A species specific satellite DNA family of Drosophila subsilvestris
advertisement

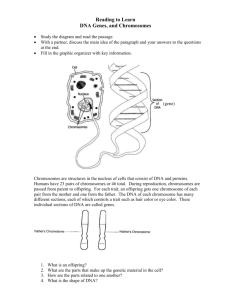
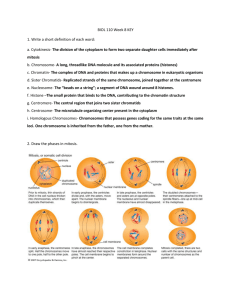
Chromosoma (1995) 103:539-544 CHROMOSOMA 9 Springer-Verlag 1995 A species specific satellite DNA family of Drosophila subsilvestris appearing predominantly in B chromosomes Jutta Gutknecht, Diether Sperlich, Lutz Bachmann Universit~t Ttibiugen, BiologischeFakult~t,Lehrstuhlftir Populationsgenetik, Auf der Morgenstelle28, D-72076 Ttibingen, Germany Received: 7 September 1994; in revised form: 1 November 1994 / Accepted: 2 November 1994 Abstract. This paper describes a species specific satellite DNA family (pSsP216) of Drosophila subsiIvestris, a palearctic species of the D. obscura group. The pSsP216 family consists of tandemly arranged 216 bp repetitive units that are predominantly localized on B chromosomes. These chromosomes appear in variable numbers in the karyotype of this species. Some pSsP216 repeats can also be detected in the centromeric heterochromatin of the acrocentric A chromosomes. Two strains, one with and the other without B chromosomes, were investigated for sequence variability and for the location of this satellite DNA on the chromosomes. Among 16 clones of the 216 bp basic repeat unit an overall similarity of about 93% and no strain specific differences were found, indicating that the B chromosomes may have derived from the A chromosomes (probably the dots) by spontaneous amplification of the pSsP216 satellite DNA family. Introduction The term "B chromosome" was introduced by Randolph (1928) in order to distinguish these small and morphologically specific chromosomes from the typically bigger "A chromosomes" in the chromosome complement of maize. B chromosomes are usually not necessary for cellular functions (Shaw and Hewitt 1990). During mitosis and meiosis these chromosomes are distributed to daughter cells by random segregation, and their number can, therefore, vary from individual to individual of the same species (Shaw and Hewitt 1990). The presence of B chromosomes in chromosome sets is a rather widespread phenomenon, especially in plants (Puertas et al. 1987; Staub 1987). There are several examples known in insects (Shaw et al. 1985), but so far only one single case for the genus Drosophila, i.e. DroEdited by: E.R. Schmidt Correspondence to: L. Bachmann sophila nasura albomicans (Ramachandra and Ranganath 1987; Hatsumi 1987). The origin of B chromosomes is still mainly speculative, e.g. they may be derivatives of X chromosomes, or telocentric fragments arising through the mispairing of centromeres (Jones and Rees 1982). There is also very little known about possible effects of B chromosomes. Positive effects seem to be scarce; many deleterious effects on the fitness/survival of plants and insects have been observed (Jones and Rees 1969; Hewitt 1979; Beukeboom and Werren 1993). By means of specific staining techniques it is possible to demonstrate that B chromosome are basically heterochromatic and that they might consequently consist to a large degree of highly repetitive satellite DNA. Here we describe the identification of supernumerary B chromosomes in D. subsilvestris, a palearctic species of the D. obscura group. Its haploid karyotype has been described as consisting of only two metacentric, one submetacentric, two acrocentric and one dot chromosomes (Lakovaara and Saura 1982). In the present study we will show that supernumerary B chromosomes can be found in strains directly derived from nature. Since this is a unique situation in the D. obscura species group a molecular analysis was performed in addition. Materials and methods Drosophila strains. Two different strains of D. subsilvestris were used. Both strains were established as mass cultures from wild flies caught in wooded areas near Ttibingen, Germany. Strain A was kept in the laboratory from 1978 and died out in 1992; strain B was established in autumn 1992. The strains have been kept in the laboratory in continuous light and at a constant temperature of 18 ~ C. DNA isolation and cloning. Genomic DNA of Drosophila was extracted as described by Preiss et al. (1988). highly repetitive DNA was isolated from satellite DNA bands visible after 5% polyacrylamide gel electrophoresis of PvulI digested genomic DNA. The DNA fragments were eluted from the gel, purified and ligated into the plasmid pUC19 (King and Blakesley 1986), and transformed 540 Fig. 1A, B. Mitotic chromosome sets of Drosophila subsilvestris from strain A (A) and at a higher magnification from strain B. (B) The preparations were made shortly after establishing the strains in 1978 (A) and 1992 (B). Arrows indicate the supernumerary B chromosomes. Magn. 1000 x into Escherichia coli JM103 cells. Recombinant clones were selected as white colonies on ampicillin plates containing X-gal (5bromo-4-chloro-3-indolyl-fl-D-galactoside) and IPTG (isopropylfl-D-thiogalactopyranoside). DNA sequencing. The nucleotide sequences were determined by the dideoxy chain termination method (Sanger et al. 1977) using the USB sequenase kit (No. 70770) for 35S radioactive sequencing and the Autoread kit (Pharmacia No. 27-1690-04) for automatic sequencing on an A.L.E (Pharmacia). DNA for the sequencing reactions was prepared according to Lee and Rasheed (1990). Filter hybridization. Fragments of digested DNA were separated by agarose gel electrophoresis and transferred to Hybond N membranes (Amersham) by the method of Southern (1975). Labeling of probe DNA with Digoxigenin and signal detection were carried out according to the instructions accompanying the "DIG DNA labeling and detection kit-nonradioactive" (Boehringer No. 109 36 57) and according to Kreike et al. (1990). Preparation of mitotic chromosomes. Neural ganglia of third instar larvae were dissected in Drosophila Ringer (5.55 g NaC1, 0.22 g KC1, 0.44 g CaC12, 1000 ml distilled water) and then treated for 20 min in a hypotonic solution of 1% sodium citrate. After 10 rain fixation in a cold 3:1 ethanol:acetic acid mixture the ganglia were resuspended in a drop of 45% acetic acid and spread onto a clean slide prewarmed to 42 ~ C. After drying, the chromosomes were dehydrated in ethanol (70%, 90%, 100%) and then air dried. In situ hybridization of chromosome preparations. Fifteen microliters of a 50% formamide, 5% dextran sulfate, 4xSSC and DIG-labeled DNA (1 ng/gl) mixture were placed on a slide, covered with a coverlip and sealed with rubber cement. (lxSSC is 0.15 M NaC1, 0.015 M sodium citrate). The probe and the chromosomal DNA were denatured simultaneously at 80 ~ C for 10 rain. In situ hybridization was performed overnight in a humid box at 37 ~ C. Unspeciflcally bound DNA was removed by washing the slides in 2xSSC for 5 min at room temperature, twice for 10 rain at hybridization Fig. 2. Part of a set of polytenic chromosomes of D. subsilvestris from larval salivary gland tissue. The degree of polytenization of the dot chromosome (indicated by an arrow) is the same as that of the other chromosomes. Magn. 500 x temperature and once more for 5 min at room temperature in 2xSSC. Subsequently, the slides were washed briefly in 100 mM Tris-HC1, 150 mM NaC1, 0.3% Tween 20, pH 7.5 and preincubated in 100 mM Tris-HC1, 150 mM NaC1, 0.3% Tween 20, pH 7.5 containing 0.5% blocking reagent (Boehringer) at 3 7 ~ for 30 min. The anti-DIG-peroxidase conjugate (in 100 mM Tris-HC1, 150 mM NaC1, pH 7.5 containing 0.5% blocking reagent) was placed on the slide with the chromosomes and incubated for 45 min at 37 ~ C followed by three washes in 100 mM Tris-HC1, 150 mM NaC1, pH 7.5. The detection was done in 50 mM Tris-HC1, 0.05% dimninobenzidine, 0.01% H202 pH 7.5 for 10 min in the dark. The slides 541 1 Ii 21 31 41 51 pSsP216 /i CAGCTGTATT CAGCGGACCA AACCAGGAGT CAACCCCGAA GCAACGGTCA CGGACAGAGA pSsP216 /2 ................................................ pSsP216 /3 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . pSsP216 /4 ............................................ pSsP216 /5 ........................................................ pSsP216 /6 ............................................................ pSsP216 /7 ............................................................ pSsP216 18 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . pSsP216 /9 ............................................ pSsP216 /i0 ............................................................ pSsP216 /ii ............................................ . . . . . . . . . . . . . . . . . . . . T ............... T--- c .................... T ............... T ............... ..................................... A ...... pSsP216/13 ..................................... A .................. pSsP216/14 ............................................................ 61 71 81 91 i01 iii pSsP216/l CTTTGGGAGC GGCAACCACA CCCAGTGTGC CAAACCAGGA GCCTCATGCT CAGACAACTT pSsP216 '12 T ........... c pSsP216/2 ...... pSsP216/3 ..... TC ...... pSsP216/4 ..... TC ................................ pSsP216/5 ...... C .............. pSsP216/6 ...... C .......................................... pSsP216/7 ...... pSsP216/8 ..... TC ................................ pSsP216/9 T .... pSsP216/10 ...... pSsP216/II T ............... C .......................................... T ......................... T---G T--- C .......... T ......... C .......... T ......... C .......... ....................... C .......... C .......... C .......... C .......... TC .......................................... C .......... C .......................................... C .......... T .... TC .......................................... C .......... pSsP216/12 ..... TC ................................ C .......... pSsP216/13 ...... C .............. C .......... pSsP216/14 ...... C .......................................... C .......... C .......................................... T---G T ......... T ......... ....................... 121 131 141 151 161 171 pSsP216/I CAC*GGGCAG GAACTTTGGC AAAGACGGCC AACAACAGCG CGTCAAACCA GGAGTCACCT pSsP216/2 ---T .............. pSsP216/3 ---* ......... pSsP216/4 ---* ................. pSsP216/5 ---* .............. pSsP216/6 ---* ................. G ................ pSsP216/7 ---* ................. G ................ pSsP216/8 ---* ......... G .......... pSsP216/9 ---* ................. pSsP216/lO ---* . . . . . . . . . . . . . . . . . pssP216/ll ---* . . . . . . . . . . . . . pSsP2t6/12 ---* . . . . . . . . . . . . . . . . . G. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . pssP216/13 ---* . . . . . . . . . . . . . . . . . G. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . wrisks pssP216/14 ---*--c G. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . increase sequence similarity TT G .... A ....... GA ................................. G .......... A---G G .............. TT ........................ G ....................... -C ...................................... T ....... T ..................... T ..................... T ................. A ......... G ...................................... A-- G. . . . . . . . . . . . . . . . T- -A . . . . . . . . . . . . . . . . . . -G. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 181 191 201 211 pSsP216 /i GCAACACCAC GATCCCAGGC AAAGATTTTG GCAAAGG /2 ......................... C ........... /3 ....... C . . . . . . . . . . . pSsP216 /4 . . . . . . . . . . . . . . . . . . . . . . . . . pSsP216 /5 . . . . . . . . . . . . . . . . . . . . . . . . . c-- pSsP216 /6 ............... C ....... pSsP216 /7 ....... pSsP216 /8 ......................... C ........... pSsP216 /9 ......................... C ........... pSsP216 /i0 ......................... C ........... pSsP216 /ll ......................... C ........... pSsP216 /12 ............. pSsP216 /13 ......................... C ........... pSsP216 /14 ................ C ........... T ......... T ................. A ........... * ........ indicate gaps introduced to Results pSsP216 pSsP216 A . . . . . . . . . . . . . . . . . AT--- Fig. 3 . N u c l e o t i d e s e q u e n c e s o f pSsP216/1-14~om D. subsilvestris aligned withpSsP216/1. As- c . . . . . . . . . . . . . . . . . . . . G--- C ........... C ........... were air dried and analyzed microscopically with reflection contrast (Landegent et al. 1985). Preparation of salivary gland chromosomes. The usual squashing method for polytene chromosomes of Drosophila was used (Ashburner 1989). The chromosomes were stained in a 60% acetic orcein solution. The configurations of the mitotic chromosome sets of D. subsilvestris (strains A and B) are shown in Fig. 1. They consist of two pairs of acrocentric chromosomes, two pairs of metacentric chromosomes (one of them is the X chromosome) and one pair of submetacentric, F-shaped chromosomes. In addition a variable number (one to five) of dot-like chromosomes was observed in most of the chromosome sets studied. With respect to these dotlike chromosomes we found differences between strains A and B. Up to five dot-like chromosomes appeared in chromosome preparations prepared shortly after strain A had been established from wild flies; only one or none at all were found after several years of laboratory maintenance. In the newly established strain B, however, a variable number of dot-like chromosomes was again present in mitotic metaphases. In preparations of polytene chromosomes (Fig. 2) from salivary glands, however, only 542 one small banded dot chromosome could be seen irrespective of the number of dot-like chromosomes appearing in the metaphases. From this observation it was concluded that the supernumerary dot-like chromosomes of D. subsilvestris must be B chromosomes. B chromosomes are absent in almost all chromosome sets of Drosophila. So far, they have only been observed in D. nasuta albomicans (Ramachandra and Ranganath 1987; Hatsumi 1987). Since only one polytene dot chromosome with a normal degree of polytenization occurred in each salivary gland nucleus it was further assumed that the supernumerary B chromosomes are heterochromatic and contain repetitive sequences that are underreplicated and hence not visible in polytenic nuclei (Zacharias 1993). To obtain more information, a search for repetitive sequences in the genome of D. subsiIvestris was performed. Genomic DNA of the two strains (A, B) of D. subsilvestris was digested with a number of different restriction endonucleases and subsequently electrophoresed on a 5% polyacrylamide gel; only PvuII and HaeIII digested DNA, respectively, gave the ladder-like pattern that is typical of tandemly arranged satellite DNA. The DNA of the smallest PvuII restriction satellite fragment (about 200 bp) was subsequently eluted from the gel and ligated into the plasmid vector pUC 19. After transformation seven positive clones were identified from each strain and sequenced: pSsP216/1-7 from population A and pSsP216/8-14 from population B. All sequences had a length of 216 bp, apart from clones pSsP216/2 and pSsP216/14, which were 217 and 215 bp in length, respectively. The sequences showed an average homology of 96.35% (93.52%-99.54%) and were thus considered as members of the pSsP216 satellite DNA family (Fig. 3). The pSsP216 repeats are slightly GC rich (55%) and exhibit a remarkably asymmetric base composition, especially with respect to A and T nucleotides (A, 31.5%; C, 30.5%; G, 25.5%; T, 12.5%). As expected from the electrophoresis data all PvuII repeats have an internal HaeIII restriction site (Fig. 3, position 147-150). The analyzed pSsP216 sequences from the two different lines of D subsiIvestris exhibited no strain specific variation. An EMBL data bank search revealed no significant similarity of pSsP216 sequences to any other known nucleotide sequence. In order to test whether the sequences of the psSp2167 family are also present in the genomes of other species of the D. obscura group, genomic DNA of D. madeirensis, D. obscura, D. ambigua, D. tristis, D. subobscura, D. pseudoobscura, D. bifasciata and D. subsilvestris (strains A and B) was digested with the restriction endonuclease PvuII, electrophoresed on a 1% agarose gel and blotted. The hybridization with labeled pSsP216/3 DNA revealed that the ladder-like pattern of typical satellite DNA occurred only with DNA from D. subsilvestris. A significant hybridization signal could be observed with DNA fragments of high molecular weight from D. ambigua. Interestingly, the hybridization signal with DNA from strain A was much weaker (i.e. almost invisible) compared with that with DNA from strain B, although almost the same amount of genomic DNA was Fig. 4. In situ hybridizationof clone pSsP216/3 to mitotic chromosomes of D. subsilvestris (strain B) larvae. Hybridizationsignals are visible on the centromeres of the acrocentric chromosomes (1), on the dot chromosomes (2) and on the B chromosomes (3). Magn. 1000 x used. This was completely unexpected as almost no difference in the intensity of the -200 bp PvuII restriction satellite band on 5% polyacrylamide gels was observed between strain A and B. The solution to this discrepancy was that genomic DNA of strain A was used that came from two different preparations: the DNA of strain A used for the identification of the psSp216 satellite DNA by polyacrylamide gel electrophoresis was prepared in 1986 when strain A had been kept for 8 years in the laboratory and the DNA of stock A used for the Southern blot analysis was prepared in 1992 after 14 years of cultivation of strain A. Therefore, most of the pSsP216 sequences were probably eliminted from the genome of strain A during this period of cultivation. This was also the case for the B chromosomes. A rough quantification of the contribution ofpSsP216 sequences to the genome of D. subsilvestris was performed by means of dot-blot hybridizations using cloned pSsp216/3 DNA and genomic DNA from strain B. The resulting estimate of 0.5%-1% as an average proportion of pSsP216 sequences in the genome (data not shown) is of course only valid for the specific strain B in summer 1993, when the genomic DNA was prepared. For chromosomal localization of the pSsP216 sequences in situ hybridizations to mitotic chromosomes prepared from larvae of strain B were carried out, using clone pSsP216/3 as a probe. Weak hybridization signals were visible with reflection contrast microscopy on the centromeres of the acrocentric chromosomes and strong ones on the dot chromosomes. In addition, extremely strong signals appeared that were so intense that it was difficult to associate them with any underlying chromo- 543 somal structure. Therefore, chromosomes of the same slides were stained with Giemsa and inspected in the normal light microscope. A comparison showed that all structures that produced intense hybridization signals were dot-like B chromosomes (Fig. 4). Discussion In this study a species-specifically amplified satellite DNA family pSsP216 could be detected. It represents a major component of additional B chromosomes of the chromosome complement of D. subsilvestris. Copies of the same family were also found in the centromeric heterochromatin of the two acrocentric chromosomes and on the dot chromosomes. The pSsP216 satellite DNA family shows all the characteristics of a typical satellite DNA: a repetitive unit of discrete length (216 bp), high intraspecific sequence homogeneiy (96.35%) and a clear tandemly repeated arrangement in the genome. B chromosomes and dot chromosomes cannot be distinguished in the microscope without specific staining techniques. One pair of dot chromosomes appears in all karyotypes of the species of the D. obscura group. Cbanding techniques showed that these dots are rather heterochromatic in all the species of the D. obscura group (Raab 1988). The dot chromosomes of D. subsilvestris stained with the fluorescent dyes 4,6-diamidino2-phenylindole (DAPI)/Chromomycin A 3 (CMA) exhibit GC-rich DNA in the centromeric and AT-rich DNA in the distal regions, a pattern that was not observed in dot chromosomes of other D. obscura group species (Raab 1988) at all. Clusters of the slightly GC-rich pSsP216 sequences (55%) are probably limited to the centromeric region of the dot chromosomes. However, if additional B chromosomes are present, extremely intense hybridization signals occur. We therefore believe that the B chromosomes contain either very big clusters of amplified pSsP216 sequences or that they are entirely composed of them. Based on these results we suggest that the B chromosomes may have originally arisen from the dots. However, dot chromosomes behave like the other normal A chromosomes during meiosis while the B chromosomes seem to segregate randomly, leading to different B chromosome numbers in different individuals (varying at least from zero to four, according to our observations). The mechanism responsible for the origin of B chromosomes in D. subsiIvestris might be inferred from the experiments of Haaf et al. (1992). After transformation of in vitro cultured African green monkey cells with human alpha satellite sequences they found that clusters of human alpha satellite sequences of approximately 15 kb were amplified in some instances up to 200 kb. Furthermore, so-called double minutes appeared and persisted after transformation over several cell generations. The double minutes exhibited "normal" replication and segregation behavior. In situ data showed that these double minutes consisted of human alpha satellite DNA. Although speculative, it appears possible to explain the ori- gin of B chromosomes of D. subsilvestris in a similar way by extrachromosomal amplification of pSsP216 sequences. If so, we have to conclude that the nucleotide sequence of pSsP216 repeats and their tandemly repeated organization carry information necessary for establishing a chromosome-like structure in combination with specific proteins - at least a structure that fulfills the requirements of a centromere/kinetochore. The presence of pSsP216 sequences in the centromeric regions of the two acrocentric chromosomes and the normal dot chromosomes may be taken as support for the assumption that clusters of pSsP216 sequences might possess some centromere-like properties. The problem with this hypothesis is that a structure that only contains amplified pSsP216 repeats lacks telomeres and should, therefore, degenerate. If we assume continuous elongation of the pSsP216 arrays by an amplification mechanism on the one hand and continuous reduction in length due to the lack of telomeres on the other an equlibrium situation could be imagined. Therefore, it may be possible to achieve an almost constant size of B chromosomes. In comparison with other satellite DNAs described so far for D. obscura group species, the pSsp216 repeats reveal no peculiarities that would classify them as an extraordinary satellite DNA. Certainly, the GC content of roughly 55% is slightly though not remarkably higher than that of other satellite DNA families of the centromeric heterochromatin, i.e. 27% in the KM190 family of D. kitumensis and D. micJvlabis (Bachmann et al. 1992) 30% in the pGH290 family of D. guanche (Bachmann et al. 1989), and 40%-45% in the ATOC180 family of D. ambigua, D. tristis and D. obscura (Bachmann and Sperlich 1993). On the other hand, the observed average variability of 3.65% between repeats is smaller than in any of the other satellite DNAs of the other species of the group, i.e. 5%-6.7% in the KM190 family, 11.3% in the pGH290 family, and 7%-15% in the ATOC180 family. Computer simulations show that such a low value of variability between repeats may be the result of a rather high recombination rate (Stephan and Cho 1994). Furthermore, the model of Stephan and Cho (1994) predicts that increasing rates of unequal exchanges lead to lower copy numbers per cluster. Indeed, the copy number of pSsP216 repeats seems to be low compared with those of the KM190, ATOC180 and especially the pGH290 repeats if we exclude the B chromosomes. However, the data are difficult to compare for two reasons: (i) in addition to general methodological problems in the estimation of copy numbers per genome, the obtained values for the pSsP216 repeats are only of limited value owing to the variable number of B chromosomes and (ii) the KM190, ATOC180 and pGH290 satellite DNAs were detected in the centromeric heterochromatin of all chromosomes (except the Y) of the respective species. Therefore, it may be possible that the copy numbers per gehome are higher but that the actual sizes of the tandemly arranged arrays are smaller. Copies of the pSsP216 satellite DNA family derived from B chromosomes should in any case be homogeneous if the origin of B chromosomes is spontaneous extrachromosomal amplification of a chromosomal template. 544 Acknowledgements. We thank Michael Raab-Vetter for technical instructions for the in situ hybridization experiments. We also thank Wolfgang Stephan for valuable suggestions for improving the manuscript. References Asburner M (1989) Drosophila: A laboratory manual. Protocol 17 Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY Bachmann L, Sperlich D (1993) Gradual evolution of a specific satellite DNA family in Drosophila ambigua, D. tristis and D. obscura. Mol Biol Evol 10:647-659 Bachmann L, Raab M, Sperlich D (1989) Satellite DNA and speciation: a species specific satellite DNA of Drosophila guanche. Z Zool Syst Evol-Forsch 27:84-93 Bachmann L, Mtiller E, Cariou ML, Sperlich D (1992) Cloning and characterization of KM 190, a specific satellite DNA family of Drosophila kitumensis and D. microlabis. Gene 120: 267-269 Beukeboom LW, Werren JH (1993) Deletion analysis of the selfish B chromosome, paternal sex ratio (PSR), in the parasitic wasp Nasonia vitripennis. Genetics 133:637-648 Haaf T, Warburton PE, Willard HF (1992) Integration of human alpha-satellite DNA into simian chromosomes: Centromere protein binding and disruption of normal chromosome segregation. Cell 70:681-696 Hatsumi M (1987) Karyotype polymorphism in Drosophila albomicans. Genome 29:395400 Hewitt GM (1979) Orthoptera: Grasshoppers and crickets. Animal Cytogenetics 3: Insecta 1. Borntr~iger, Berlin Jones RN, Rees H (1969) An anomalous variation due to B chromosomes in rye. Heredity 24:265-271 Jones RN, Rees H (1982) B chromosomes. Academic Press, New York King PV, Blakesley RW (1986) Optimizing DNA ligation for transformations. Focus 8:1-3 Kreike CM, de Koning JRA, Krens FA (1990) Non-radioactive detection of single-copy DNA-DNA hybrids. Plant Mol Biol Rep 8:172-179 Lakovaara S, Saura A (1982) Evolution and speciation in the Drosophila obscura group. In: Ashburner M, Carson HL, Thomp- son JN Jr (eds) Genetics and biology of Drosophila, vol 3b. Academic Press, New York London, pp 1-59 Landegent JE, Jansen in de Wal N, van Ommen GJB, Baas F, de Vijlder JJM, van Duijn P, van der Ploeg M (1985) Chromosomal localization of a unique gene by non-autoradiographic in situ hybridization. Nature 317:175-177 Lee S, Rasheed S (1990) Mini-screening of recombinant bacteria. Bio Techniques 9:676-679 Preiss A, Hartley DA, Artavanis-Tsakonas S (1988) Molecular genetics of enhancer of split, a gene required for embryonic neural development in Drosophila. EMBO J 12:3917-3927 Puertas MJ, Ramirez A, Baeza F (1987) The transmission of B chromosomes in Secale cereale and Secale vavilovii populations. II. Dynamics of populations. Heredity 58:81-86 Raab M (1988) Untersuchung des Heterochromatins durch B~inderungstechniken und cytologische Lokalisierung von hochrepetitiven DNA-Sequenzen in Artender Drosophila obscura Gruppe. Diploma thesis, University of Tiibingen Ramachandra NB, Ranganath HA (1987) Characterization of heterochromatin in the B chromosomes of D. nasuta albomicans. Chromosoma 95:223-226 Randolph LF (1928) Types of supernumerary chromosomes in maize. Anat Rec 41:102 Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463-5467 Shaw MW, Hewitt GM (1990) B chromosomes, selfish DNA and theoretical models: where next? In: Futuyama D, Antonovics J (eds) Oxford surveys in evolutionary biology, vol 7. Oxford University Press, Oxford, pp 197-223 Shaw MW, Hewitt GM, Anderson DA (1985) Polymorphism in the rates of meiotic drive acting on the B chromosome of Myrmeleotettix maculatus. Heredity 55:61-68 Southern EM (1975) Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98:503-517 Staub RW (1987) Leaf striping correlated with the presence of B chromosomes in maize. J Hered 78:71-74 Stephan W, Cho S (1994) Possible role of natural selection in the formation of tandem-repetitive noncoding DNA. Genetics 136:333-341 Zacharais H (1993) Larger nuclei in the larval brain of Drosophila nasutoides often shows underreplication, whereas metaphases provide a reliable DNA standard. Genome 36:294-301