Chapter 13 Properties of Gases
advertisement

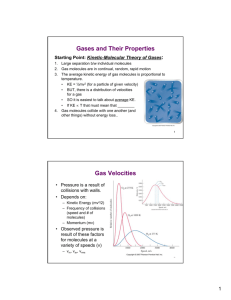
Chapter 13 Properties of Gases Many chemical insights are gained by studying the simplest chemical phase, gases 13-1 Gases Solid - fixed volume and shape Liquid - fixed volume but shape can change Gas - both volume and shape can change Will explore the other phases in chapter 15 A picture is worth a 100 words. Figure 13.1 volume of solids and liquids fixed because atoms/molecules in contact with each other (condensed state) Solids further fixed into array so cannot move, that is why shape fixed Liquids condensed, but not fixed so atoms/molecules can move, that is why volume not fixed Gases mostly empty space, molecules not touching so free to move as well as volume can change 13-2 Measurement of Pressure & 13-3 Atmospheric Pressure mixed together Gases are in constant motion and the molecules are colliding with each other as well as the wall of the container they are in. The collective force of these molecules hitting a surface is responsible for the pressure of a gas. Strictly speaking pressure is defined ad P = F/A where P is pressure, F is force, A is area In SI units force is measured in Newtons (1N 1 kg@m/s2) Roughly the force exerted by gravity on an apple at earth’s surface Area would be in m2 The SI unit of pressure is a Pascal (Pa) = 1 Newton/m2 This is a very small force spread out over a very large area, so it is a very small unit. It is so small that we usually don’t use it, and instead use the unit ‘bar’ where 1 bar = 10,000 Pa The air around us is a gas, and that gas exerts a pressure on every chemical experiment we do in the lab, so it is an important pressure to know. First, how do we measure it? This force can be easily measured with a barometer. At its simplest, a barometer is a tube filled with mercury that is turned upside down. When the 2 tube is turned upside down most people think intuitively that it is the vacuum in the closed end that holds the mercury in the tube. If fact you have it bassackwards. It is the pressure of the air on the bottom end of the tube that is pushing to keep the mercury in the tube. The height of the column of mercury supported by the pressure of a gas is directly proportional to the pressure of the gas pushing on the bottom of the tube. Thus one of the most common units used to express pressure is related to how high a column of mercury it supports. So you will see pressure expressed as mm of Hg, referring to the height of the mercury column. Notice this is not a measure of force/area, instead it is an easily measured number that is proportional to pressure. Another unit you will see is Torr, named after Torricelli, the man who invented the barometer. 1 mm of Hg = 1 Torr Key concept: So what is the pressure of the atmosphere around us? At sea level, on a clear day it is 760 mm Hg or 760 Torr or 1.01325 Pa or 1.01325 bar or 14.7 psi or 1 Atm. Example Problem 1. I have a vacuum system that measures pressures down to .025 mbar. Express these pressures in bar, mm Hg, Torr, atm and psi ,025 mBar x 1x10-3 bar = .025x10-3 bar 1 mbar ,025 mBar x 1x10-3 bar x 1 Atm 1 mbar 1.01325 bar x 760 mm Hg = .0188 mm Hg 1 Atm ,025 mBar x 1x10-3 bar x 1 Atm 1 mbar 1.01325 bar x 760 Torr = .0188 Torr 1 Atm ,025 mBar x 1x10-3 bar x 1 Atm 1 mbar 1.01325 bar ,025 mBar x 1x10-3 bar x 1 Atm 1 mbar 1.01325 bar = 2.47x10-5 Atm x 14.7 psi = 3.63x10-4 psi 1 Atm 3 Problem 2 The pressure at the center of the earth is estimated to be 350 Gpa. Express this pressure in bar, mm Hg, Torr, atm and psi 350 Gpa x 1x109 Pa x 1 bar 1 Gpa 10,000 Pa = 3.50x107 bar 350 Gpa x 1x109 Pa x 1 bar x 1 Atm 1 Gpa 10,000 Pa 1.01325 bar x 760 mm Hg = 2.63x1010 mmHg 1 Atm 350 Gpa x 1x109 Pa x 1 bar x 1 Atm 1 Gpa 10,000 Pa 1.01325 bar x 760 Torr = 1 Atm 350 Gpa x 1x109 Pa x 1 bar x 1 Atm 1 Gpa 10,000 Pa 1.01325 bar = 3.24x107 atm 2.63x1010 Torr 350 Gpa x 1x109 Pa x 1 bar x 1 Atm X 14.7 psi = 5.08x108 psi 1 Gpa 10,000 Pa 1.01325 bar 1 atm = 2.54x105 tons/sq in 13-4 Boyle’s and Charles’s Laws The first property of gases was discovered by Boyle in the 1660's What he found was that when the temperature is constant, when V8, P9 That is, pressure and volume are inversely proportional Draw figure like 13.8 on board Mathematically this can be expressed as V%1/P or V =c/P where c is a constant Going to our picture of a gas composed of particles bouncing around, and P coming from them bouncing off a surface Draw figure like 13.9 on board Makes sense, as volume increases the # of times a molecule hits the wall 9 to P9 Roughly 100 years later Jacques Charles uncovered the relationship that as T8,P8 or that T and P were directly proportional or that V%T Sketch a direct relationship on board Not only that, but when he worked at different pressures or with different gases all the line came to the same spot Figure 13.11 4 So with this series of experiment he discovered that all gases have zero volume at -273.15 oC. Why? Because at that temperature the gas molecules stop moving so they can exert pressure. He had uncovered absolute zero 13-5 Avogadro’s Law The final key to understanding gases, and indeed to understanding chemical and molecules came in the early 1800 When Gay-Lussac discovered that there was a direct relationship between volumes of gas reactants and products This, in turn was interpreted by Avogadro so say that any gas at a specific volume and temperature would contain the same number of molecules, and that V%n 13-6 Ideal-Gas Equation So at this point we have V%1/P V%T V%n Combining this together we have V%nT/P Putting in a proportionality constant V = K nT/P and multiplying both sides by P we have PV = K nT and giving K a special name and symbol: the molar gas constant R, we have the ideal gas law PV =nRT Key Equation: PV=nRT and R = .08206 L@atm/K@mol ( R has other values when P is measured in other units) Example problems A. Full blown Ideal gas problem ..... given any 3 of P,V,n,or T, calculate the missing variable. One of those big gas cylinders contains about 50L of gas. IF it was filled to 5 2,500 psi with N2 gas at 25oC, how much gas (in mole and grams and lbs) is in the tank First notice that this problem has P, V and T defined so we must be looking for n, the number of moles of gas PV=nRT Let’s rearrange the question to get n alone on one side n = PV/RT Before we fill in the blanks, what are the units of R .08206 L@atm/K@mol so V must be in L P in atm T in K So we have some conversions to do before we fill in the blanks P = 2500 psi x 1atm = 170 atm 14.7psi T = 25oC+ 173.15 = 298.15K n = (170 atm @ 50L)/ (.08206 L@atm/K@mol @ 298K) = 347 moles 347 mole x 2(14 g/mol) = 9732g 9732 g x 2.2046 lbs/1000g = 21 lbs of N2 gas B. Problems where 1 variable changes (and some of the other variables may not be given!) B.1 I fill a balloon with 1 L of helium for my daughter’s birthday on a day that is raining because with low pressure front just moved through so the air pressure is 736.6 mm of mercury. What is the volume of the balloon two days later when a high pressure front has moved through and its nice and bright and sunny, and the air pressure is 774.7 mm of Hg. Notice the set up, only two of the variables are given P and V. What do we do now? We assume the other variables are constant and don’t change. (So T is the same both days and n the # of moles of He is the same - nothing leaked out of the balloon) IN this case put all the variables on one side of the equation, and all the constants on the other 6 Variables Constants PV = nRT Constants are constants are the same both days so P*V = a constant so PV(day 1)=PV(day2) The starting volume was 1 l 1l x 736.6mm = V x 774.7mm V2 = V1 X 736.6/774.7 = .951 l, so my balloons are 95% of their original size - they have shrunk 5%! Notice I did NOT change pressure to atm. Why not? Didn’t use R to solve the problem Had same units on both sides of equation so canceled out! So it is easier to solve this way than a full blown PV=nRT B.2 Let’s continue with my daughter’s balloons. If a balloon contains 1 liter of helium when it is filled in our house at 65o F (291.5K). I put it in the car, in the sun, and the temperature gets up to 100oF (310.9 K). What is its volume now? PV=nRT Variables : V,T Constants: n,P Rearranging Variables Constants V/T = nR/P V1/T1 = V2/T2 Whenever you use T in a gas problem, you ALWAYS have to convert to K V1=1 Liter, T1 = 65o F =18.33oC = 291.48K), the other temp is V2 = X, T2= 100oF = 37.78oC = 310.93K V1T1 = b = V2/T2 and 1/291.48 = X/310.93 , X=1.07 L my balloon is another 7% larger. 7 B.3 Suppose we have 12.2 L of oxygen containing 0.5 mol of O2 at 11 atm and 25oC. IF we converted this to ozone (O3) what is the volume of the ozone (at same P and T)? 1st what is the balanced reaction? 3O2 62O3 Is this a redox reaction? (No, elemental O oxidation state 0) Variables: n,V Constants T,P PV=nRT Variables V/n = Constants RT/P V1/n1 = V2/n2 We have V1 and n1 What is n2? Molar conversion using stohiciometric coefficeints .5 mole O2 x 2 mol O2 = .333 mol O3 = n2 3 mol O3 12.2 L /.5 mole = X/.333 mol; X = 8.13L Does this make sense? We have a smaller number of moles, so the volume should have decreased and it did. I think 4 examples are enough Clicker questions Set up a word problem, identify the equation they need to solve it or set up some 3 of 4 problems with an error 13-7 STP and Determination of Molar Masses (I am deviating a little bit from the text) STP If we have 1 mole of an ideal gas at RT and 1 atm what is its volume? PV=nRT V = nRT/P =1(.08206)(273.2)/1.00 = 22.42 L 22.42L is a volume about 28x28x28 cm 8 The volume 22.42 L is called the molar volume Note the conditions of 1 atm and 273.2K are called standard temperature and pressure (STP), and this is a common reference for gases. Let’s check if some of our common gases are ideal gases at STP Gas Molar Volume (L) 22.397 O2 N2 22.402 H2 22.433 He 22.434 All above ideal NH3 22.079 Not ideal, about 2% low This confirms Avogodro’s hypothesis that a mole of each gas should occupy the same volume. Key Concept: 1 mole of an ideal gas has a volume of 22.42: @ STP Example calculation: What is the volume of 6 moles of H2 gas at STP The long way - use PV=nRT STP = 273.15K & 1 atm n=6 PV=nRT V = nRT/P = 6 @ .08206 @ 273 /1 = 134 L Quick and Dirty use 1 mole %22.42L @ STP, 1 mole %22.42 L 6 mol x 22.42L = 134L 1 mol Density and Molecular mass One very important feature of the ideal gas law is that it helps you to see te close relationship between a gas's density and it molecular weight. First, do you remember what density is? It is the mass per volume or: density=Mass/volume I just showed you that 1 mole of all gases, no matter what their molecular weight 9 will have the same volume Molar volume = 22.42 L If you think about it, the molecular mass of 1 mole of a gas depends on its chemical composition, thus H2 hydrogen gas has a molecular weight of 2.016 grams so its density is 2g/22.42 L, while CO2, a gas used to put out fires, has a molecular mass of 12.01 +16+16 or 44.01, and its density is 44.01/22.42 L. Let's see if we can use PV=nRT to make an equation about density. Let’s start with the term n, number of moles n=mass/molar mass Therefore PV=(mass/molar mass) RT Our equation for density is: density = mass/Vol So how do we rearrange the equation to look like this? We already have mass on the right-hand side of the equation, so we need to divide it by volume and then move molar mass, R and T to the left-hand side Thus we get P x Molar mass/RT = mass/vol = density -orMolar mass = density (RT/P) Key Equations: Molar mass = density (RT/P) density of a gas = molar mass x (P/RT) Let's try one example using this equation You may have heard (Or do as a demo?) that when you have your lungs full of helium you talk like Donald Duck. This is because He has a different density than the N2/O2 mix of our atmosphere, so your vocal cords vibrate differently and the sound waves are transmitted differently when the gas has a different density. Let’s calculate the density of He and N2 and compare them. Atomic mass of He= 4.003 Molar Mass = density (RT/P); Molar Mass X (P/RT) = density at STP P=1, T=273.2 d=Molar mass*1/.08206*273.2=0.18 g/L at STP (gram/mole) *(atm) * Kmol/Latm)* 1/K 10 What is the density of N2? 28.01/(.08206*273.2) = 1.25 g/L Now can you tell be why they use He to fill balloons? And CO2 to put out fires? 13-8 Partial Pressures and Mole Fraction Partial Pressure Now what happens if we have a mixture of gases? If you calculate the volume of an ideal gas, and compare it to the volume of the molecules in the gas, we will find that the gas molecules occupy a very small part (<.2%) of the volume For myself, class does not have to do: Volume of N2 molecule 65 A3, Volume of 1 mole of N2 = 65 x 6.022x1023 = 3.9x1025 A3 1 A = 1x10-8 cm so 1A3 = 1x10-24 cm3 = 1x10-24 mL 3.9x1025 x 1x10-24 = 39 mL Volume of 1 mole of N2 Gas = 22.42 L = 22420mL So 39/22420 = .0017 or .17% of gas volume is occupied by actual gas molecule! Bottom line. When you mix gases there is so much space, the gas molecules ignore each other so you can treat each gas in the mixture independently! So, if you have a mixture of two gases Key Equations: Pgas 1 = n1RT/V Pgas2 = n2RT/V and the pressure of the mixture = P1 + P2 We call the pressure of each individual gas the partial pressure. Mole Fraction Mole fraction is another way to measure the concentration of a material in a uniform mixture like a mixture of gases. The Definition of mole fraction is: Key Equation: Mole fraction (÷1) = n1/(n1 + n2 + n.....) = n1/ntotal 11 Notice that as a fraction the sum of the fractions in a whole =1 so in a mixture ÷1 + ÷2 +... =1. I talk about mole fraction right now because there is an very useful relationship between mole fraction and partial pressures of gases Going back to our equation on partial pressures Ptotal = P1 + P2 + ... = n1RT/V + n2RT/V .....= (n1 + n2 +...) RT/V =ntotalRT/V Let’s look at a single gas, P1. What is P1/ Ptotal ? =( n1RT/V ) / (n1RT/V + n2RT/V ....) But all the RT/V’s drop out so we have P1/Ptotal = n1/ntotal = mole fraction of gas 1! Key equation: P1/Ptotal = n1/ntotal = mole fraction Practice problems involving partial pressures and mole fractions: A typical problem starts with figure 13.20 400 mL 950 Torr O2 500 mL 700 Torr N2 Part 1. What is the total pressure inside the system when you open the stopcock? Treating each gas separately If you have 400 mL of O2 at 950 Torr and you let it expand into 500 mL of additional space PV=nRT Variables P,V, constant n,R,T PV = nRT =PV P1 V1 = P2 V2 P1V1/V2 = P2 V2 = the new TOTAL volume = 400 + 500 = 900 mL (950 Torr x 400 mL)/(900mL) PO2 = 422.2 Torr If you have 500 mL of N2 at 700 Torr and you let it expand into 400 mL of additional space PV=nRT Variables P,V, constant n,R,T 12 PV = nRT =PV P1 V1 = P2 V2 P1V1/V2 = P2 V2 = the new TOTAL volume = 400 + 500 = 900 mL (700 Torr x 500 mL)/(900mL) PN2 = 388.9Torr Ptotal = PO2 + NN2 = 422.2 + 388.9 = 811.1 Torr Part 2. What is the mole fraction of each gas in the mixture? ×O2 = PO2/Ptotal = 422.2/811.1 = .52 ×N2 = PN2/Ptotal = 388.9/811.1 = .48 Quick check does .52 + .48 = 1? Yes! So no obvious error Gases produced in the lab over water The book goes on to talk about the fact that if you make gas in the lab, you usually to it over water, so the gas you create has some water vapor in it that you have to account for in your calculations. Since we won’t do a lab like that, let’s skip it! 13-9 Maxwell-Boltzmann Distribution Throughout this chapter we have talked a gas as being a bunch of particles wizzing around in a large space. Before we develop this into a complete theory we need to think a little bit about the speed of the gas molecules. We can measure how fast a gas particle moves. We can actually measure the speed of gas molecules using a set up like the one shown in figure 13.22 Just like all the cars on the freeway don’t all go the same speed, all the molecules in a gas don’t all go the same speed. Instead there is a distribution of speeds. This distribution, called a Maxwell-Boltzmann distribution, is shown in figure 13.23 Notice two things. First, the distribution changes with T, and second it is not a nice symmetric distribution (Some of you may have had statistics, and worked with a Gaussian distribution that is nice and symmetric) In the next section we will talk about the Kinetic Energy of the gas molecules. How do you calculate kinetic energy? ½ mv2 13 I just showed you that gas molecules have a range of v’s. Which V should you use? What do I do when I have a range of grades on a test? I use a mean or an average. So that is what we will do in the next section, talk about the mean (average) KE and velocity of a distribution of gas molecules 13-10 Kinetic Theory and Root-Mean-Square Speed So let’s put all the pieces together and make a comprehensive theory that describes gases. This theory is called the kinetic theory of gases Key Theory: Kinetic theory of Gases 1. The molecules of a gas are in constant motion. They randomly collide with each other and with the walls of the container. (The random collisions with the container is what we measure as pressure) 2. All collisions are elastic, that is no energy is lost due to heat or friction 3. The average distance between the molecules is much larger than the actual size of the molecules 4. Any interactions between particles are negligible. That is there is no net attraction or repulsion between particles 5. The mean (average) kinetic energy of the molecules is directly proportional to the K temperature of the gas The last statement is very important. It says that K.E. %T Remembering that we have a range of kinetic energies we have: average kinetic energy = 3/2 RT now KE = ½ mV2 so if we know an average KE, we can also figure out an average velocity! Cutting out the math intermediates Key Equation: 14 Where vrms is the root mean square average velocity. What does root mean square actually mean? Well we have a distribution of velocities right? Now we square each velocity and get a new distribution of v2 Next we get the mean (average) of the v2 distribution Finally we take the square root of (the average of the v2 distribution) R is our gas constant, but here we are dealing with energy so we use the gas constant of 8.314 J/K@mol Mkg is the molar mass of the molecule IN KG (needs to match units in R) Sample calculation urms =(3RT/Mkg)1/2 M N2 =28.02 g = .02801 kg T=absolute =273.2 +25 =298.2 Plug and chug sqrt[3(8.3145kg (m/s)2/K mol)298.2 K/.0281 kg/mol] = 514.5 m/s 514.5 m/s = about 560 yd/sec = .32 mile/sec = 19 mi/min=1,145 mph!!!!! (You might compare this to the sped of a helium atom at 25 o C. Helium is much lighter (mm=4) so should it go faster or slower?) Another question - If a gas molecule is going 1000 mph. How come if somebody breaks a bottle of a smelly substance in the lab, it may take a minute or two before everybody is gassed out? The answer is to remember that there are lots of gas molecules in the air all bouncing into each other. Remember, What is molar volume at STP ? 22.42 L and how many molecules of air are there ? 6.02x1023 so that is 6.02/22.4 or 2.7 x1022 molecules/liter. That’s a lot of chances for collisions. In fact in a typical gas at STP an atom will only go 1x10-7 M before a collision (0.1 ìM) So the molecules are colliding with each other frequently and in each collision the atoms change both direction and speed sp it takes a long time to get across the room 13-11 Graham’s law of Effusion & 13-12 Mean Free Path Fun and interesting calculations, we can figure out that our N2 molecules @ STP actually goes about 63 nm before its first collision -see example 13.17 for details and that it collides about 8.1 x109 times /second! But nothing I want you to memorize, so let’s skip these sections 15 13-12 Van der Waals Equation An ideal gas is one that follows the ideal gas law, PV=nRT Most gases are ideal low pressures, say below 10 atm but what happens then? Figure 13.30 First, what is the Y axis? PV/RT From the ideal gas law PV=nRT PV/RT =n in this plot n=1, so if you are an ideal gas you follow the dashed line You can see that all the gases, even He are NOT ideal at high pressure. The deviations from the ideal gas can be traced back to a couple of postulates in our kinetic theory of gases Postulate 3. The average distance between the molecules is much larger than the actual size of the molecules At high pressures, we have so many gas molecules crammed into a small space that this postulate is no longer true. To correct for this we have to subtract the volume of the gas molecules from V. So the Vcorrected = V-nb where V = our original V n= # of moles b = is the volume of a mole of gas If our ideal gas law is PV=nRT we can throw in the above correction for V and get P(V-nb) =nRT The next problem with our kinetic theory is in the 4th postulate; Any interactions between particles are negligible. That is there is no net attraction or repulsion between particles There can be attractions between particles. If there is an attraction, this makes the molecules ‘stick’ together a little bit, and makes the pressure lower so Pcorrected = P - a(n/V)2 Where a is a empirical attraction factor. Why (n/V)2 ? n/V would be the concentration of one particle. Since this is an interaction between two molecules we have to deal with the concentration of both molecules so we have an (n/V)2 term 16 These corrections were formulated by Johannes van der Waals and put into a single equation called the vander Waals equation Key equation: To use this equation you need the two correction factors, a & b This is not an equation I expect you to memorize. Typical test questions I will ask on this section are: Given the above equation, and constants a & b, calculate a corrected pressure -orGiven the above equation explain what all the symbols mean -orExplain what is wrong with the kinetic molecular gas theory as originally written and explain how the above equation corrects these issues