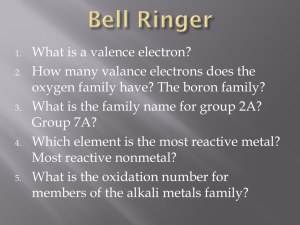Name:_____________
Chemistry 112
Final Exam
Section I . New Material Each questions worth 25 points- You may NOT skip any
questions from this section.
1. In the ground state of Cd
How many electrons occupy atomic orbitals with n=3?
How many electrons occupy orbitals where R=2
How many electrons occupy orbitals where R=1 & mR = -1
How many electrons have an ms of +1/2
How many electrons occupy an orbital where n=5 and & mR = -4
2. Write electron configurations fr the following atoms or ions:
N
S2-
Cu
An excited state of Na
Nd
2
3. Estimate ÄHRXN for the reaction K(s) + ½ Cl2(g) 6 KCl(s)
using the following data:
Lattice energy
-695 kJ/mol
Ionization Energy for K
419 kJ/mol
Electron affinity of Cl
-349 kJ/mol
Bond Energy of Cl2
239 kJ/mol
Enthalpy of sublimation for K
64 kJ/mol
4. For each of the following molecules or ions:
1. Provide a Lewis structure.
2. Give the arrangement of electrons in the molecule.
3. Give the molecular configuration of the molecule.
4. Tell if the molecule contains polar bonds.
5. Tell if the molecule is polar.
SF5
PO43+
AlCl3
NF3
3
Section II
Old Material
You may do any 4 questions. (All questions worth 25 points. - If you do more
than 4 questions, I will throw out the worst one(s).)
 0
0