AM Noon Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt Figure 6. Variation in plasma cortisol levels during the day. Even in the absence of significant stress, cortisol levels exhibit considerable variation. The graph shows levels experienced in a “typical” individual. Note especially the marked rise in cortisol during the night. It is not entirely clear how the stress and diurnal rhythm effects are controlled separately. In part it may be a consequence of differential occupancy of GR Type I and Type II receptors, or by altering GR Type I/Type II heterodimer formation. On the other hand, there is evidence that arginine vasopressin (AVP) is involved. Although AVP alone has little effect, in conjunction with CRH, AVP causes a marked rise in ACTH secretion. Unlike CRH, AVP has little effect on POMC synthesis; however, AVP release by the hypothalamus is not as sensitive to inhibition by glucocorticoids. Thus, AVP may mediate the additional increase in ACTH release required for response to acute stress during periods of chronic stress, when CRH levels are suppressed by glucocorticoids. Mechanism of ACTH Action The zonæ fasciculata and reticularis express the ACTH receptor, a cell surface protein coupled to adenylyl cyclase. Binding of ACTH results in an elevation in cyclic AMP levels, and therefore increased cAMP-dependent protein kinase activity. This kinase then phosphorylates a variety of other proteins. The response of the adrenal to stimulation depends on the intensity and duration of exposure to ACTH. (Note that gonadal steroidogenic tissues respond in a generally similar manner to the relevant trophic hormones.) Transient increases in ACTH elicit rapid effects (time-scale minutes): phosphorylation-dependent activation of cholesterol esterase (which releases cholesterol from the lipid droplet storage), and of the proteins responsible for cholesterol transport into the mitochondria. A protein called StAR (Steroidogenic acute regulatory protein) is thought to mediate the hormonedependent transport of cholesterol to adrenal and gonadal inner mitochondrial membranes by an as yet poorly understood mechanism. Lack of this protein results in a rare form of congenital adrenal hyperplasia; this observation, along with other studies, strengthens the hypothesis that the presentation of cholesterol to P450scc is a major control point in steroid hormone biosynthesis. Prolonged increases in ACTH elicit medium term effects (time scale 6 to 36 hours): increased synthesis of all of the steroidogenic enzymes, and especially of P450scc, and increased uptake of cholesterol from circulation. Chronic stimulation of the adrenal by ACTH results in adrenal hyperplasia (time scale weeks to months): the medium and long term effects appear to be mediated by CREB (cAMP Response Element Binding protein) and several other transcription factors activated by cAMP-dependent protein kinase. Most of the adrenal steroidogenic enzyme genes contain a cAMP Response Element, and therefore their transcription is controlled in part by CREB. In addition, prolonged high levels of ACTH result in proliferation of adrenal steroid producing cells (this is 28 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt also thought to be mediated by cAMP). ACTH-stimulation is required to prevent adrenal atrophy (time scale lifetime): the adrenal requires a constant low level of ACTH to maintain normal responsiveness to hormonal stimuli. This last effect is of significant clinical relevance for patients treated with pharmacological levels of glucocorticoid agonists for prolonged periods. The high levels of synthetic glucocorticoid are perceived by the hypothalamus and pituitary and result in complete suppression of ACTH release. Consequently, if the treatment is continued for several months, the adrenal cortex regresses and loses its ability to produce cortisol. If the glucocorticoid treatment is stopped suddenly, the patient may suffer from the lack of cortisol; it is therefore necessary to gradually reduce the levels of treatment to allow the pituitary and adrenal to recover. Depending on the level of suppression, it may take the pituitary a month or more to recover its ability to produce ACTH, and may take from several months to a year for the adrenal cortex to regain the capacity to synthesize normal levels of cortisol. Measurement of ACTH and CRH Levels While it should, in principle, be possible to measure ACTH levels in serum, in practice, obtaining meaningful values is very difficult. This is in part due to the fact that ACTH is readily degraded during sample storage prior to assay. More importantly, ACTH levels vary widely, due to the diurnal variation, to stressinduced changes, to its short serum half-life (~10 minutes), and to the fact that ACTH is released in pulses by the pituitary. All of these factors make ACTH RIA results of limited value in diagnosing adrenal disorders. Under normal conditions, measuring CRH levels is also almost useless. Hypothalamic CRH is released into the portal vein, and travels directly to the pituitary. Therefore, unless the sample is taken directly from the portal vein (which has been done, but is a difficult and risky procedure), the dilution that occurs in circulation tends to reduce systemic CRH levels to below the limit of detection. In addition, CRH is a paracrine factor that is thought to mediate some types of stress responses (especially those involved in inflammation); the peripheral production of CRH in these conditions is under separate regulation from that in the hypothalamus, and is unrelated to the levels sensed by the pituitary. In other words, even if you can detect CRH in serum, the levels will not tell you anything about pituitary ACTH release. The placenta releases CRH into circulation; this may result in physiologically relevant increases in ACTH levels, and in increased adrenal activity during pregnancy. Some tumors produce ectopic ACTH or CRH, causing some forms of the cortisol hypersecretion disorder Cushing’s syndrome (see Chapter 3). Tumor production of these hormones tends to be less variable than that of the hypothalamus and pituitary, and in these cases, measurement of serum ACTH and/or CRH may have some diagnostic value. 29 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt Supplement: Characteristics of Adrenal Steroidogenic Enzymes Expression of steroidogenic enzymes An orphan member of the steroid hormone receptor superfamily called Steroidogenic Factor-1 (SF-1) or Adrenal-4-binding protein has been implicated in mediating tissue specific expression of the steroid biosynthetic enzymes, of StAR (see box on previous page), and, in adrenal cortex, of the ACTH receptor. Hormone-induced expression appears to be indirectly regulated by cAMP via CREBinduction of expression of additional transcription factors, which then act together with SF-1 to increase steroidogenic enzyme synthesis. The exception to this appears to be P450-18, which may be directly regulated by cAMP-stimulated phosphorylation of CREB; this is difficult to reconcile with the apparent phosphatidyl inositol second messenger system that mediates angiotensin II action (see Chapter 3). Mice homozygous for a knockout mutation of the SF-1 gene lack adrenals and gonads, suggesting that the SF-1 protein is required for differentiation of steroidogenic cell types. The homozygous mice die 1 to 8 days after birth, due to mineralocorticoid and glucocorticoid insufficiency. The ligand, if any, for SF-1 has not been identified. A second orphan member of the steroid hormone receptor superfamily called DAX-1 may be preferentially involved in expression of adrenal enzymes; some X-linked forms of congenital adrenal hypoplasia are due to lack of this protein. P450scc Cholesterol side chain cleavage enzyme catalyzes the rate limiting step for all steroid hormone biosynthesis. It is therefore the main regulatory control point in each steroidogenic cell type. P450scc is located in the inner mitochondrial membrane; this is physiologically relevant because cholesterol cannot enter the mitochondria without assistance, and therefore allows substrate presentation to be a major mechanism for regulation of P450scc activity. Expression of P450scc is controlled by ACTH in all zonæ of the adrenal with additional control by angiotensin II in the zona glomerulosa. P450scc activity is controlled primarily by LH in the testis, and by both LH and FSH (depending on the cell type) in the ovary. These trophic hormones all increase P450scc activity of by several mechanisms, including increased mobilization of cholesterol (de-esterification and transport into the mitochondria) and increased amounts of the enzyme (via increased transcription). Longer term effects include stimulating the production of all of the steroidogenic enzymes, increased cholesterol uptake from the circulation, and hypertrophy and hyperplasia of the endocrine cells. Deficiency in P450scc has been observed in a few cases; as expected, it results in congenital adrenal hyperplasia and sexual differentiation abnormalities due to an inability to produce all steroid hormones. There is a single gene for P450scc located on chromosome 15. 3 -hydroxysteroid dehydrogenase/ 5- 4-isomerase 3β-HSD, a non-cytochrome P450 enzyme located in the endoplasmic reticulum, converts 3β-hydroxy∆5-steroids to 3-keto-∆4-steroids. Both dehydrogenase and isomerase activities are mediated by the same protein. 3β-HSD activity is found in the adrenal, gonads, placenta, and liver, as well as other tissues. The fetal zone of the fetal adrenal has very low concentrations of 3β-HSD, while the definitive zone has significant 3β-HSD activity; this is consistent with the functions of the two zonæ in producing DHEA and cortisol, respectively. Some tissues (especially liver) exhibit sexually dimorphic levels of 3β-HSD expression. At least two genes code for active isozymes of 3β-HSD; both, along with a pseudogene, are located on chromosome 1. Type I, which has a higher affinity for steroid substrates, appears to be found predominantly in the placenta and in peripheral tissues (based on mRNA levels), while Type II is the major form in the adrenals and gonads. Deficiency in Type II 3β-HSD is one cause of congenital adrenal hyperplasia (~5% of cases), and of androgen excess in women. The latter is either a direct result of elevated DHEA levels or (more 30 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt likely) a result of peripheral conversion of DHEA to androgens by the non-adrenal specific form(s) of 3β-HSD. No cases of Type I deficiency have been reported, probably because the resulting lack of placental progesterone production terminates pregnancy. 17 -hydroxylase/17-20-lyase P450-17α, a smooth endoplasmic reticulum enzyme, is required for cortisol, C19 steroid, and C18 steroid production. In humans, it is found in adrenal and gonadal tissue, but not in placenta. The single P450 can perform both 17-hydroxylation and the lyase reaction, although mutant forms that retain only 17-hydroxylase or only lyase activity have been discovered. The 17-hydroxylation occurs more rapidly than the lyase reaction, allowing release of 17-hydroxylated substrates for cortisol synthesis. There appear to be other regulators of partitioning, possibly including relative amounts of cytochrome reductase, or alterations in membrane lipid composition. There is a single P450-17α gene on chromosome 10. Deficiency in P450-17α is a rare cause of congenital adrenal hyperplasia, and usually results in sexual differentiation abnormalities (ambiguous genitalia, hypogonadism/pseudohermaphroditism) due to lack of sex steroid hormone biosynthesis, and in hypertension (sometimes with suppressed aldosterone levels in spite of high DOC and corticosterone as a result of a suppressed zona glomerulosa). There may be differences in expression of mutant P450-17α in adrenal and gonads, since some cases are either normotensive or have low-normal reproductive functioning, or both. Glucocorticoid insufficiency is extremely rare, probably due to high levels of zona fasciculataproduced corticosterone fulfilling cortisol functions. Human P450-17α appears to have low affinity for C21 ∆4 steroids; humans probably use the ∆5 pathway (i.e. ∆5: pregnenolone → 17α-hydroxy-pregnenolone → DHEA → androstenedione, rather than ∆4 (pregnenolone → progesterone → 17α-hydroxy-progesterone → androstenedione) for most steroidogenesis. 21-hydroxylase P450-21, a smooth endoplasmic reticulum enzyme, is required for the synthesis of both aldosterone and cortisol in humans, and for corticosterone production in rats. Deoxycorticosterone, a weak mineralocorticoid normally produced in small amounts by the adrenal, is produced in significant quantities from progesterone by liver 21-hydroxylase activity during pregnancy, and may account, at least in part, for pregnancy-induced hypertension. Deficiency in P450-21 accounts for >90% of all cases of congenital adrenal hyperplasia. P450-21 has a closely related pseudogene located in close proximity to the expressed gene on chromosome 6; gene conversions and unequal crossing-over between the gene and the pseudogene account for many new cases of 21-hydroxylase deficiency. Mild defects in 21-hydroxylase are much more common than complete lack of the enzyme; in most cases, these result in hypertension due to elevated aldosterone levels, while cortisol levels are normal. Approximately 15-20% of the general population (with higher incidence in some ethnic groups, such as Askenazi Jews) are carriers of at least mild defects in 21hydroxylase activity. 11 -hydroxylase P450-11β is an adrenal specific mitochondrial enzyme related to P450scc. P450-11β is required for cortisol synthesis, but in humans is probably not involved in aldosterone synthesis. Bovine P450-11β is capable of performing both 11β and 18 hydroxylations (the mechanism for preventing aldosterone synthesis in the bovine zona fasciculata is not understood), while humans and rats use two separate enzymes (note however, that rats use corticosterone as their major glucocorticoid, and therefore requiring two enzymes for aldosterone synthesis may be a way of regulating the relative amounts of the two steroid hormones). Deficiency in P450-11β causes congenital adrenal hyperplasia (~5% of cases); untreated it usually results in hypertension due to elevated DOC or other adrenal steroid metabolite levels (aldosterone synthesis is usually still possible due to P450-18, but is suppressed by the DOC-related hypertension), and in either precocious puberty (males) or female pseudohermaphroditism due to high androgen levels. 31 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt 18-hydroxylase (or aldosterone synthetase or 18-methyloxidase) P450-18 is very closely related to P450-11β (the genes are located in close proximity on chromosome 8), but is normally only expressed in the zona glomerulosa. The expression of P450-18 is under the control of angiotensin II. Inappropriate expression of P450-18 in the zona fasciculata (usually as a result of an unequal crossing-over event that puts the P450-18 gene under control of the P450-11β promoter) results in an aldosterone overproduction disorder called dexamethasone-suppressible aldosteronism. P450-18 is capable of catalyzing all of the reactions involved in converting DOC to aldosterone, and in fact, probably uses DOC as its major substrate. Mutations in 18-hydroxylase have been observed that result in abolished activity, or in enzymes that are still capable of 11β-hydroxylation but which have lost either 18-hydroxylase or 18-oxidase activity, or both. Selected Non-Adrenal Enzymes 17 -hydroxysteroid dehydrogenase 17β-HSD catalyzes the interconversion of 17-keto and 17β-hydroxysteroids. At least four genes located on different chromosomes produce enzymes with 17β-HSD activity; the isozymes differ in substrate specificity, in reaction direction (i.e. some forms predominantly catalyze the reduction of 17-ketosteroids, and some the reverse reaction), and in tissue distribution. These observations suggest that isozyme distribution may regulate levels of effective hormone activity in different tissues (since the 17-ketosteroids are much less active than their 17β-hydroxy counterparts). At least two isozymes of 17β-HSD can also convert progesterone to the inactive 20α-hydroxyprogesterone. Deficiency in some isozymes of 17β-HSD has been shown to be responsible for some rare types of ambiguous genitalia; 17β-HSD deficiency does not affect adrenal steroid hormone levels. Aromatase (or Estrogen Synthetase) Aromatase is the smooth endoplasmic reticulum cytochrome P450 that catalyzes the conversion of C19 3-keto- ∆4 androgens to C18 estrogens. It is found predominantly in the gonads (especially the granulosa cells of the ovary) and in placenta, but is also present in significant amounts in adipose tissue, skin, and brain. Deficiency in aromatase is extremely rare, possibly because lack of placentally synthesized estrogens compromises fetal survival. There is a single gene for aromatase located on chromosome 15. 11 -hydroxysteroid dehydrogenase 11β-HSD catalyzes the interconversion of cortisol and cortisone. At least two genes encode enzymes with this activity; the Type I enzyme, found in the liver and many other tissues, catalyzes the reaction in both directions, and has relatively low affinity for cortisol; this enzyme can activate circulating (and exogenously administered) cortisone. The Type II enzyme has high affinity for cortisol and corticosterone, is found predominantly in mineralocorticoid-responsive tissues and in placenta, and catalyzes solely the oxidation of 11β-hydroxysteroids to their inactive 11-ketosteroid counterparts. The Type II enzyme is responsible for the specific mineralocorticoid response. Defective Type II 11β-HSD or overexpression of an inhibitor of the enzyme results in Syndrome of Apparent Mineralocorticoid Excess. References Simpson & MacDonald (1981) “Endocrine physiology of the placenta.” Ann. Rev. Physiol. 43: 163-188. Dallman (1993) “Stress update: adaptation of the hypothalamic-pituitary-adrenal axis to chronic stress.” Trends Endocrinol Metab 4: 62-69. 32 Chapter 2. The Adrenal Cortex -- Part I Endocrine -- Dr. Brandt Seeman & Robbins (1994) “Aging and hypothalamic-pituitary-adrenal response to challenge in humans.” Endocr. Rev. 15: 233-260. Wajenberg et al. (1994) “Ectopic adrenocorticotrophic hormone syndrome.” Endocr. Rev. 15: 752-787. Pepe & Albricht (1995) “Actions of placental and fetal adrenal steroid hormones in primate pregnancy.” Endocr. Rev. 16: 608-648. Stocco & Clark (1996) “Regulation of the acute production of steroids in steroidogenic cells.” Endocr. Rev. 17: 221-244. Parker & Schimmer (1997) “Steroidogenic factor 1: A key determinant of endocrine development and function.” Endocr. Rev. 18: 361-377. Mesiano & Jaffe (1997) “Developmental and functional biology of the primate fetal adrenal cortex.” Endocr. Rev. 18: 378-403. 33