Assignment 11 Reading !

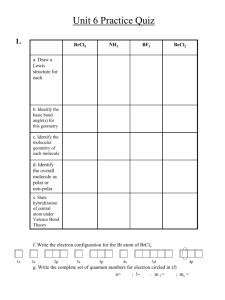
PHY 142 !
Assignment 11 !
Summer 2013
Reading : Modern Physics 1, 2
Key concepts: Bohr model of hydrogen; photoelectric effect; deBroglie wavelength; uncertainty principle; nuclear decays; nuclear binding energy.
1.
!
Comment on these early assumptions about atomic structure, indicating which one(s) survived when quantum mechanics was discovered in the 1920’s.
a.
!
Electrons circulate about the nucleus in definite orbits.
b.
!
Radiation by an atom results from acceleration of the electrons.
c.
!
The frequency of radiation is the same as the frequency of revolution of an electron in its orbital motion.
d.
!
The energy carried off by radiation is equal to the loss of energy by the radiating atom.
2.
!
Comment on the following statements about the restrictions imposed by the uncertainty principle.
a.
!
In principle, it renders the program of Newtonian mechanics hopeless.
b.
!
It forbids a system to have a precise value of the angular momentum.
c.
!
It poses no problem to the practical use of classical physics for systems of macroscopic size.
d.
!
It means that the Bohr model’s use of precise orbits for electrons in atoms cannot be valid.
3.
!
Compton’s analysis of the effect named for him assumed that the process is an elastic collision between an incoming X-ray photon and a free (conduction) electron on a metal surface. Because of momentum conservation the electron must recoil after the collision, taking away some of the photon’s energy with it.
This loss of energy is what makes the frequency of the scattered photons lower, by the equation
E
= hf . But many of the X-rays in the experiment came off the metal with essentially unchanged frequency. Explain this in terms of collisions between photons and electrons that are bound tightly to their atoms, so that the whole atom must recoil. [How does the energy lost by a light particle in an elastic collision depend on the mass of the heavy particle it hits?]
PHY 142 !
Assignment 11 !
Summer 2013
4.
!
There is another “uncertainty” rule, relating energy and time, usually written as
Δ
E
⋅ Δ t . It says the uncertainty in energy of a state varies inversely with the average time for an appreciable change in that state. “Appreciable change” usually means change to a different state, e.g., a quantum jump in an atom, decay of a radioactive nucleus, or decay of an unstable particle into other particles.
!
a.
!
Since the ground state of an atom is stable, its energy has no uncertainty, but an atom in an excited state will “decay” to the ground state, emitting a photon. For a typical case the lifetime of the excited state is about 10
− 8
s and the photon’s energy is about 1 eV. Estimate the fractional uncertainty
Δ E / E in the photon’s energy.
b.
!
Measurements of the mass of the ρ particle, for which mc
2 ≈ 750 MeV, show an uncertainty of about 150 MeV. Assuming this is not just experimental uncertainties, estimate the lifetime of this particle.
[Use = 6.6
× 10
− 16
eV-s.]
5.
!
Some numerical comparisons.
a.
!
What is the energy of a photon of wavelength 500 nm, in eV?
b.
!
What is the wavelength of a 10 MeV γ -ray?
c.
!
When two molecules of hydrogen combine with one of oxygen to form two molecules of water, about 6 eV of energy is released in the form of heat. The rest energy mc
2
for a proton or neutron is about 1 GeV. Count up the number of protons and neutrons involved in the reaction and determine the fraction of mass that is converted to energy. [The electron masses are negligible.] d.
!
Fission of
235
92
U produces about 200 MeV of released energy. What is the fraction of mass converted to energy in this case?
6.
!
Our supply of helium gas on earth comes mainly from natural gas wells. The organic gases in those wells presumably originated in plant and animal materials on the earth’s surface at some early time. But helium was never part of those materials. Where did the helium underground originate?
PHY 142 !
Assignment 11 !
Summer 2013
7.
!
Shown is a rough sketch of the curve of binding energy per nucleon as a function of nucleon number. a.
!
Use it to explain why nuclei more massive than
56
26
Fe are not generated in normal stars, which get their energy from nuclear fusion.
E b
/A
100 200 A b.
!
How can we explain the presence of the more massive atoms on earth?
c.
!
Use the curve to explain the production of energy by fission of heavy nuclei.
!
!
8*.
!
In the Bohr model of hydrogen, in the n th stationary state the electron is in a circular orbit around the proton with radius and velocity
!
r n
=
α mc n
2
, v n
=
α c
.
n
Here α = ke
2
/ c is a dimensionless number (the “fine-structure constant”) which is almost exactly 1/137. You are to use these facts and the Correspondence
Principle to derive the formula for
f 0
and the ground state energy of the atom.
a.
!
For radiation emitted in a transition from the ( n + 1) st state to the n th state, find the frequency
f n
in terms of the frequency
f 0
in the Bohr formula. What does this frequency approach for very large values of n ?
b.
!
Find the classical frequency of revolution of the electron in the n th state.
[Classical physics says this should the be frequency of the radiation.] c.
!
Using the Correspondence Principle, set these two frequencies equal to each other for very large n and evaluate
f 0
, showing that the ground state energy is E
1
= − 1
2
α 2 mc
2
. [Using mc
2 = 0.51
MeV for the electron, we find the ground state energy to be − 13.6
eV, in agreement with experiment]
PHY 142 !
Assignment 11 !
Summer 2013
9*.
!
A bead of mass m is constrained to slide back and forth on a frictionless wire of length L , attached at each end to a rigid wall. The wire runs along the x -axis, and collisions with the walls are elastic.
a.
!
Write the energy of the bead in terms of its momentum p . Ans :
E
= p
2
/2 m .
b.
!
Estimate the “uncertainty” Δ x in the bead’s position? Ans : Δ x = L . c.
!
Using this for Δ x , find the minimum value of Δ p x
. Ans : Δ p x
≥ /2 L .
d.
!
For a cycle of the motion p x
= 0 so the average of
p
2
is equal to ( Δ p x
)
2
.
Use this to estimate the minimum average energy. Ans : E ≥
2
8 mL
2
.
10.
!
A beam of electrons of momentum p moves as shown toward a narrow slit of width a . Leaving the slit, the beam spreads vertically, with angular half-width
δθ . You are to derive a formula for δθ , in terms of p , a and h .
a.
!
Find the deBroglie wavelength of the electrons, and treat the problem as one of Fraunhofer diffraction of a wave by a single slit.
[Where is the first minimum in the single slit pattern?] Ans : δθ ≈ h / pa .
b.
!
Use the uncertainty principle in the form Δ y ⋅ Δ p after the electrons passes through the slit. [Take y
Δ
= h to determine p y
≈ p y
.] p y
11*.
!
A particle confined to a certain space must have a minimum amount of kinetic energy, so it must be in motion. In the lowest possible energy state, use use Δ x ⋅ Δ p x
≥ , and use = 10
− 34 p x
= Δ p
(SI units) in the numerical calculations below.
x
, a.
!
Consider a particle of mass m confined between x = 0 and x = L . What is its minimum average speed v in the lowest energy state? b.
!
Evaluate this speed for a “classical” situation with m = 1 g and L = 10 cm.
c.
!
Evaluate the speed of an electron ( m atomic dimensions, L
≈ 1 nm.
!
≈ 10
− 30
kg) confined to a space of
PHY 142 !
Assignment 11 !
Summer 2013
!
12.
!
Materials used in buildings often contain uranium, which decays into radium, among other products. Radium decays ( τ ≈ 2300 yr) into radon. Radon decays
( τ ≈ 6 days) into polonium, emitting an α -particle. Radon is a gas, so it collects in closed spaces; if breathed its decay inside the body can cause damage. In such a closed space at t
= 0 let there be of of radon atoms at time t
N 1
radium atoms. We seek
. Let the average lifetimes be τ
1
N
and τ
2
2
( t ) , the number
, respectively.
a.
!
Show that the rate of radon buildup is given by dN
2
/ dt = N
1
/
τ
1
− N
2
/
τ
2
.
b.
!
Because τ for radium is so long, we can treat
N 1
as essentially constant.
With that assumption, show that the above equation, and the condition that
N 2
= 0 at t = 0, are satisfied by N
2
( t )
=
τ
τ
2
1
N
1
( 1 − e
− t /
τ
2 ) .
c.
!
What is the amount of radon in the space after time long compared to τ
2
?
13.
!
The isotope
14
6
C ( τ = 8270 yr) is formed in the atmosphere by interactions with cosmic rays. It combines with oxygen to form
CO 2
, as does ordinary (stable)
12
6
C . The known small fraction
f 0
of
14
6
C in atmospheric carbon remains constant over time. Plants use both forms of
CO 2
, so
14
6
C constitutes a fraction
f 0
of the carbon in plants. When plant material is used in non-living forms
(wood for furniture, etc.) replenishing of
14
6
C from the atmosphere stops.
You take a sample of pure carbon from a piece of wood which stopped growing T years ago. It contains N atoms, of which
N 14 rate R of these atoms, i.e.,
R
= − dN
14
/ dt .
are
14
6
C . You measure the decay a.
!
Express
N 14
in terms of τ and R .
b.
!
Express
N 14
in terms of
f 0
, τ , N and the age T of the wood. c.
!
Express T in terms of τ , N ,
f 0
and R . Ans : T = τ ln
⎛
⎝⎜ f
0
N
τ R
⎞
⎠⎟
.
PHY 142 !
Assignment 11 !
Summer 2013
!
!
14.
!
We have been calling τ the average lifetime of a decaying object, for which the number remaining at time t is
N
( t ) = N
0 e
− t / τ
. An alternate measure of lifetime is the half-life , which is the time it takes for half the sample to decay. You are to give further details about these times.
First, show that τ really is the average time a member of the sample lives. a.
!
Note that the probability of the object being “alive” at time t is
N
( t )/ N
0
= e
− t / τ
, so the probability of its decay between t and t proportional to this:
P
( t ) dt = Ae
− t / τ dt
+ dt is
. The total probability of decay at any time between 0 and ∞ must be 1. Set
∫
0
∞
P ( t ) dt = 1 and determine A .
b.
!
The average time at which the object decays will be the average of t over this probability distribution: t =
∫
0
∞ t ⋅ P ( t ) dt . Show that this gives t = τ .
Now relate this to the half-life.
c.
!
The half-life that τ
1
2
τ
1
2
is the time at which half of the objects have decayed. Show
= ln 2 ⋅ τ ≈ 0.693
⋅ τ .