Clifton E. Barry III, PhD , National Institutes of Health
advertisement

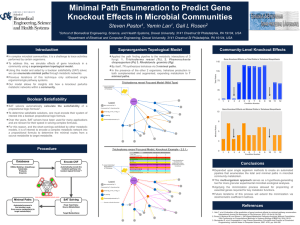
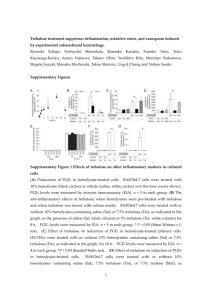
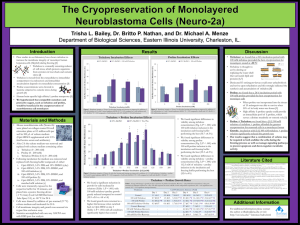
Clifton E. Barry III, PhD Tuberculosis Research Section National Institute of Allergy & Infectious Diseases , National Institutes of Health Thursday, February 18, 2016 11:00 AM Chemistry Room #1315 Exploiting the biochemistry of trehalose to develop real-time imaging probes for tuberculosis Mycobacterium tuberculosis is a devastating human pathogen that is globally responsible for 3 million deaths per year and infects one-third of the entire human population. The development of new drugs and vaccines is complicated by the century-old, poorly sensitive and poorly specific methods used for its detection in human lungs. The disaccharide trehalose plays an integral role in the biology of this organism, acting as both shuttle and destination for the very long chain mycolic acids that form the outermost layers of the outer cell envelope. The enzymes that trans-esterify mycolic acids on to, and off of, trehalose are three isoforms of the most abundant secreted protein in the cell known collectively as Antigen 85. We probed the selectivity of these enzymes using a library of trehalose analogs and identified positions on trehalose that could be substituted while still maintaining the ability to act as substrates for Antigen 85. Armed with that knowledge we synthesized and assayed 18F-trehalose analogs for use as real-time imaging agents using Positron Emission Tomography-Computed Tomography (PET-CT) in TB-infected animals and have shown that these specifically label viable bacteria. These probes appear highly specific for TB and their utility for monitoring response to treatment, vaccine efficacy and risk of progression to active disease after infection in humans are currently being explored.