Photoacoustic Measurement of Bandgaps of Thermoelectric Materials 15
advertisement

Photoacoustic Measurement of Bandgaps
of Thermoelectric Materials
by
MASSACHUSETTS
INSTITUTE
OF TECHNOLoG~Y
15 2O1,
George Ni
AU 15 20iMR
Submitted to the Department of Mechanical Engineering in partial
fulfillment of the requirements for the degree of
Master of Science in Mechanical Engineering
at the
MASSACHUSETTS INSTITUTE OF TECHNOLOGY
May 2014
C Massachusetts Institute of Technology 2014. All rights reserved.
Signature redacted
A uthor..........................
..............................
Department of Mechanical Engineering
May 19,2014
Signature redacted
C ertified by.............
...............................
Gang Chen
Carl Richard Soderberg Professor of Power Engineering
.e-00)
-
Signature redactedh
hesis Supervisor
S
Accepted by.........................
David E. Hardt
Chairman, Department Committee on Graduate Students
1
2
Photoacoustic Measurement of Bandgaps
of Thermoelectric Materials
by
George Ni
Submitted to the Department of Mechanical Engineering on May 19, 2014,
in partial fulfillment of
the requirements for the degree of
Master of Science in Mechanical Engineering
Abstract
Thermoelectric materials are a promising class of direct energy conversion materials,
usually consisting of highly doped semiconductors. The key to maximizing their thermal
to electrical energy conversion lies in optimizing three inter-related material properties,
thermal conductivity, electrical conductivity, and Seebeck coefficient.
All three
properties are affected by the carrier concentration of the thermoelectric material. In
practice, tedious trial-and-error testing is needed to determine the optimal carrier
concentration for the maximum figure-of-merit, ZT. Theory and computer simulations of
thermoelectric properties can aid the determination of new thermoelectric materials, but
several challenges remain. The bandgap is a key piece of bandstructure information, but
is difficult to determine for heavily doped thermoelectric materials. Under heavy doping
conditions, the effective mass and bandgap both change due to the formation of Urbach
band tails and other defect states within the bandgap. Furthermore, bandgaps of heavily
doped materials are difficult to observe optically, due to significant amounts of carriers in
defects states within the bandgap. Conventional optical measurement techniques relying
on transmittance change require extremely thin samples, on the order of microns for
thermoelectrics. Photoacoustic spectroscopy is used in this work to optically probe the
thermoelectric bandgap, without the need to produce thin samples. Photoacoustic
spectroscopy allows simultaneous measurements of the thermal conductivity and optical
absorption coefficient. In this work, a relative measurement is devised to reduce the need
to carefully control experimental parameters such as light input and microphone gain.
Semiconductor theory is discussed to account for the band-filling effects, and a method is
proposed to extrapolate the true electronic bandgap from the Burstein-Moss shift of the
absorption edge due to heavily doping.
3
4
Dedication
To always striving to overcome life's obstacles, and seeing the positives learned in every
stumble.
5
6
Acknowledgements
No single endeavor is accomplished without the support of numerous others. It is not
until I reached this point in my thesis writing that I realized the Acknowledgements
section means far more to the author than the reader. I had assumed that these sections
were written out of courtesy. How wrong I was. I would like to thank Professor Gang
Chen for his continuing support, through the many difficulties I faced at MIT. He has
shaped me as a scientist and engineer, and I will always treasure the personal relationship
we shared. The opportunity to work at MIT is one that I will keep with me forever.
To my parents, I would like to thank them for the quarter century that they spent to
ensure that I turn out ok. My father became a role model to me through his hard work
and compassion for his students. My mother spent countless hours homeschooling me
after public school, and shuttling me to and from numerous extra curricular activities.
Above all, they taught me the importance of integrity. Without their guidance, I would
not be the person I am today.
I could not have come this far without the insightful discussions with the members of the
Nanoengineering group. I learned a great deal, both technical and professional, from the
colleagues I was surrounded by. Being in the same year, Bolin Liao, Wei-Chun (Edi)
Hsu, and Lingping Zeng helped me a great deal with academic discussions. Dr. Selcuk
Yerci and Dr. Hadi Ghasemi both enabled me to grow immensely through their
mentorship. I can only hope to continue to have such mentors in the futures. Further, I
thank Sangyeop Lee, Keivan Esfarjani, Mona Zebarjadi, Daniel Kraemer, Ken McEnaney,
Kimberlee Collins, Jonathan Tong, and Matthew Branham for technical and nontechnical discussions. To Zhiting Tian, Shuo Chen, Sheng Shen, Tengfei Luo, Nuo Yang,
Anurag Bajpayee, Jianjian Wang, John Cuffe, Amy Marconet, and many others, I thank
for making my first time away from home less lonely than it could have been. Without
the support, I would not have been able to put my energy into my academic work.
To my wife, Wenjia Xu, I find myself thankful everyday to have met someone patient
enough to wait while I pursue my dreams. Her unwavering support keeps me grounded
in times of uncertainty, and allows me to go farther than I could alone.
7
8
Chapter 1:
Introduction...........................................15
1.1
Energy Usage ..............................................
15
1.2
Thermoelectric Materials...........................................
16
1.3
Three Therm oelectric Effects ..........................................................................
16
1.4
Therm oelectric Perform ance.............................................................................
17
1.5
Improving Thermoelectric Performance...........................................................
18
1.6
Skutterudites ...............................................
19
1.7
Bandgap M easurem ents ...................................................................................
22
1.8
Photoacoustic Spectroscopy.........................................
24
1.9
Fourier-Transform Spectroscopy ....................................
25
1.10
Thesis O verview ............................................................................................
Chapter 2:
2.1
Photoacoustic Spectroscopy.............................................
Rosencwaig-Gersho Theory.....................................
28
29
29
2.1.1
Heat-Flow and Temperature Profile ..........................................................
30
2.1.2
Acoustic Signal ..........................................
34
2.2
Photoacoustic Response - Sensitivity...............................................................
35
2.3
Photoacoustic M odel of InSb...........................................................................
38
2.4
Q uantitative M easurem ents .............................................................................
39
2.5
Experim ental Setup ....... ..............................
40
2.6
M easurement of Indium Antimonide..................................................................
43
2.7
M easurem ent of Indium Arsenide .....................................................................
44
2.8
Measurement of Undoped Skutterudites................................
46
2.9
Measurement of Doped Skutterudites....
47
9
..................................................
...................................
2.10
Quantitative Measurements ............................................................................
48
2.10.1
Thermal Measurements..........................................................................
48
2.10.2
Optical Measurements ............................................................................
51
2.11
Sum m ary ............................................................................................................
Chapter 3:
3.1
Semiconductor Optical Properties ......................................................
Free Carrier Absorption ...................................................................................
54
55
55
3.1.1
The Drude Model......................................................................................
55
3.1.2
Index of Refraction ...................................................................................
58
3.1.3
MATLAB Model - Absorption Coefficient...............................................
59
Fundamental Absorption - Direct Bandgap .....................................................
60
3.2.1
Joint Density of States - Direct Bandgap .................................................
61
3.2.2
Absorption Dependence on Doping..........................................................
63
3.2.3
MATLAB Model - Direct Gap Absorption...............................................
65
3.2.4
Determining the Band Gap - Direct Absorption .......................................
66
3.2
3.3
Fundamental Absorption - Indirect Bandgap .................................................
67
3.3.1
Joint Density of States - Indirect Bandgap...............................................
67
3.3.2
Phonon Population Dependence ...............................................................
69
3.3.3
Determining the Bandgap - Indirect Absorption......................................
70
3.4
Heavily Doped Semiconductors .......................................................................
70
3.5
Summ ary ..............................................................................................................
74
Chapter 4:
Summary and Future Work ...............................................................
76
Chapter 5:
References............................................................................................
78
10
11
List of Figures
Figure 1:Schematic of a Co 4 SbI 2 crystal. 5'8 9' Two voids are present in this crystal, one in
the front upper left octant, and one in the rear bottom right octant. Empty voids may
be filled with rattling atoms that scatter phonons and reduce thermal conductivity. 20
Figure 2: Interferogram of FTIR signal.
As the moving mirror in the Michelson
interferometer sweeps back and forth, the signal intensity changes.....................
Figure 3: Diagram of a Michelson Interferometer.
26
The light source is split and
recombined. The two split beams have different and variable path lengths, which
causes destructive and constructive interference. MI is the fixed mirror, and M2 is
the moving mirror that changes the path lengths...................................................
27
Figure 4: Cross-sectional view of a simple cylindrical photoacoustic cell, showing the
positions of the solid sample, backing material, and gas column. (Rosencwaig and
Gersho, 1976)
3
1, lb, 1 are the thicknesses of the sample, backing, and gas
respectively. ag is the thermal diffusion coefficient of the gas. ...........................
29
Figure 5: Sensitivity plot for CoSb3. The photoacoustic signal for various absorption
coefficients is plotted.
An optically insensitive region occurs at high absorption
coefficients, and can be used to measure thermal properties................................
36
Figure 6: Simulation using literature InSb absorption coefficients as input into a RG
model. The PAS signal and material absorption coefficient are compared side by
side to show the corresponding absorption edges match.....................................
39
Figure 7 PAS spectra of a foamy carbon reference sample. All PAS spectra of samples
are norm alized to this reference............................................................................
12
42
Figure 8: PAS measurement of InSb, undoped, compared with using literature absorption
coefficients in an RG model. There is good agreement in the absorption edges..... 43
Figure 9: PAS measurements of InAs wafers at varying doping concentrations. Higher
doping levels shift the absorption edge to higher energies....................................
44
Figure 10: PAS measurement of crushed, doped InAs. The signal strength is higher due
to scattering and reabsorption. The scattering is also dependent on the wavelength
(energy) of the excitation signal. ...........................................................................
Figure 11: PAS Measurement of undoped Co 4SbI 2 ............................
Figure 12: PAS measurement of Lao. 45Ndo.45Fe 3 .5 Coo. 5 SbI 2.05 .
............ .. .......... . .
45
46
The green line shows a
saturation of the PAS signal throughout all measurable energies, due to too high of
an absorption coefficient........................................................................................
47
Figure 13: Thermal conductivity measurement comparison between PAS and Laserflash
methods. Agreement is within 10% accuracy......................................................
49
Figure 14:Photoacoustic thermal conductivity measurement for single crystalline InSb.
The blue line is the measured data, and the red line is the average thermal
condu ctiv ity ...............................................................................................................
51
Figure 15: Comparison of doped Cuo.oo1 Bi2Te 3 absorption coefficient extracted from a
quantitative PAS measurement, undoped Bi2Te 3 with absorption coefficient found
from literature. The discrepancy of absorption edge is due to band-filling from high
d op ing . ......................................................................................................................
52
Figure 16: Photoacoustic thermal measurement of single crystalline InSb. Blue is the
measured data. Red and green are data from literature. There is good agreement in
13
the band transition region, though the data becomes unreliable at higher absorption
coefficients. This is due to saturation of the photoacoustic signal at -10 5m 1 ......... 53
Figure 17:Predicted photoacoustic signal from measured optical absorption coefficient
using the photoacoustic method. The purpose is to show the insensitivity of the
photoacoustic signal at energies above 0.2eV to the absorption coefficient. Changes
of lOx in absorption coefficient at 0.2-0.3eV result in less than 5%change in
photoacoustic signal..............................................................................................
54
Figure 18: Drude model fitting to free carrier absorption for two different InSb samples
from literature. ..........................................................................................................
59
Figure 19: An illustration of photon absorption by a valence electron for a h eavily doped
material. The Fermi level of the material is within the conduction band............. 63
Figure 20 Modeled absorption coefficient dependence on photon energy for InAs samples
of different doping levels.......................................................................................
66
Figure 21: The bandgap of InAs can be determined by drawing a linear slope to the xintercept energy.
The red line is the literature values of absorption coefficient
squared for InAs. The green line shows the drawn linear slope. InAs' bandgap is
0 .3 5eV .......................................................................................................................
67
Figure 22: Schematic showing the estimation of the bandgap from Fermi-level
m easurem ents.......................................................................................................
71
Figure 23: Diagram relating Fermi-Dirac "knee" to steep increase in absorption
coefficient.................................................................................................................72
Figure 24: Absorption coefficient for undoped InSb, at 130K.....................................
14
74
Chapter 1: Introduction
1.1
Energy Usage
In recent years, worldwide energy consumption has grown dramatically, while the
new discovery of fossil fuel energy sources has slowed down. Another growing concern
over the use of fossil fuels is the rise in environmental pollution and global warming.
This has led to recent efforts in developing new sources of energy, as well as advances in
energy efficiency. Though renewable energy sources such as solar, wind, and geothermal
will inevitably be needed after fossil fuels are exhausted, current renewable energy
technology is still nascent. As of 2011, renewable energy sources only make up 19% of
the world's energy supply.' In addition to finding new sources of energy, there is a need
to improve energy efficiency. According to US Department of Energy, in 2012 58% of
US energy consumption was lost as waste heat. 2 By reducing the amount of energy lost
as waste heat, the available useful energy is increased.
Thermoelectric materials are a recent focus in the search for new energy sources.
These materials convert heat energy directly into electrical energy. They can be used to
recapture otherwise wasted heat energy from current inefficient conversion processes, or
be used to harness new sources of thermal energy. Thermoelectric materials need to be
tuned to achieve optimal performance.
In order to properly understand their electronic
properties, it is important to accurately determine their bandgap energy through
measurements.
The focus of this thesis is to apply a previously unused spectroscopy,
photoacoustic spectroscopy, to this field.
15
1.2
Thermoelectric Materials
Thermoelectric materials can be used to increase energy efficiency by directly
converting waste heat sources into electricity.3
Usually made of heavily-doped narrow
bandgap semiconductors 6, these materials have coupled transport of heat and electrical
energy, and have been used in many applications such as in solid state coolers, energy
generation for satellites, and temperature sensing.7-9
Thermoelectric materials are
essentially heat pumps, able to convert heat flux into electrical power.
Among their
benefits are a lack of mechanical parts and compact size.
1.3
Three Thermoelectric Effects
The first thermoelectric effect is the Seebeck effect, which states that a material
under a temperature gradient will experience an accompanying voltage gradient. This
voltage gradient is caused by unequal transport properties between electrons and holes in
the material. The majority carriers in the material will flow towards the cold side. A
material constant between the temperature gradient and voltage drop is named the
Seebeck coefficient, and is defined as
S=AV
AT
(0.1)
where, S is the Seebeck coefficient, AT the temperature gradient, and AV the voltage
drop. The negative sign can be understood by considering a p-type thermoelectric leg
experiencing a temperature gradient. The holes at the hotter end of the thermoelectric leg
diffuse towards the cold side, leaving behind negative charges. Thus, for a p-type
thermoelectric leg, the Seebeck coefficient is positive, if the positive electrode is placed
onto the hot side.
16
The second thermoelectric effect is the Peltier Effect, which relates the electric
current in a material with the heat flux. If an electric current passes through a material, a
heat flux will be generated according to the equation
Q=fl*J
(0.1)
A material constant H, the Peltier coefficient, is defined and it is equal to S*T through
the Kelvin relation.10 Q is the heat flux, and I is the electrical current.
The last thermoelectric effect is the Thompson Effect, which predicts the heating
and cooling of a material experiencing an electrical current and a temperature gradient.
The Thompson Effect occurs because the Seebeck Coefficient of a material generally
changes with temperature. The Thompson coefficient is defined as
dT
dx
q=-
J
(0.1)
where q is the heat production per unit volume, and J is the current density, I is the
Thompson coefficient, and dT/dx is the temperature gradient.
1.4
Thermoelectric Performance
Thermoelectric devices can be used for both power generation and heat pumping.
When a heat flux is applied to the device, the voltage drop can be used to drive a load.
Conversely, when an electrical current is applied to the device, heat is pumped across the
material.
The same thermoelectric device can thus be a heater, cooler, or power
generator. The expression for thermoelectric power generation efficiency is
T"= T,
T 1
[I+Z
l+ZT + T / T,
where ZT, a dimensionless figure-of-merit, is
17
(0.2)
ZT = S 2 UT
(0.2)
K
and Y is the electrical conductivity, and K is the thermal conductivity. Kis the sum of the
lattice (Kiattice) and electron
(Keectronic)
contributions. ZT is an important dimensionless
number, and is commonly used to characterize thermoelectric efficiency. The numerator
S2
is often called the power factor. The thermoelectric efficiency can be thought of as
the Carnot efficiency, modified by an expression dependent on ZT which is less than
unity.
Currently, highest reliable reported ZT's achieved are around 1.5-2 for bulk
"
thermoelectric materials. 3
1.5
Improving Thermoelectric Performance
Improving thermoelectric performance is difficult, due to the coupled nature of the
relevant properties. ZT can be improved through two directions: improving the power
factor, or lowering the thermal conductivity; however, nature does not allow
improvement of one factor without losses in other factors. For example, increasing the
carrier concentration is an easy way to raise electrical conductivity. However, doing so
will generally decrease the Seebeck coefficient through changes in the Fermi level. In
addition, if carrier concentration is increased to boost electrical conductivity, the electron
contribution to thermal conductivity will increase as well. Maximum ZT is obtained by
carefully considering the underlying behaviors of the carriers, and optimizing each
relevant property.
Phonon Glass Electron Crystal (PGEC) is a concept for increasing ZT, by
preferentially scattering phonons over electrons. 4 "2 This is possible due to the differing
lengths scales on which the two particles operate. Thermal conductivity is composed of
18
lattice and electron thermal conductivity, so PGEC materials attempt to reduce lattice
thermal conductivity.
For example, the host crystal may be partially substituted, to
increase mass disorder. The grain size can be tuned to selectively scatter phonons in the
micron to nanometer range. Certain materials have more options available to minimize
thermal conductivity.
Skutterudites have large crystal vacancies where interstitial
dopants can rattle and selectively scatter phonons of specific frequencies. On the other
hand, the electron scattering can be minimized by reducing impurity scattering through
modulation doping.1 3 In this approach, the impurities are grouped together into
precipitates, so that electrons may have unimpaired travel through the host matrix. In ptype SiGe, the powerfactor was increased 40%, while in n-type SiGe, the increase was
20%. Overall, the figure-of-merit was increased 10%.13
1.6
Skutterudites
Skutterudites are a promising class of thermoelectric materials, because they
exhibit naturally high electrical conductivity, and present a unique approach to reduce
lattice thermal conductivity. The skutterudite base crystal structure of Co 4 Sb1 2 includes
two empty voids per unit crystal, which can be filled with various impurity atoms for
dopoing and reducing lattice thermal conductivity.
shown below in Figure 1.
19
The Co4Sb12 crystal structure is
.
-
cobalt
-
antimony
Figure 1:Schematic of a Co4Sbl2 crystal.12,14,15 Two voids are present in this crystal, one in the front upper
left octant, and one in the rear bottom right octant. Empty voids may be filled with rattling atoms that scatter
phonons and reduce thermal conductivity.
Co 4 Sbi 2 exhibits useful electrical behaviors for thermoelectric applications,
including high electrical conductivity and Seebeck coefficient.
Co 4 Sb 1 2 is naturally p-
type, and has a positive Seebeck coefficient, although the p/n behavior is highly sensitive
to dopants and defects.1 6 P-type Co 4Sb12 exhibits very high hole mobility, which gives
high electrical conductivity values; depending on carrier concentration (1018-10 '9c-n 3 ),
typical hole mobilities are around 2000 cm2 Vs-
.
14,16-1
A common method to p-dope
Co 4 Sb12 is substituting Fe for Co. For n-type Co 4 Sbi-, electron mobilities are one order
of magnitude lower, around 200 cm 2V- Is- at a carrier concentration of 1018 cm- . N-type
Co 4 Sb12 can be created by substituting Co with Ni, Te, or Pd.14, 8- 2
20
0
In order for n-type
Co 4 Sb1 2 to reach similar electrical
conductivities as p-type Co 4 Sb1 2 , the carrier
concentration must be around 100 times greater.
The cause of this disparity between p-type and n-type Co 4 Sb1 2 is due to large
differences in the effective masses of the hole and electron. Caillat et all measured an
electron effective mass of ~-3 me for a carrier concentration of 1019 cm-3 ; for holes, an
effective mass of only 0.07 was measured.1 6 This suggests high asymmetry between the
valence and conduction bands, and offers promising possibilities for materials
engineering.
A variety of processes are available to drastically lower the undesirably high
thermal conductivities of Co 3Sb1 2 , which are on the order of 10-15 Wm'K-1.
14,16,21-23
Through n-type doping of concentrations between 1020 to 1021cm- 3 , thermal conductivity
can be reduced to around 3.5-5 Wm~'K-1.21-27 In 1018 cm~ 3 doped p-type Co 4 Sb12 , thermal
conductivity can be reduced to around 4 Wm-'K-' at 400 0 C.
It was postulated that
electron-phonon scattering is the cause of this thermal conductivity reduction.
A second method to reducing thermal conductivity in Co 4Sb1 2 is by using filler
atoms in the crystal structure voids. Slack et al proposed to fill the skutterudite voids
with atoms that rattle inside.
7
These rattling atoms act as scattering centers for phonons
by resonating at specific frequencies. Sales et al reached a thermal conductivity of only
1.6 Wm-'K-1 by filling Fe 3CoSb1 2 with Ce.
First Nolas and then others proved that by
only partially filling all the available voids, thermal conductivity could be further
reduced.2 1 2 3 2, 8 In addition, using multiple types of fillers have been used to further reduce
thermal conductivity.
The weights of the fillers can be tuned, so that each filler type
targets phonons of different frequencies.
20,24-26
21
For example, Tang et al demonstrated a
thermal conductivity reduction to values less than 1 Wm-IK-1 in CamCenFeCo 4.xSb 12. 2 6
Other fillers used include Yb, Ba, La, and In.
In summary, skutterudites provide attractive opportunities for good thermoelectric
performance, including good electrical and thermal properties. Skutterudites have good
hole mobility values, leading to high electrical conductivities. In addition, there is high
valence band and conduction band asymmetry, which is conducive to high Seebeck
coefficients.
Finally, skutterudites have a unique avenue to reducing thermal
conductivity, by allowing the addition of rattling atoms to disrupt phonon transport.
1.7
Bandgap Measurements
In thermoelectrics, the precise optimization of carrier concentration is important to
maximizing performance. Experimentally, this requires tedious preparation of numerous
samples. Calculations of material properties are important to move from trial-and-error
production to designing thermoelectric materials. One important bandstructure parameter
is the bandgap, which determines optimum operating temperatures for thermoelectric
materials. Previous efforts to calculate the bandgap of the skutterudite Co 4 Sb 12 using
density functional theory (DFT) have produced a wide range in bandgaps.12,29-32
Similarly, experimental measurements of Co 4 Sb1 2 fail to agree with each other and
calculations. The estimates of Co 4 Sb1 2 bandgap range from no gap to 0.5eV.12,29-32
The bandgap strongly influences electronic behavior. The Fermi-Dirac distribution
for charge carriers shows that most electronic activity occurs near the Fermi-level and
bandgap. For example, at high temperatures relative to the bandgap, significant amounts
of minority carriers appear, which decrease the Seebeck coefficient. In addition, the
bandgap is proportionally related to the effective mass of a material. For heavily doped
22
thermoelectric materials, their bandgap and effective mass may be altered from the
undoped state.
DFT computations are not yet able to accurately simulate dopant
concentrations in the 10 cm-3 range, due to a lack of computational power. Therefore, it
is important to perform bandstructure characterization through bandgap measurements on
thermoelectric materials.
Conventional efforts in measuring bandgaps involve determining the spectraldependent absorption coefficients of the materials.
At photon energies above the
bandgap energy, the absorption coefficient increases significantly, up to several orders of
magnitude. The absorption coefficient can be measured using transmission and reflection
measurements on samples of varying thickness. Beer-Lambert's law is used to extract
the absorption coefficient, and is given as
I= 10 exp(-#x)
(0.3)
where Io is the incoming intensity, I is the transmitted intensity, P is the spectral
absorption coefficient, and x is the sample thickness. For heavily doped thermoelectric
materials, this method requires samples of varying thickness, down to 10-5 or 10-6 meters,
to ensure a non-opaque sample.3 3 Transmission measurements thus require timeconsuming sample preparation, and accurate measurement of the sample thickness is
difficult at these thicknesses.
In addition, sample thickness uniformity cannot be
guaranteed.
Another way to measure bandgaps is to measure the reflectivity, and using the
Kramers-Kronig relations (KK), determine the absorption.
KK relations relate the
imaginary and real parts of a complex function, such as the dielectric function of a
material.
KK relations require an integration from oo to -oo, which in practical
23
applications can be achieved by measuring reflectance in a wide energy range.
For
thermoelectrics, this method is not appropriate because of the low energy bandgaps
measured. Conventional FTIR systems have difficulty measuring at energies lower than
0.05eV, due to the KBr beamsplitter commonly used. A better way to measure bandgaps
is required for thermoelectric materials.
1.8
Photoacoustic Spectroscopy
Photoacoustic spectroscopy is one method to directly measure the absorption
coefficient of opaque materials.
In 1880, the photoacoustic effect was discovered by
Alexander Graham Bell. Eventually, photoacoustic spectroscopy (PAS) was developed
as an optical absorption measurement technique for gases, and later liquids and solids. In
PAS, a monochromatic, periodic intensity light source is shone on a sample, and an
acoustic wave is induced. The frequency of the acoustic signal is the same as that of the
intensity periodicity. The acoustic signal is related to the optical absorption and thermal
properties of the materials; the details will be discussed later in this thesis (Section
Chapter 2:).
PAS has been used in a wide variety of fields to measure absorption properties;
both liquids and solids may be measured by PAS. In semiconductors, PAS can be used to
measure diffusion length for carriers, the surface recombination velocity, and the bulk
lifetime.
34-36
Eaves et al measured bandgaps in the semiconductors GaAs and Ge, as well
as Cr impurity levels in GaAs.3 7 PAS has also been used in the biological field, to detect
the presence of protein levels, bacteria, chemicals, and more. 35' 38 In this thesis, PAS will
be used to determine the bandgaps of thermoelectric materials, and the effects of carrier
concentration on absorption.
24
PAS is a useful tool in measuring absorption spectra, especially in opaque samples.
PAS also requires minimal sample preparation, as the sample geometry can be in bulk,
crushed, or powder form. However, quantitative PAS requires the sample geometry to be
known and easily modeled. PAS also allows depth profiling of the sample, by varying
the modulation frequency of the light source.3 6 Shorter modulation frequency results in
shorter thermal diffusion lengths, meaning the sample is measured at shallower depths.
In theory, a range of absorption coefficients can be measured without requiring different
sample thicknesses, by varying the modulation frequency.
In addition, combined with specific heat measurements, PAS can be used to
measure bulk thermal conductivity or thermal diffusivity without requiring knowledge of
the absorption coefficients of the material.
1.9
Fourier-Transform Spectroscopy
By combining Fourier-Transform Spectroscopy (FTS) with PAS, a broadband
measurement of absorption properties can be performed.
In an FTS measurement, an
excitation signal consists of a spectrum of wavelengths, each wavelength having a
characteristic periodic intensity. The composite resulting excitation signal is known as an
interferogram. Below is an example interferogram.
25
Sample Interferogram Signal
60000
U,
'E 40000
20000
-20000
-0.0002
0.0000
0.0002
Mirror Position (m)
Figure 2: Interferogram of FTIR signal.
As the moving mirror in the Michelson interferometer sweeps back
and forth, the signal intensity changes.
Once the excitation signal interacts with the sample, an output signal is received. This
output maintains the characteristic periodic information. A Fourier transform can convert
the output signal from a time-domain signal to a frequency-domain signal, and the
contribution of each frequency can be determined.
In an FTS-PAS measurement, the
Fourier transform will give the photon energy dependent absorption.
The Michelson interferometer is one of many interferometers able to produce FTS
excitation signals.
The Michelson interferometer converts a constant intensity light
source into a periodic intensity light source.
operating principle.
26
The following diagram illustrates the
0
0
C
0
Stationary Mirror
Figure 3: Diagram of a Michelson Interferometer.
The light source is split and recombined.
The two split
beams have different and variable path lengths, which causes destructive and constructive interference. Ml is
the fixed mirror, and M2 is the moving mirror that changes the path lengths.
A light source emits a constant intensity signal, which meets a beamsplitter. The signal
splits in two, with half transmitting directly through to a moving reflecting mirror. The
other half diverts to a stationary reflecting mirror.
Finally, the two split light beams
recombine into one light beam. The moving mirror is the key to the modulated intensity.
While the moving mirror is at different positions, there will be a path length difference
between the two split light beams.
This will alternatively cause destructive and
constructive interference between the two beams. The periodic intensity signal is thus
produced from recombining the two beams.
27
The period of the intensity oscillation is dependent upon the wavelength of light.
This is important because the characteristic period can be used to connect the output
signal to the input signal wavelength.
The Fourier Transform Infrared Spectrometer (FTIR) is a common light source
for FTS measurements. The FTIR includes a broadband blackbody source, and typically
operates in the micron wavelength range. The broadband light from this source enters a
Michelson interferometer, which converts the constant intensity light source into a
periodic intensity light source. The periodic light source then interacts with the samples,
and a detector picks up the resultant signal.
1.10 Thesis Overview
The goal of this thesis work is to develop a new way to accurately measure the
narrow bandgaps of thermoelectric materials, using FTIR-PAS. Section Chapter 2: will
discuss the principles of photoacoustic spectroscopy, and several methods to measure
thermal conductivity and absorption coefficient, the latter which is necessary in
determining the bandgap energy.
Section Chapter 3: explores several fundamental
models for optical absorption in semiconductors, and shows how the absorption
coefficient can be used to determine to indirect and direct bandgap energies. Finally, the
section ends with a discussion on how to interpret the measured absorption coefficient in
the case of extremely doped materials, such as thermoelectrics.
28
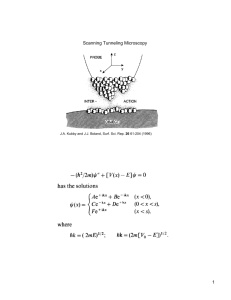
Chapter 2: Photoacoustic Spectroscopy
In this section, the principle of determining thermal and optical properties will be
shown from photoacoustic signal will be discussed together with sensitivity analysis.
Experimental results of PAS measurements will be shown for InAs, InSb, and
Cuo.o 1 Bi 2Te 3 samples.
The PAS method is capable of measuring optical absorption
coefficients and thermal conductivities of bulk samples.
These materials have known
thermal, optical, and electronic properties, and will be used to verify the techniques
developed in this thesis. The developed techniques can then be applied to skutterudite
materials.
2.1
Rosenewaig-Gersho Theory
In
1976,
Rosencwaig and Gersho
developed a model
(RG Theory)
for
Figure 4 shows the idealized sample chamber
understanding the photoacoustic effect.:
for a PAS measurement system.
Backing
Material
Boundary Layer
of Gas
Incident Light
Sample
-(Lb+L)
-L
Gas (Helium)
0
2Tc/ag
X
Lg
Figure 4: Cross-sectional view of a simple cylindrical photoacoustic cell, showing the positions of the solid
sample, backing material, and gas column. (Rosencwaig and Gersho,
1976)
3
L, Lb, Lg are the thicknesses of the
sample, backing, and gas respectively. ag is the thermal diffusion coefficient of the gas.
29
Light input with oscillating intensity enters from the right, and reaches the sample to the
left. The light is absorbed by the sample, and converted into heat energy.
The heat
conducts through the sample into the gas in the chamber, causing the gas to thermally
expand.
Because the light source is periodic, the gas also periodically expands and
contracts, producing an acoustic signal. The acoustic signal depends on the absorption
and thermal properties of the sample.
2.1.1
Heat-Flow and Temperature Profile
The input signal for PAS is a light source sinusoidally varying in intensity. For
now, the light source is considered to be monochromatic.
Therefore, the response
calculated will be from a single wavelength of light. The sinusoidal input intensity is
modeled as
1= -I(1+cos(wt))
where
(0.3)
o is the frequency of the intensity variation, not the frequency of the
electromagnetic wave.
absorption coefficient
Io is the amplitude of the sinusoidal intensity, I. For a given
P,
the volumetric heat generation q(x) within the material at some
depth x will be
q(x)= -#IO exp(/x)(l + cos(ot))
2
(0.4)
The lack of a negative sign in the exponential is due to the coordinate system used in
Figure 4. The heat equation can be written for each section of the PAS cell shown in
Figure 4, taking into account the heat generation in Eq.(0.4).
-2
ax2
= ---
a. at
A exp(#x)(1 + exp(jwt))
30
-1
x
0
(0.4)
a
2
T
1
- = -
ax2
a
2
-- lb
a b at
T
ax
T
1
2
a
!x
-1
(0.4)
T
0 X I1
at
(0.4)
a is the thermal diffusivity, T the temperature, and A = NO
2k
At this point, it is convenient to define two parameters, the thermal diffusion
coefficient, and the thermal diffusion length. The thermal diffusion length defined as
= (
,)1/2
and the thermal diffusion coefficient is a = 1/p. The thermal diffusion
length represents the penetration depth of the temperature gradient into the material, and
is dependent on the intensity periodicity of the incoming light. Slower oscillation of the
light input equates to deeper penetration depth.
In PAS, this relation can be taken
advantage of for depth profiling of the sample.
The solutions to the temperature profile T are given below, for each section of the
photoacoustic cell.
T(x,t) = e, +e~x+dexp(#x)+(Uexp(ux)+ Vexp(-ax)- Eexp(#x))exp(jot)
-l (0.6)
Tb(x,t)
=
()(x + l+b)W + Wexp(b(x + 1) +ot)
-1 - lb
x
0
x
(0.5)
1
lb
ig (0.7)
x
T (x,t) = (1--)0 +O exp(-u x + jwt )
A
where c=(1+j)a, d=--
,and E=
1
A
2
2-
There are two sets of coefficients needed to define the temperature profile: one set
for the time-varying temperature component, and one set for the steady-state component.
31
U, V, W, and 0 are complex and represent the amplitudes of the time-varying portion of
the temperature profile.
el, e2, d, Wo, and 0 are real coefficients, and represent the
steady state portion of the temperature profile. The boundary conditions between the cell
backing, sample, and gas are required to determine these coefficients.
At the boundaries between two layers, the temperature must be continuous, and
the heat flux must be continuous. The boundary conditions are
T (0,t)= T(0,t)
Tb(-l,t)
k,
k
=
(0.7)
T(-l,t)
(0.7)
aT
aT
9 (0,t) = k, s (0, t)
ax
(-I,t)
TT
S x
t)
(0.7)
(0.7)
Although there are 8 coefficients to determine, the 4 boundary conditions can apply
separately to the steady state and time varying portions of the temperature profile.
Applying the boundary conditions, the following equations are produced relating
the steady state coefficients,
00 = el + d
(0.7)
W = e, - el + dexp(-1)
(0.7)
-(kg / 1g )60 = kse2 + kfd
(0.7)
(kb / lb)W = kse2 + k,#dexp(-#1)
(0.7)
For the time-varying coefficients, the following relations can be found,
6=U+V-E
32
(0.7)
W = U exp(-aul) + V exp(csl) - E exp(-#l)
(0.7)
-kgagO = ks(YU - ksV - k,# E
(0.7)
kba bW =
U exp(-aYl) - ksaV exp(7,l) - kSJE exp(-6l)
(0.7)
Matrix equations in MATLAB can be used to solve for the four unknowns in each set of
equations.
The most useful coefficient for the photoacoustic measurement is 0, the timevarying coefficient for the temperature profile in the gas. The photoacoustic signal
depends on the varying pressures in the gas, which depends on the time-varying
temperature of the gas.
An analytical expression for 0 an be solved as
0=
NO
2k,(#
2
--
2
)
(r - 1)(b + 1)exp(al) - (r + 1)(b - 1)exp(-aul) + 2(b - r)exp(-#l)
(g + 1)(b + 1)exp(asl) - (g - 1)(b - 1)exp(-asl)
(0.7)
where b and g are the ratio of thermal fluxes in the backing and gas,
b=
ka
b
k as
(0.7)
ka
ksas
(0.7)
and r is a ratio of the optical penetration depth and the thermal penetration depth.
)
r =(1-
2as
0 has both steady state and time-varying components.
33
(0.7)
2.1.2
Acoustic Signal
The pressure change due to the expansion and compression of the gas can be
determined from the temperature profile at the gas. The temperature of the gas must first
be modeled, and then the volumetric expansion determined. The pressure change follows
the volumetric expansion.
To determine the temperature of the gas, it is assumed that the gas temperature
profile approaches steady-state at a certain distance away from the sample. It can be seen
from the temperature profile that the temperature variations quickly diminish further
away from the sample surface.
At a distance of 2;r / ag from the sample, the gas
temperature is essentially constant. An approximation is made to split the gas into two
components. The gas near the sample surface experiences temperature change, and acts
as a gas piston on the rest of the gas, which undergoes adiabatic compression.
The spatially averaged temperature is found using the time varying portion of
Eq.(0.7), the time-varying temperature profile in the gas. The average temperature is
found by integrating over the length of the air piston,
12
Kg
'
[eexp(-agx+ jwVt)Px
(0.7)
The air piston portion of the gas is assumed to be an ideal gas. The displacement due to
the periodic heating is modeled as
3x(t)= 2;r
Y
-=
TO
6Mg
1
exp(j(ot - -))
4
2T
-
34
(0.7)
where To is the average steady state temperature of the sample surface, found by
summing the ambient temperature and 0, the steady state temperature profile of the
sample-gas boundary.
This displacement of the air piston adiabatically compresses the rest of the gas in
the chamber. Eq.(0.7) gives the adiabatic gas law, where y is the ratio of the constantvolume and constant-pressure specific heats.
PV' = constant
(0.7)
SP(t)=
(0.7)
8V
V
Using Eq. (0.7), the periodic gas displacement, and substituting for 6V in Eq. (0.7), the
expression for the acoustic pressure change is found:
8P(t) =
y________
exp(j(cot -
2l agT
1
-
4
r))
(0.7)
6P(t) is important because it gives the magnitude of the photoacoustic signal from
the sample. The photoacoustic signal derived in Eq. (0.7) is for a monochromatic light
source. In the case of an actual photoacoustic measurement, a broadband light source can
be used, and the total photoacoustic signal is a linear superposition of the individual
responses from the different light wavelengths. In FTIR-photoacoustic spectroscopy, the
individual photoacoustic responses each have different frequencies, depending on the
intensity modulation frequency of each wavelength of light.
2.2
Photoacoustic Response - Sensitivity
The photoacoustic signal determined in Eq. (0.7) depends on the coefficient 0,
which determines the time-varying temperature in the gas; it is very complex and
35
The sensitivity of the PAS measurement to absorption
depends on many factors.
coefficient differs depending on the thermal properties of the material, and measurement
parameters. However, in many limiting cases, the photoacoustic signal can be simplified
with some knowledge of the sample being studied.
The different sensitivities of the
photoacoustic measurement mainly depend on whether the sample is thermally or
optically thick, and the ratio between the thermal and optical lengths.
,
Simulations were performed to understand the photoacoustic sensitivity of CoSb 3
using thermal properties from literature.
2
Figure 5 below shows the simulation results.
Test Absorption Coefficient vs. Energy
4.5.............,......
...
,... ...
.....
4
/"
3.5
-
3
-
2.5
2-
o ically
Optically
S nsitive
Insensitive
1.5 --
10
10
*...'
''"
10
.
.'
.
.'''
.
.'
.
104
.-- "
10
'
'
.1
10a
'
"
0
-
0.5
10
Absorption Co efficient (m-1)
Figure 5: Sensitivity plot for CoSb3. The photoacoustic signal for various absorption coefficients is plotted. An
optically insensitive region occurs at high absorption coefficients, and can be used to measure thermal properties.
The sample was given a range of absorption coefficients, and thermal properties similar
to CoSb 3. The signal change shows that for this situation, the photoacoustic measurement
can detect changes in absorption coefficient between 102 to 105 m-1. The modulation
36
frequency of the light source was kept constant (300s-) for different wavelengths of light.
In normal operation, the change in modulation frequency would have to be accounted for.
This would shift the sensitive region downward to lower absorption coefficients.
The
absorption coefficient range 102-105 m 1 should be sufficient to determine the bandgap
using the method discussed later in Section 3.2.4 of this thesis.
Figure 5 shows two useful sensitivity regions: an optically sensitive (green) and
optically insensitive (red) region. In the optically insensitive region, the photoacoustic
signal depends largely on the thermal properties of the sample. The thermal diffusion
length is much larger than the optical penetration depth, and so where the light is
absorbed is not as important. In this case,
Q
P<<ps, and p,<<
1, and the signal is roughly
fs
2agks
(0.8)
The signal is proportional to the inverse of the effusivity of the sample, and independent
of the absorption coefficient.
Using the optically insensitive region, the PAS measurement can be used to
measure thermal properties. The PAS measurement depends on a and cp. Given one of
those parameters, and assuming a large absorption
coefficient (>106 m-1 )
the
photoacoustic model in Section 2.1 can be used to fit an unknown thermal property. This
will be further discussed in Section 2.10.1.
In the optically sensitive region, the photoacoustic signal depends on the optical
properties of the sample.
The optical penetration depth is deeper than the thermal
diffusion length, and so the amount of heat traveling to the sample surface depends on the
absorption profile.
In the case where
pr>p,,
37
and p<<I,, i.e. where the sample is
thermally thick, and the optical penetration depth is longer than the thermal diffusion
length, the PAS signal is approximately
Q
pS
2agkS
(0.9)
and is proportional the absorption coefficient.
2.3
Photoacoustic Model of InSb
To test the validity of the photoacoustic model, literature absorption coefficients
of InSb determined by transmission measurements were input, and the resulting PAS
signal generated was compared to a PAS measurement (later, in Section 2.6). Indium
antimonide is a narrow bandgap material (0.17eV at 300K), and is well-studied enough
that it will be used as a validation material for this thesis. 40 InSb has both well-known
thermal properties, and optical absorption coefficients at different carrier densities.
Thermal conductivity and specific heat used were 18W/m-K and 200 J/kg-K. 41 The
specific heat was verified using a Netzsch DSC404F 1. Figure 6 below shows the results
of the simulation.
38
107 >
0.8
CT
0
0.6
106w
U) 0.4
10 5 0
0
Cn
a-
0.0
0.2
0.4
0.6
104
'
0.2
PAS Signal
-u- Absorption Coefficient
0.90,
Energy(eV)
Figure 6: Simulation using literature InSb absorption coefficients as input into a RG model. The PAS signal and
material absorption coefficient are compared side by side to show the corresponding absorption edges match.
2.4
Quantitative Measurements
Under practical
situations,
the
RG model
cannot
accurately
predict a
photoacoustic signal. Several additional factors are present in the experimental setup,
which are difficult to account for. For this work, the PAS detector uses an FTIR as a
light source. The power output received by the PAS detector is unknown. The sample
chamber's acoustic frequency response is also unknown.
The chamber response will
depend on the volume of the gas, and the thermal diffusion length within the gas. 42
Smaller volumes have curved acoustic responses, with higher signal at high and low
frequencies; conversely, larger volumes give flatter responses.
microphone and circuitry sensitivity is unknown.
43
In addition, any
Rather than chase down all these
unknowns, it is easier to negate their effects somehow.
The proposed solution of this thesis is to do a relative measurement, and then
determine the parameter of interest using a relative calculation. By using a reference
material with known properties, all the experimental unknowns can be normalized and
39
neglected.
Certain sample properties must be known, depending on what property is
being fitted in the relative measurement. For example, to determine optical absorption
coefficients, both the specific heat and thermal conductivity must be known. The specific
heat must be measured in a separate measurement, perhaps with differential scanning
calorimetry (DSC).
The thermal conductivity can be determined from the optically
insensitive measurement regime.
In a semiconductor PAS measurement, the energy
region above the bandgap can be used to determine the thermal conductivity.
This
thermal conductivity then can be used to help determine the absorption coefficients for
the region below and around the bandgap.
2.5
Experimental Setup
The photoacoustic
measurements are done using an FTIR-Photoacoustic
Spectroscopy (PAS) set up. A Thermo Nicolet 6700 FTIR is used as a light source for
the PAS detector. The light sources used in the FTIR are an ETC EverGlo infrared light
source or Whitelight visible light source. The EverGlo light source was used in the 252.5[tm range (0.05-0.5eV), while the Whitelight source covered the 5-1tm range (0.25leV). A KBr beamsplitter was used for the lower energy measurements, and is capable
of handling light with energy from 0.05eV to above leV. A CaF 2 beamsplitter was used
for the higher energy measurements, in conjunction with the Whitelight source.
A PAS detector was used in conjunction with the Thermo FTIR, and was placed
inside the bench area of the FTIR. The PAS detector used was an MTEC Inc. PAC300
unit. The PAC300 is able to hold samples up to 10mm in diameter, and 5mm thick. Both
monolithic disc, crushed, and powder samples were used. For quantitative measurements,
a 9.5mm diameter disc of 2mm thickness was used. The gas gap between the sample and
40
top of the chamber was 2mm. An additional volume of 0.07cm2 was used to house the
microphone; this additional volume was accounted for in the PAS model.
The PAS
detector included a gain dial, which amplified the signal. Different gains were used
during the reference and sample measurements.
The PAS measurement is highly sensitive to humidity, so care was taken to
maintain a dry environment. The FTIR was continuously purged with a stream of dry air,
to minimize the amount of CO 2 and water vapor light absorption before reaching the PAS
chamber. A small amount of CaCl2 was placed in the FTIR chamber to further reduce
humidity. Typically, a 10-minute wait time was used to purge away excess CO 2 . The
PAS detector itself was purged for 5 minutes with a dry zero-grade helium source, and
then sealed prior to measurement. From the measured signal, contamination in the FTIR
chamber or PAS detector could be seen by either negative or positive pointing peaks,
respectively.
A background scan was first obtained, by using a "foamy" carbon reference
sample, provided by MTEC Inc. A carbon sample is highly absorptive, and its optical
absorption length is negligible compared to the thermal diffusion length.44 48 Therefore
the photoacoustic response is independent on the absorption coefficient, and a
background signal only dependent on the system properties can be obtained.
Below is the PAS signal from the foamy carbon reference, using the EverGlo light
source. The signal becomes very weak around 0.45eV, and reliable measurements cannot
be made above this energy, for this configuration.
41
I
I
I
I
0.1
0.2
0.3
0.4
200
100
-
.0
0.5
Energy(eV)
Figure 7 PAS spectra of a foamy carbon reference sample.
All PAS spectra of samples are normalized to this
reference.
Depending on the signal strength, the amount of sample scans needed varies.
Typically, for a high-energy(eV) measurement, a scan time of about 10-20 minutes was
needed, at a resolution of 16cm- . At low energies, perhaps only 5 minutes was needed at
the same resolution.
To quantitatively determine the amount of light entering a sample, the sample
reflectivity was measured using the same FTIR, coupled with a Harrick ERA reflection
stage. The reflection angle is 120, which is considered close enough to normal (cos 12'
0.98). The reflection is needed to determine the intensity of light entering the sample,
since different samples have different reflectivities.
Samples were polished using a
rotating wheel polisher until the surfaces were specularly reflecting. A series of polishing
suspensions were used, with the smallest particle size at 50nm.
42
2.6
Measurement of Indium Antimonide
InSb single crystal wafers were measured using the PAS detector, and compared
to the PAS model results in Section 2.3. Figure 8 below shows the PAS measurement of
InSb. For comparison, the results of the RG simulation from Section 2.3 are shown side
by side.
0
'
40--
C
Experiment
Literature+RG Model
20-
8.0
0.2
Energy(eV)
0.4
Figure 8: PAS measurement of InSb, undoped, compared with using literature absorption coefficients in an RG
model. There is good agreement in the absorption edges.
The PAS model can be verified by comparing the absorption coefficients measured using
the PAS method, and those measured using traditional transmission measurements.
The InSb sample measured is a single crystal undoped wafer, 9.5mm in diameter
and 0.5mm thick. The doping level is below 1016 cM-3. As expected, the absorption edge
appears around 0.17eV,
and is in excellent agreement with literature bandgap
measurements. The plateau feature starting around 0.2eV is very flat, and can be used to
estimate the thermal conductivity of InSb.
43
2.7
Measurement of Indium Arsenide
Indium Arsenide is another well-studied narrow bandgap material (0.357eV at
300K). It is commercially available at different doping concentrations, and will be used
to illustrate the effects of doping on the absorption edge. Doping shifts the absorption
edge to higher energies, due to the Moss-Burstein effect. The doping effect on absorption
edge is important to understand, as thermoelectrics are often heavily doped materials.
Single crystal wafers 9.5mm in diameter and 0.5mm in thickness are measured
using PAS, at different doping concentrations. Figure 9 below shows how the absorption
edge shifts to higher energies due to doping. More discussion is written in Section 3.2.2.
I
200
100
-
<3e16 at/cm 3
-
3-10e17 at/cm 3
lel8at/cm 3
0)
0.2
so 1
0.3
0.4
0.5
Energy(eV)
Figure 9: PAS measurements of InAs wafers at varying doping concentrations. Higher doping levels shift the
absorption edge to higher energies.
The blue plot is the undoped wafer, which shows an absorption edge at 0.35eV.
Several peaks are visible at 0.2eV and 0.3eV, and are attributed to humidity and CO 2 in
the PAS chamber.
The red and green plots show heavier doping levels, with
progressively higher energy absorption edges. In addition, the free carrier absorption at
44
lower energies (<0.2eV) increases for higher doping levels. Measurement of Crushed
Samples (InAs)
When uniform, polished samples are unavailable, the PAS detector can be used
for qualitative measurements of samples. For example, a crushed sample will produce a
strong acoustic signal, though it will be difficult to perform quantitative measurements.
Incident light will be reflected, scattered, and reabsorbed into the sample. The amount of
light entering the sample can not be accurately determined. Below are results for a PAS
measurement on doped InAs (n=3- lOx I0 7 cm-3).
200
2 150
10050
0
0.2
0.6
0.4
Energy(eV)
Figure 10: PAS measurement of crushed, doped InAs.
0.8
The signal strength is higher due to scattering and
reabsorption. The scattering is also dependent on the wavelength (energy) of the excitation signal.
The crushed InAs PAS signals looks qualitatively similar to the single-crystal wafer InAs
PAS signal in Figure 9.
The absorption edge ends at 0.35eV, as expected for InAs.
However, the sample light input is unknown, but is evidently higher. The acoustic signal
is generally 2-5 times stronger for the crushed samples, although this is highly dependent
on the sample crushing. Also, light scattering effects cause the free carrier absorption to
be stronger than normal.
45
2.8
Measurement of Undoped Skutterudites
Skutterudites, as typified by Co 4Sbl 2, have a bandgap that is highly sensitive to
the lattice position of the Sb atoms. By changing the position less than 1%, the calculated
bandgap changed from 0.05eV to 0.22eV. 2 9,3 0 As stated in Section 1.6, there is little
agreement in measured bandgaps of undoped Co 4 Sb] 2 . Therefore, there is a clear need
for experimental verification of the optical bandgap for Co 4 Sb] 2. In addition, current
state of the art skutterudites contain heavy doping in the form of Fe substitions for Co,
and filler atoms such as La, In, and Ce. The doping in these materials may cause the
bandgap to shift, considering the high sensitivity to lattice positions. Measurements of
both highly doped and undoped Co 4 Sb] based skutterudites will be shown in this section.
A PAS measurement was performed on an undoped Co 4 Sb 2 , and the results
are shown below.
80
100
601
80
-Signal
40 .60
Reflection
Corrected Signal
20
40
20
0.2
0.3
0.4
0.5
Energy (eV)
11: PAS Measurement of undoped Co 4 Sb12
.
Figure
Though the sample is undoped, it still has a carrier concentration of 5x10 1cM- 3, deduced
from Hall effect measurements.
This residual carrier concentration comes from excess
46
elements and defects, and causes the high absorption below -0.3eV.
However, there is
still a feature above 0.35eV, and suggests that the bandgap is around 0.32eV.
2.9
Measurement of Doped Skutterudites
A doped sample of Lao.4 5Ndo. 4 5 Fe 3.5 Co0 .5 Sb12 .05 was measured using the PAS
detector. The results are shown below in Figure 12.
250
100
200
80
150-
60
S100 -
- 40 5
-
Cn
50
a-
-
8.0
PAS Signal
Reflectivity
Adjusted PAS
20
0.2
0.4
Energy (eV)
Figure 12: PAS measurement of La).4SNdO.4 5Fe 3.5 CoO.Sb 12 .05 . The green line shows a saturation of the PAS signal
throughout all measurable energies, due to too high of an absorption coefficient.
After correcting for reflection losses, the PAS measurement of the doped skutterudite
shows no bandgap features. Most likely, the entire signal is optically saturated, meaning
that the absorption coefficient is very high. It is possible that due to the high level of
doping (~1020cM3) the bandgap could have disappeared. There could also be strong
absorption from the formation of sub-bandgap defect states. In Figure 5, the sensitivity
analysis predicts that the PAS measurement for undoped Co 4Sb 12 will be saturated at an
absorption coefficient of around 104 m-1. For heavily doped InSb with carrier
47
concentrations of above 10 18cm- 3 , the sub-bandgap absorption coefficient is already at
this level. Considering that the doped skutterudite has a carrier concentration two orders
higher, it is very likely that the sub-bandgap absorption has caused the PAS optical
saturation.
To determine whether the signal flatness is due to saturation of the PAS signal, or
due to flatness of the absorption coefficient, we can do a thermal conductivity analysis.
Assuming the PAS signal is optically saturated, the PAS measured thermal conductivity
was 2.2 W/m-K. In comparison, the laser flash measured thermal conductivity was 2.05
W/m-K. This confirms that the entire PAS spectrum is due to signal saturation from the
sample, and not merely due to constant optical absorption.
2.10 Quantitative Measurements
The photoacoustic measurement is not linearly proportional to the absorption
coefficient. The PAS signal is affected by the comparison between the absorption
coefficient and the thermal diffusion coefficient, which changes with the light wavelength
in an FTIR. At shorter light wavelengths, the FTIR modulates the intensity faster, and so
the thermal diffusion coefficient decreases.
This gives rise to a need for good
quantitative analysis using the RG PAS model to determine quantitatively the absorption
coefficients and thermal properties. In this section, methods will be shown to measure
thermal diffusivity and absorption coefficient of a sample using the PAS method.
2.10.1 Thermal Measurements
To test the reliability of the RG PAS model for the PAS measurement, a
comparison of the thermal measurement was made with a separate measurement made
through the Laserflash technique. 49 A commercial equipement, the Netzsch LFA457 was
48
used to measure the thermal diffusivity of a Cuo. Bi2Te3 nanocrystalline bulk sample.
Combining the specific heat (from DSC) with the thermal diffusivity gave a thermal
conductivity. The specific heat was also input into the RG PAS model, and a thermal
conductivity was fitted to the optically insensitive portion of a PAS measurement.
To do a relative PAS measurement, an undoped single-crystalline InSb wafer was
used as a reference.
measurement.
The Cuo.OIBi2Te3 was measured, and normalized to an InSb
The InSb properties were assumed known, and values were taken from
literature. A kl1 1sb of 18W/m~K, and cp.Ins of 200J/kg-C' were used. The
Cp-InSI
was
verified using a Netzsch DSC404Fl.
For CuO.o 1 Bi2Te,, a
C)-Bi2Te3
of 148J/kg-C' was measured. Below, the results of
fitting Cuo.()oBiTe 3 's thermal conductivity using PAS are compared to the Laserflash
measurement.
1.0-
0 .
--
-I-
00.01
0.30
PAS method
Laserflash method
0.35
0.40
Energy (eV)
Figure 13: Thermal conductivity measurement comparison between PAS and Laserflash methods. Agreement is
within 10% accuracy.
49
The results of PAS measurement show that the RG PAS model reasonably predicts the
PAS signal of a sample. The PAS measurement of thermal conductivity is within 10%
difference of the Laserflash thermal conductivity. In addition, the Laserflash technique
only claims 10% accuracy.
Cuo.oBi 2Te 3 is an unsuitable material to verify the optical measurements, so the
undoped InSb single crystal wafer will be used to verify the optical measurements. To
determine the thermal conductivity value fit from InSb, a third reference material will be
needed.
Carbon foam is used as the reference material.
The thermal properties for
carbon foam are determined using the previously verified thermal conductivity
measurement, with a variety of tested thermoelectric materials as reference samples. The
carbon foam used values are: density is 1490 kg/m 3 , thermal conductivity is 0.59 W/m-K,
and specific heat is 42 J/kg-K. In practice, the carbon foam properties depend on the
effusivity, so all three values are combined into one term.
The photoacoustic thermal measurement of single crystalline InSb is given in the
figure below.
50
~20
~15
E
0
C-)
-
.)
I
Conductivity
-Thermal
Average
1
0.30
0.35
0.40
0.45
0.50
Energy (eV)
Figure 14:Photoacoustic thermal conductivity measurement for single crystalline
InSb.
The blue line is the
measured data, and the red line is the average thermal conductivity.
The blue line shows the thermal conductivity calculated from each photoacoustic
measurement point. The red line shows the average thermal conductivity over the entire
range. An average of 18.7 W/m-K was determined from the measurement, compared to
literature thermal conductivity values of 16.5-18 W/m-K for single crystalline InSb.
In this measurement, the scatter is ~22%, which is slightly worse than the Cuo.o 1 Bi2Te3
measurement (10%).
This is due to the lower absorption of the InSb sample, and
therefore the lower signal to noise ratio in the measurement.
2.10.2 Optical Measurements
Once the thermal diffusivity of a sample is known, either through a PAS
measurement or alternate measurement, the PAS measurement can be used again to
determine the sample absorption coefficient. In the optical measurement, the optically
sensitive portion of the measurcment is used.
51
Again, the reference material used is
undoped InSb, single-crystalline wafer.
Figure 15 below shows the absorption
coefficient determined from a Cuo01 Bi2Te 3 measurement.
(
3x10 5
3 2x105
C)
PAS (Doped)
Litergture (Undoped)
0.2
0.3
Energy(eV)
--
0
0.1
Figure 15: Comparison of doped CuO.oOBi 2Tej absorption coefficient
extracted from a quantitative PAS
measurement, undoped Bi 2Te3 with absorption coefficient found from literature. The discrepancy of absorption
edge is due to band-filling from high doping.
Although the slopes of the absorption coefficient do not line up, the magnitudes are the
same. The difference in absorption coefficient slopes is attributed to the doping effect in
the PAS sample.
As described in Section 3.2.2, highly doped samples will shift the
absorption edge to higher energies.
A better comparison would be to compare optical properties of samples with the
same or similar carrier concentration. Single crystalline InSb was purchased with similar
carrier concentration (10 15ciM 3) with data from literature( 10 -310 4 cM- 3). 2
52
3
.5
107
E
06
-103
0
.
Data Fit (rn- 1
Literature 1
)
C2
Literature 2
10 2
0.1
0.2
0.3
0.4
Energy (eV)
Figure 16: Photoacoustic thermal measurement of single crystalline
InSb.
Blue is the measured data. Red and
green are data from literature. There is good agreement in the band transition region, though the data becomes
unreliable at higher absorption coefficients. This is due to saturation of the photoacoustic signal at ~10 m-1.
In the band edge region, there is good agreement in the fitted absorption coefficient
(blue), with transmission data from literature (red&green). 2 ,54 At higher energies of 0.20.3eV, the fitted data deviates an order of magnitude from the transmission data from
literature. This is due to the decreasing sensitivity of the measurement to the absorption
coefficient, as the photoacoustic signal becomes saturated. From sensitivity calculations,
it was found that the maximum measurable absorption coefficient is on the order of 105
m'. Furthermore, the order of magnitude deviation from 0.2-0.3eV result in roughly 5%
change in the photoacoustic signal.
The fitted absorption coefficient in Figure 16 is
inputed into the photoacoustic model to determine the photoacoustic signal, to quantify
the change in signal at the 0.2-0.3eV range. The results are shown below.
53
0.0008
T 0.0006CO
.9
0.0004
0
M 0.0002
0
a- 0.0000M
0.1
0.2
0.3
0.4
0.5
Energy (eV)
Figure 17:Predicted photoacoustic signal from measured optical absorption coefficient using the photoacoustic
method.
The purpose is to show the insensitivity of the photoacoustic signal at energies above 0.2eV to the
absorption coefficient.
Changes of lOx in absorption coefficient at 0.2-0.3eV result in less than 5%change in
photoacoustic signal.
2.11 Summary
PAS is a versatile spectroscopy tool that is capable of measuring thermal and
optical properties of samples, by taking advantage of different sensitivity regimes.
A
variety of well-studied samples (InSb, InAs) were measured using the PAS method, and
higher doping concentrations were shown to shift the absorption edge to higher energies.
A modified RG theory was used to model the experiment, and relative measurements and
simulations allowed the quantitative thermal conductivity and absorption coefficient to be
determined. For the thermal conductivity of Cuo.o1 Bi 2 Te 3, the PAS measurement gives
agreement within 10% of the laserflash measurement method.
For the absorption
coefficient, the correct magnitudes were measured, and a slight deviation is attributed to
the difference in doping concentrations in samples between the PAS and literature values.
54
Chapter 3: Semiconductor Optical Properties
In semiconductors, optical absorption is heavily dependent on the bandgap energy
and doping level. The Fermi-level may be within the bandgap, or in the conduction or
valence bands for heavily doped semiconductors.
In this section, basic theory and
models are presented for various absorption processes such as free carrier absorption and
fundamental band-to-band absorption.
Fundamental absorption is described for both
direct and indirect bandgaps.
3.1
Free Carrier Absorption
Free carrier absorption occurs due to electrons in the conduction band, and is
significant at low frequencies.55-58 These free electrons can be caused by high doping
levels in the material, and by thermal excitation of electrons in the valence band or
doping levels. Free carrier absorption is a two-step process, and requires a phonon to
shift a free electron's momentum.
3.1.1
The Drude Model
The Drude model can be used to model free carriers in metals and metal-like
heavily doped semi-conductors. Electrons are modeled as semi-classical free particles,
meaning an effective mass is used instead of the free electron mass. Also, a damping
term is included. The Drude model starts from the motion equation,
m
d2 r
dr
+ my d- -eE
dt2
dt
2
55
(0.9)
where m is the effective mass of the carrier, r is its position, y is the damping constant, e
is the electron charge, and E is the electric field. Using a variable change, the current J
can be related to the position:
dr
dt
(0.9)
J = -n -e-v
(0.9)
m dJ myJ(09
-- d/ +
=-eE
ne dt
ne
(0.9)
where ne is the carrier concentration. Eq. (0.9) is a differential equation relating current J
with electric field E. By definition, J is related to E through the relation
J=c-E
(0.9)
Assuming sinusoidal solutions for both J and E:
J = Joe~'"'
(0.9)
E = Eoe-'"'
(0.9)
w is the angular frequency, and Jo and EO are wave amplitudes. Substituting sinusoidal
solutions for J and E, into the motion equation yields the relation
-iwJ + yJ =
2nE(0.9)
This can be further simplified to determine the complex conductivity
-E
m(-iw+y)
Y=
ee 2
m(-iw+y)
56
&*, where
(0.9)
(0.9)
n
It is convenient at this time to define a low frequency conductivity Co =
2
my
.
This is
also known as the DC electrical conductivity.
Maxwell's equations are used to relate conductivity to the index of refractivity,
which has the imaginary component k*, which is associated with energy dissipation. k* is
related to the absorption coefficient, which we are interested in.
From Maxwell's
equations, the following relation can be obtained:
E
V 2EIa2=
1
aJ
+
O
2 at2e,72
1WE oa2at
C2a2
12
c2 at2 +,,c 2 1-iw Iy
2E
Ecore
a
(0.9)
at
Again, if time and space dependent sinusoidal
is the local electric permittivity.
solutions for E are used, Eq. (0.9) simplifies to:
k2 =
C
2 +(
.
)
1-iw/y
(0.9)
where pt is the local magnetic permeability. The dielectric constant E can be obtained by
using Eq. (0.9) with Eq. (0.9).
=
2
(0.9)
2
2
W2
++i Wcore
S= coee,-
coeW2
+2
py
(0.9)
W3 +W2
fl2
W,
ne
MEoEcore
A useful constant wp is defined here, the plasma frequency.
(0.9)
wp is defined as the
frequency where the real part of the dielectric function becomes 0. At this frequency, an
57
ideal material will reflect all light.
permittivity, while
3.1.2
Ccore
It is important to note that Co is the vacuum
is the dimensionless local dielectric constant of the material.
Index of Refraction
The dielectric constants and index of refraction are related through the relation:
e=e'-iE"=N2= n -ik *
(0.9)
where N is the index of refraction, composed of real and imaginary parts n and k*. A few
relations are useful between c and N.
E= n 2 -k*
2
(0.9)
e "= 2nk*
n2
k *2
(0.9)
1(E'+ e'+E "2)
(0.9)
2
2
(-E'+
(0.9)
e'2+8"2)
To determine a useful expression for k, some approximations should be used. In the
infrared region, where photon energies are close to many semiconductor bandgap
energies, w >> y , w >> w,, and n >> k .59-63
The following approximation for the real
portion of the dielectric constant can be made.
I=
=E
cor
core 2
W2 +,22
n2
~Ecore
(0.9)
From the approximation in Eq.(0.9), we can approximate the imaginary portion E" in
"
Eq.(0.9) as:
2
E ~2nk* = 2N Eek- = Ecore 3Py
3
58
(0.9)
(0.9)
2w
k* is directly related with the absorption coefficient through the following relation:
a=2wk*
(0.9)
C
a=
SWY
(0.9)
Wa-
In conclusion, it was shown that for semiconductors under the conditions of w >>
w >> w, , and n >> k , the absorption coefficient a is proportional to w- 2 or
k2 .
The free
carrier absorption coefficient given in Eq. (0.9) also shows a direct dependence on carrier
concentration.
3.1.3
MATLAB Model - Absorption Coefficient
A MATLAB model was created to model the Drude free carrier absorption
coefficient using the full expression for the k- given in Eq. (0.9).
Figure 18 shows
literature free carrier absorption coefficients 53 for two samples of InSb with different
doping levels, and the Drude model fit.
InSb, n=2.6e18 cm- 3
InSb, n=7e17 cm-3
1.E+05
1.E+06
E
-Drude Model
Fit
Literature
- 1.E+05
-Drude
E
-
- 1.E+04
i)
0
0
U
C
Model
Fit
-Literature
-Ltrtr
S1.E+03
.
o 1.E+04
0.
0.
'4
I .E+02
1.E+03
0.05
0.10
0.15
0.20
0.05
0.25
Energy (eV)
0.10
0.15
0.20
Energy (eV)
Figure 18: Drude model fitting to free carrier absorption for two different InSb samples from literature.
59
0.25
Below 0.2eV, the Drude model has good agreement with the free carrier
absorption. For the Drude model fit, the dielectric constant used was 16, the effective
mass was dependent on carrier concentration, and the scattering time was fitted. 4 65 For
the high and low concentration fits, a scattering time of 1x1013 s and 3.5x1014 s were used
respectively.
At even higher energies, the absorption coefficient does not follow the
Drude model closely. This is evident in the higher concentration model (Figure 18, left).
This is because other absorption processes are happening. InSb has a bandgap around
0.17eV, and so some band-band transitions are occurring around 0.2eV and higher. In
heavily doped samples, Urbach band tails will start to appear, leading to additional
absorption below the bandgap energy.
3.2
Fundamental Absorption - Direct Bandgap
In direct gap semiconductors, photons can excite valence electrons across the
bandgap, while effectively conserving the electron momentum.
While a photon has
significant energy, its momentum is relatively small compared to the electron. Co 3 Sb12
has a lattice parameter of 9A; correspondingly, the Brillouin zone edge is 7x101 0m-1. For
a photon in the near IR range, its wavenumber is on the order of 10 6 m-1, roughly 4 orders
of magnitude lower than the length of the Brillouin zone.
Fundamental absorption can be used to determine a material's band gap. While
photons of energy higher than the bandgap will be absorbed, photons of insufficient
energy will pass through. The absorption coefficient due to fundamental absorption in
direct bandgap materials is dependent on the bandstructure of the material. A general
form of the absorption coefficient is given as
60
ho * number of transitions per second per volume
Total incoming energy flux
(0.9)
The numerator of Eq.(0.9) is strongly dependent on the joint density of states for the
material system. The absorption coefficient can be defined as a spectral coefficient, or
effective coefficient (broadband).
3.2.1
Joint Density of States - Direct Bandgap
The joint density of states between a material's conduction band and valence band
affects the absorption coefficient. In this thesis, a simple parabolic model is used as an
example. In actual materials, the joint density of states will differ, based on the crystal
structure.
In future work, computational methods such as Density Functional Theory
(DFT) can be used to simulate real materials.
For a single parabolic band in an isotropic material, the density of states is
p(E)=
I(
)32-1
21r 2 h2
(0.9)
where m* is the effective mass, h is the reduced Planck's constant, and E is the energy.
However, for fundamental absorption both the valence and conduction bands must be
considered, whereas Eq. (0.9) only account for a single band. The joint density of states
will have a form similar to Eq.(0.9), except with a reduced mass m, substituted for the
effective mass. The derivation for the joint density of states will be given below.
Starting with the energy dispersions for the valence and conductions bands:
E =
h2 k 2
g
E = hk
'
(0.9)
+E
"2m
2mh
61
(0.9)
Here, Eg is the bandgap energy, and the minima of the conduction band. E" and E, are
the energies of the conduction and valence band energy, with respect to the top of the
valence band. Ep is defined as positive.
The photon energy h can be written in terms of the energy dispersions and the
bandgap energy.
Taking into consideration that k is conserved in direct bandgap
materials, the photon energy is simply the sum given as
ho= E +E =
h)- E9 =
M-
h2 k2
+E
h2 m2
(0.9)
(0.9)
2m
mmh
me+ mh
(0.9)
From Eq. (0.9), re-arranging Eg to the left side, it can be seen that the energy
expression is similar to the case with one parabolic energy band. The reduced mass m, is
defined in Eq. (0.9). A simple derivation will show the joint density of states is similar to
the one given in Eq. (0.9).
p(h2)
1
2m
2
h-E
(0.9)
The joint density of states has a square root dependence on the photon energy minus the
bandgap energy. To determine the bandgap in undoped semiconductors, the absorption
coefficient squared is plotted against the photon energy. The intercept of the absorption
coefficient squared will point to Eg.
62
3.2.2
Absorption Dependence on Doping
In thermoelectrics and other degenerately doped semiconductors, the Fermi level
is located within either the conduction or valence band. This has significant effects on
the absorption characteristics for carriers. The measured absorption edge for high Fermi
level semiconductors generally does not indicate the band gap.
In the intrinsic case,
electrons fill the valence band, and the conduction band contains mostly unoccupied
states. An electron absorbing an incoming photon can be excited to the conduction band,
into one of the unoccupied states. However, if the semiconductor is significantly doped,
then many of the previously unoccupied states are now occupied.
In this case, the
valence band electron must absorb a photon with high enough energy to reach an
The higher the Fermi level, the higher the threshold for photon
unoccupied state.
absorption.
C
0
E
Conduction Band
E
EF
EF
Photon
hv
EG
Valence Band
Figure 19: An illustration of photon absorption by a valence electron for a h eavily doped material. The Fermi
level of the material is within the conduction band.
The absorption coefficient is dependent on the number of unoccupied states
available, and the number of carriers to excite. A simple expression is shown below:
63
f(E,)(1- f(En))
(0.9)
where E, and E, are the energies of the initial and final states, and
distribution for fermions.
f
is the Fermi-Dirac
Given a known Fermi level, expressions for f(En) and
1- f(E,) must be found.
It is useful to find an expression for the electron energy in terms of other known
quantities, such as photon energy, and effective mass.
First, the conduction band
dispersion relation is written.
E =
kBs is the Burstein shift.
h2k 2
BS
2m
+E
g
(0.9)
Next, a relation between photon energy and the band
dispersions is written.
hw=E +E =
BS+E
k = 29 (ho) - E)
(09)
(0.9)
Using the expression for photon energy, Eq. (0.9), the conduction band dispersion can be
re-written in terms of the photon energy and bandgap. Re-arranging Eq.(0.9) into Eq.
(0.9), and then combining with Eq. (0.9), results in Eq. (0.9), an expression for the
conduction band dispersion rewritten in terms of photon energy, bandgap, and other
material parameters:
En= mrh2(ho)-E)
m,e
(0.9)
This result for the electron energy can be inserted into the Fermi-Dirac distribution, to
calculate the filling expression in Eq. (0.9).
64
The number of unoccupied states for electrons to be excited to is given below.
1- f(E,,)=l-
1(0.9)
m (ho - E ) E -E
exp( '
9 + 9.
f)+1
ex
mkBT
BT
For this expression, the Fermi level must be known or calculated. The photon energy is a
controllable variable in the measurement, and the bandgap energy can be fitted to the
measurement data.
The number of carriers available for excitation f(E,) is given below, using Eq.
(0.9) the expression for electron energy. Note that the hole effective mass is used, instead
of the electron effective mass. To calculate the occupation level of the valence band, Eq.
(0.9) in the previous derivation must be changed to reflect the energy dispersion for the
valence band.
1
Ed
fm(Er(h
exp(
k)+I
h
Ef
(0.9)
-
mhkBT
kBT
Combining the expressions for joint density of states, and the Fermi filling
fractions, Eq. (0.9), (0.9), and (0.9), a final expression
a ~ Cp(ho)f(E,)(l - f(E,))
(0.9)
can be obtained to show the frequency dependent and doping dependent behavior for
semiconductor optical absorption.
C is a constant, and p(ho) is the joint density of
states.
3.2.3
MATLAB Model - Direct Gap Absorption
A MATLAB model was created to simulate the absorption coefficient dependence in
Eq.(0.9). Literature values for electron and hole effective mass were used.66 Various
65
carrier concentrations were used, to view the shift in the absorption edge. As expected,
Figure 20 shows the absorption edge shifting to higher energies as the material is more
heavily doped.
3.6e16cm3
6e17cm3
- -3.8e1 8cm3
--
U)
4
3
C
0
--- --- --- --- -- --
0
0.0
0.5
1.0
Energy (eV)
Figure 20: Modeled absorption coefficient dependence on photon energy for InAs samples of different doping
levels.
3.2.4
Determining the Band Gap - Direct Absorption
For a lightly-doped semiconductor, the band gap can be determined directly from
absorption measurements. Eq.(0.9) shows that the absorption coefficient is proportional
to (ho - E.
)12
.
To determine the precise band gap, the square of the absorption
coefficient should be plotted versus the photon energy. The band gap can be found by
determining the intercept of the plot with the energy axis. Figure 21 shows the literature
values of InAs' absorption coefficient squared, plotted versus energy.
The blue line
contains the literature values, while the red line shows the drawn linear slope.
intersection of the red line with the x-intercept is the direct bandgap energy.
66
The
3 101
-U
2
3x10 2
E
Absorption Coefficient Squared
Bandgap Fit
--
n 2x10 1 2
0*
U/)
S1x1012.
L-)
00
0.0
0.2
0.4
0.6
0.8
Energy
Figure 21: The bandgap of InAs can be determined by drawing a linear slope to the x-intercept energy. The red
line is the literature values of absorption coefficient squared for undoped
InAs 6
.
The green line shows the
drawn linear slope. InAs' bandgap is 0.35eV.
3.3
Fundamental Absorption - Indirect Bandgap
In indirect bandgap semiconductors, electrons cannot be promoted from the
valence band to the conduction band via photon absorption alone.
Instead, a two-step
process occurs with the addition of a phonon emission or absorption.
This additional
phonon process is required to make up the momentum difference between the valence
and conduction band extrema.
transition
of an
electron,
Because two processes must occur for the interband
indirect
semiconductors
have
much
lower
absorption
coefficients compared to direct semiconductors.
3.3.1
Joint Density of States - Indirect Bandgap
For an indirect semiconductor, the joint density of states must be re-derived to
account for the electron momentum shift from the phonon absorption/emission process.
The joint density of states expression given in Eq. (0.9) assumes that the electron
momentum is unchanged. The general form of the joint density of states will depend on
67
the density of states of each the conduction and valence bands, taking into account the
additional energy gain/loss from the phonon. Eq.(0.9) shows the general form of the joint
density of states for indirect absorption.
p(ha)oc
J
( E )p, (E,)dE,
(0.9)
For the indirect case, the energies E, and E, will have to be redefined, to include ke, the
wave vector of the conduction band minimum.
2m
+E9
(0.9)
E=
h2(k n- kC)2
"
2m,
EP =-
g
P
(0.9)
2mh
"
The energy balance should be redefined also, where hoq represents phonon absorption
with oq as the frequency of a phonon with k bridging the conduction and valence band
extrema.
hW + hq = En+ E,
(0.9)
Likewise, the density of states pc for the conduction band, and pv for the valence band are
defined below:
1 ( 2 me)(
2
p(E
)3/2, -E)
n
p,(E)=
2m
(
2
(0.9)
n
)3/2
(E)12
(0.9)
In order to solve the integral in Eq.(0.9), it would be useful to change the energy variable
in the conduction band density of states, Eq.(0.9). Eq.(0.9) relates Ep to En, and the joint
density of states integral can be written in terms of only Ep.
68
It is convenient to introduce an energy 6, for the integral limits.
3 = ho + hq - E9
(0.9)
Intuitively, 6 represents the maximum kinetic energy of an electron promoted to the
conduction.
Finally, the indirect band gap transition joint density of states can be written as
follows, using Eq.(0.9), Eq.(0.9), Eq.(0.9), and Eq.(0.9).
1
1
p(hw)=
p(h1)=
47
4M e M
4m m
2(
4
h
/
32
12
)/
E )" (E,)12
h )3/2-(hW+hW
8
q
-E
)2
(0.9)
(0.9)
Eq.(0.9) shows the joint density of states, and hence the absorption coefficient,
proportional to the square of the photon energy.
3.3.2
Phonon Population Dependence
In indirect band gap fundamental transitions, the phonon population plays an
important role in the absorption coefficient. The larger the population, the more likely
that the phonon absorption two-step transition process occurs.
Likewise, at lower
temperatures, phonon emission is the dominant process, due to the lower population of
available phonons for absorption.
In the phonon absorption case, the probability is related to the Bose-Einstein
distribution for phonons. The absorption coefficient relationship is below, using the joint
density of states from Eq.(0.9).
exp(hw q kBT) -
69
(0.9)
(
a oc p(hw )
In the phonon emission case, the density of states should be rederived, using -hoq in the
energy expressions.
The absorption coefficient is related to the Bose-Einstein
distribution + 1.
a oc p(ho)(1+
3.3.3
1
exp(hWoq / kBT) -1
(0.9)
Determining the Bandgap - Indirect Absorption
Determining the bandgap in indirect semiconductors is not as straightforward as
in direct semiconductors, due to the absorption coefficient dependence on the phonon
population; the phonon population is dependent on the temperature.
Nevertheless, if
these effects are ignored, an approximate method to determining the bandgap can be used.
From Eq.(0.9), it is seen that the joint density of states in the indirect bandgap case is
dependent on (ho+hoq -Eq )2 .
Therefore, if the square root of the absorption
coefficient is plotted with respect to energy, a linear slope can be used to approximate the
bandgap.
3.4
Heavily Doped Semiconductors
Heavily doped semiconductors require additional analysis to determine the
bandgap from absorption measurements. In the previous sections, the doping effects on
semiconductors were considered by adjusting the joint density of states with the FermiDirac distribution, to account for partially filled occupation levels in the bands by carriers.
In theory, experimental absorption data on doped semiconductors can be analyzed using
Eq. (0.9), to determine what the absorption data would look like without the effect of
carriers. However, for heavily doped semiconductors, the Fermi level will be relatively
70
high above the band minimum (in the case of an n-type material). According to FermiDirac statistics, the band states near the band minimum will be highly filled, and very
little light absorption will occur. Experimentally, no useful signal can be collected and
manipulated using Eq. (0.9).
Given that absorption data from heavily doped
semiconductors contain no useful information on the bandgap, a different method must
be used to extrapolate the bandgap.
It will be shown that the Fermi-level can be
approximated using the absorption data. Coupled with carrier concentration data, the
bandgap can be calculated. Once the Fermi level has been determined from the PAS
measurements, the bandgap can be estimated through the following algorithm illustrated
in Figure 22.
E
EB
EA = EF(hw)
E
k
EC
Figure 22: Schematic showing the estimation of the bandgap from Fermi-level measurements.
EA
is the Fermi level measured through the absorption coefficient, and is in terms of the
photon energy.
EB is the chemical potential, and is related to the doping level of the
semiconductor. Ec is the energy in the valence band associated to
absorption.
By taking E,
-
E1
in the fundamental
(E, + E,,), the bandgap energy can be estimated.
The
challenge lies with accurately determining the Fermi-levels EA, EB, and Ec. The next few
71
paragraphs will discuss in detail determining of these three energies, and the difficulties
encountered.
EA
is the Fermi level in terms of the photon energy, and can be determined by
making the approximation that the "knee" in the Fermi-Dirac distribution is related to the
beginning of the steep increase in the absorption coefficient, marking the absorption edge.
The Fermi-Dirac knee is determined by drawing a linear slope through the absorption
edge. In Figure 23, the knee of the red curve (doped InSb) is labeled, and corresponds to
the point labeled in the Fermi-Dirac distribution.
Absorption Edge Shift Due To F-D Distribution
f(E,T)
Fermi-Dirac Distribution
-
1.E+06
"Fermi-Dirac shift"
.! 8.E+05
Q
- "Fermi-Dirac knee"
TwOK
o .E+05
-Undoped
0
3.E+05
-Doped
T> K
0
0.E+00
'Er
0.1
0.2
0.3
0.4
0.5
0.6
No Absorption
Absorption starts
(Undoped EG)
Energy (eV)
Figure 23: Diagram relating Fermi-Dirac "knee" to steep increase in absorption coefficient.
The smooth curve of the Fermi-Dirac distribution is approximated as a linear slope, equal
to the slope at E=Er. The intercept of the linear slope with the energy axis is the FermiDirac knee. Because the Ferni-Dirac distribution is known, the relationship of the "knee"
to the Fermi-level can be determined. For room temperature, the knee is approximately
0.05eV below the Fermi-level. For the InSb test case, the data used is taken at 130K. For
130K, the "knee" is 0.022eV below the Fermi-level. Thus, for the doped InSb sample in
Figure 23, the Fermi-level is determined to be about 0.45eV.
72
Once the Fermi level has been determined from the PAS measurements, the
energy leveled filled by the degenerate doping must be determined (EB and Ec). Carrier
concentrations can be measured by Hall effect measurements. For the literature data used,
the carrier density was already measured. The energy levels filled by the free carriers can
be calculated by doing bandstructure calculations. For this work, the Kane model for
non-parabolic bands was used. A parabolic band model was tested first, but it was found
that at the high doping levels of the InSb, the parabolic band model was not very accurate.
In the future, density functional theory can be used to accurately calculate the energies EB
and Ec.
The Kane model 6 7 requires the bandgap as an input, and is explicitly written as
E(I+
EG
)=
h 2k 2
K(0.10)
2m
To deal with the bandgap being unknown, an initial estimate of the bandgap must be
made. An iterative process is used to update the bandgap input, until the bandgap output
matches the bandgap input.
,
For three sets of doped InSb samples, at carrier concentrations of 7.5x10' 7 m-3
2.6x10 " cm-3, 6x10" cm-3, the algorithm in Figure 22 produced bandgaps of 0.17eV,
0.19eV, and 0.16eV.
73
8.E+05
4.E+05
E
jE 4.E+05
-Undoped
0
0
-Doped
0.
L_
0 1.E+02
0.4
0.3
0.2
Energy (eV)
Figure 24: Absorption coefficient for undoped
InSb,
at 130K.
53
The absorption coefficient slope in the doped
InSb (red) is due to a combination of the bandstructure effect, and the band filling effect.
The bandstructure
effect is shown in the absorption coefficient of the undoped InSb (blue)
To check whether the Fermi-Dirac "knee" can actually be approximated by the
absorption coefficient energy-intercept, we will examine the effect of bandstructure
versus the effect of band filling on absorption coefficient by comnparing the slopes of
absorption coefficient between the doped and undoped samples. It can be seen that the
undoped sample's absorption coefficient change is approximately 3.1xl 06m-ieV-1. The
6
doped InSb absorption coefficient change is about double, 7.3x0 m-'eV-1. This means
that the bandstructure effect and the band filling effect are about equal in magnitude,
because the doped InSb absorption coefficient slope is composed of both bandstructure
and band filling contributions.
In the case of T=130K, this amounts to an additional
adjustment of -0.022eV in the bandgap determined.
3.5
Summary
For heavily doped semiconductors such as thermoelectrics, the bandgap is
difficult to measure because the absorption coefficient is dependent on both the
74
bandstructure and band filling.
The joint density of states shows how the absorption
coefficient of undoped samples can be used to determine the bandgap of the material, as
well as the gap type (direct vs. indirect). For lightly doped materials, where there is less
but still significant absorption in the bandgap region, the absorption coefficient can be
modified by taking into consideration the Fermi-Dirac distribution, to obtain the
absorption coefficient contribution due to bandstructure.
This is the absorption
coefficient predicted by the joint density of states, from which the bandgap can still be
determined. In the case of heavily doped materials, gap information in the absorption
coefficient is lost, due to extreme band filling.
Given material parameters such as
effective mass, the Fermi level and band gap can be determined by fitting experimental
data to absorption coefficient models.
75
Chapter 4: Summary and Future Work
The work presented in this thesis aims to overcome the challenge of accurately
and efficiently measuring the infrared bandgaps of heavily doped semiconductors.
commercial FTIR with a PAS detector was used in this work.
A
From a single
Photoacoustic spectroscopy measurement, both the thermal conductivity and the optical
absorption coefficient can be measured. However, the specific heat of the sample needs
to be measured separately. Directly calculating the photoacoustic response of a sample is
difficult, because there are many experimental unknowns in a commercial FTIR-PAS
setup. A relative measurement using a reference sample was devised to circumvent these
unknowns, and allow measurement of sample properties without knowing experimental
parameters such as light input power or microphone gain. The absorption coefficient
measured with the photoacoustic method directly gives information about the
bandstructure and band filling of the semiconductor.
In addition to measuring
the absorption coefficient
of heavily doped
semiconductors, the work presented in this thesis involved analyzing the data to
extrapolate the true bandgap from the measured absorption edge.
In heavily doped
semiconductors, the challenges are two-fold: the absorption edge is raised in energy from
the bandgap region due to extreme band-filling, and the band minimum region yields
little useful information to "see" a bandgap.
Theory is provided to account for the
Burstein-Moss absorption edge shift, in the case where bandgap features are still visible
in the absorption coefficient.
In addition, a method is proposed to extrapolate the
bandgap in heavily doped materials where no useful bandgap information is gained from
the absorption coefficient.
76
Future work in this area should aim towards further extending the theoretical
work presented in the second part of this thesis. The bandgap extrapolation method relies
on knowing the effective mass of the material, which is generally only well measured for
pure, high-quality semiconductors.
The effective mass most likely changes in heavily
doped semiconductors, such as thermoelectrics. Methods to obtain an effective mass
include the "method of four coefficients", where electrical conductivity, Seebeck
coefficient, Hall coefficient, and Nernst coefficient are measured.
density functional theory simulations to estimate effective mass.
Also possible are
Finally, the effect of
defect states caused by high impurity concentrations are not considered in this work. At
the doping concentrations of thermoelectrics, it can be expected that defect states will
play a large role in the carrier concentration.
77
Chapter 5: References
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
Committee, S. Renewables 2013. 1-178 (2013).
https:/flowcharts.llnl.gov.
Nolas, G., Poon, J. & Kanatzidis, M. Recent Developments in Bulk Thermoelectric
Materials. MRS bulletin 31, 199 (2006).
Snyder, G. J. & Toberer, E. S. Complex thermoelectric materials. Nature Materials
7, 105-114 (2008).
Riffat, S. B. & Ma, X. Thermoelectrics: a review of present and potential
applications. Applied Thermal Engineering23, 913-935 (2003).
Sofo, J. 0. & Mahan, G. D. Optimum band gap of a thermoelectric material. Phys.
Rev. B 49,4565 (1994).
Winder, E. J., Ellis, A. B. & Lisensky, G. C. Thermoelectric Devices: Solid-State
Refrigerators and Electrical Generators in the Classroom. 1-7 (1997).
Sharp, J., Bierschenk, J. & Lyon, H. B. Overview of Solid-State Thermoelectric
Refrigerators and Possible Applications to On-Chip Thermal Management. Proc.
IEEE 94, 1602-1612
GYLFE, J. & WIMMER, R. REACTOR-THERMOELECTRIC POWER
SYSTEMS FOR UNMANNED SATELLITE APPLICATIONS. in Inter-Society
Energy Conversion EngineeringConference (American Institute of Aeronautics
and Astronautics, 2012). doi: 10.2514/6.1966-2308
Thompson, W. On the Dynamical Theory of Heat. Trans. Royal Society of
Edinburgh:Earth Science 3, 91-98 (1851).
He, J., Kanatzidis, M. G. & Dravid, V. P. High performance bulk thermoelectrics
via a panoscopic approach. Materials Today (2013).
doi: 10.1016/j.mattod.2013.05.004
Nolas, G., Morelli, D. & Tritt, T. M. Skutterudites: A phonon-glass-electron
crystal approach to advanced thermoelectric energy conversion applications.
Annual Review of MaterialsScience 29, 89-116 (1999).
Zebarjadi, M. et al. Power factor enhancement by modulation doping in bulk
nanocomposites. Nano Lett. 11, 2225-2230 (2011).
Fleurial, J., Caillat, T. & Borshchevsky, A. Skutterudites: an update.
Thermoelectrics, 1997. ProceedingsICT'97. XVI InternationalConference on 111(2002).
Kim, I.-H. & Ur, S.-C. Electronic transport properties of Fe-doped CoSb3 prepared
by encapsulated induction melting. MaterialsLetters 61, 2446-2450 (2007).
Caillat, T., Borshchevsky, A. & Fleurial, J. P. Properties of single crystalline
semiconducting CoSb3. J. Appl. Phys. 80, 4442 (1996).
Slack, G. A. & Tsoukala, V. G. Some properties of semiconducting IrSb3. J. Appl.
Phys. 76, 1665 (1994).
Dyck, J., Chen, W., Uher, C. & Chen, L. Thermoelectric properties of the n-type
filled skutterudite BaCoSb doped with Ni. JournalofApplied ... (2002).
Tang, X., Zhang, Q., Chen, L., Goto, T. & Hirai, T. Synthesis and thermoelectric
properties of p-type-and n-type-filled skutterudite RyMxCo4-xSb 12 (R: Ce, Ba,
YM: Fe, Ni). J. Appl. Phys. 97, 3712 (2005).
78
20.
21.
22.
23.
24.
25.
26.
Shi, X., Salvador, J., Yang, J. & Wang, H. Thermoelectric Properties of n-Type
Multiple-Filled Skutterudites. JournalofElec Materi 38, 930-933 (2009).
Nolas, G. S., Cohn, J. L. & Slack, G. A. Effect of partial void filling on the lattice
thermal conductivity of skutterudites. Phys. Rev. B 58, 164 (1998).
Sales, B., Mandrus, D. & Williams, R. Filled skutterudite antimonides: A new
class of thermoelectric materials. Science 272, 1325 (1996).
Tang, X., Chen, L., Goto, T. & Hirai, T. Effects of Ce filling fraction and Fe
content on the thermoelectric properties of Co-rich CeyFexCo4- xSbl2. Journal
of Materials Research 16, 837 (2001).
Peng, J. et al. High temperature thermoelectric properties of double-filled
InYbCoSb skutterudites. J. Appl. Phys. 105, 084907 (2009).
Yang, K. et al. Synthesis and thermoelectric properties of double-filled
skutterudites CeyYb0. 5-yFel. 5Co2. 5Sbl2. JournalofAlloys and Compounds
467, 528-532 (2009).
Tang, X., LI, H., Zhang, Q. & Niino, M. Synthesis and thermoelectric properties of
double-atom-filled skutterudite compounds Ca m. JournalofApplied ... (2009).
27.
28.
29.
30.
31.
32.
33.
34.
35.
36.
37.
38.
39.
Zebarjadi, M., Esfarjani, K., Yang, J., Ren, Z. F. & Chen, G. Effect of filler mass
and binding on thermal conductivity of fully filled skutterudites. Phys. Rev. B 82,
195207 (2010).
Stoica, M. & Lo, C. S. Electrical transport properties of Co-based skutterudites
filled with Ag and Au. arXiv cond-mat.mtrl-sci, (2012).
Singh, D. J. & Pickett, W. E. Skutterudite antimonides: Quasilinear bands and
unusual transport. Phys. Rev. B 50, 11235 (1994).
Sofo, J. & Mahan, G. Electronic Structure and Transport Properties of CoSb3: A
Narrow Band-Gap Semiconductor. MRS Proceedings545, (1998).
Ackermann, J. & Wold, A. The preparation and characterization of the cobalt
skutterudites CoP 3, CoAs 3 and CoSb 3. JournalofPhysics and Chemistry of
Solids 38, 1013-1016 (1977).
Mandrus, D. et al. Electronic transport in lightly doped CoSb_ {3 }. Phys. Rev. B
52, 4926 (1995).
Stepanov, N. P., Nemov, S. A., Zhitinskaya, M. K. & Svechnikova, T. E. Optical
properties of doped bismuth telluride crystals in the region of plasma effects.
Semiconductors 41, 786-789 (2007).
Sablikov, V. The photoacoustic effect in semiconductors. physica status solidi (b)
(1983).
Schmid, T. Photoacoustic spectroscopy for process analysis. Analytical and
bioanalyticalchemistry (2006).
Vargas, H. & Miranda, L. Photoacoustic and related photothermal techniques.
Physics Reports 161, 43-101 (1988).
Eaves, L., Vargas, H. & Williams, P. Intrinsic and deep-level photoacoustic
spectroscopy of GaAs (Cr) and of other bulk semiconductors. Appl. Phys. Lett. 38,
768-770 (1981).
Rosencwaig, A. Photoacoustic spectroscopy. Annual Review ofBiophysics and
Bioengineering9, 31-54 (1980).
Rosencwaig, A. & Gersho, A. Theory of the photoacoustic effect with solids. J.
Appl. Phys. 47, 64-69 (1976).
79
40.
41.
42.
43.
44.
45.
46.
47.
48.
49.
50.
51.
52.
53.
54.
55.
56.
57.
58.
59.
60.
61.
62.
Fan, H. Y. Infra-red absorption in semiconductors. Reports on Progress in Physics
19, 107 (1956).
ROSS, I. M. & SAKER, E. W. XXVIII. Applications of Indium Antimonide.
Journalof Electronicsand Control 1, 223-230 (1955).
Aamodt, L. C., Murphy, J. C. & Parker, J. G. Size considerations in the design of
cells for photoacoustic spectroscopy. J. Appl. Phys. 48, 927 (1977).
Jones, R. W. & McClelland, J. F. Phase references and cell effects in
photoacoustic spectroscopy. Applied Spectroscopy 55, 1360-1367 (2001).
Foster, P. J. & Howarth, C. R. Optical constants of carbons and coals in the
infrared. Carbon 6, 719-729 (1968).
Williams, M. W. Optical properties of glassy carbon from 0 to 82 eV. J. Appl.
Phys. 43, 3460 (1972).
Noda, T., Inagaki, M. & Yamada, S. Glass-like carbons. Journal ofNonCrystallineSolids 1, 285-302 (1969).
Katerberg, J. A. & Anderson, A. C. Low-temperature specific heat, thermal
conductivity, and ultrasonic velocity of glassy carbons. JournalofLow
TemperaturePhysics 30, 739-745 (1978).
Takahashi, Y. & Westrum, E. F. Glassy Carbon low-temperature thermodynamic
properties. The Journalof Chemical Thermodynamics 2, 847-854 (1970).
Parker, W. J., Jenkins, R. J., Butler, C. P. & Abbott, G. L. Flash Method of
Determining Thermal Diffusivity, Heat Capacity, and Thermal Conductivity. J.
Appl. Phys. 32, 1679 (1961).
Levinshtein, M., Rumyantsev, S. & Shur, M. HandbookSeries on Semiconductor
Parameters.(World Scientific, 1995).
Steigmeier, E. F. & Kudman, I. Thermal conductivity of III-V compounds at high
temperatures. PhysicalReview 132, 508 (1963).
Gobeli, G. W. & Fan, H. Y. Infrared absorption and valence band in indium
antimonide. PhysicalReview 119, 613 (1960).
Ukhanov, Y. I. OpticalPropertiesofSemiconductors. (Nauka Publishing
Company, 1977).
Kurnick, S. W. & Powell, J. M. Optical absorption in pure single crystal InSb at
298 and 78 K. PhysicalReview 116, 597 (1959).
Visvanathan, S. Free Carrier Absorption Arising from Impurities in
Semiconductors. PhysicalReview (1960).
Schroder, D. & Thomas, R. Free carrier absorption in silicon. Solid-State Circuits
(1978).
Das, A. K. & Nag, B. R. Free-carrier absorption and electron mobility in n-type
PbTe. JournalofPhysics and Chemistry ofSolids 39, 259-267 (1978).
Fujiwara, H. & Kondo, M. Effects of carrier concentration on the dielectric
function of ZnO:Ga and In203:Sn studied by spectroscopic ellipsometry: Analysis
of free-carrier and band-edge absorption. Phys. Rev. B 71, 075109 (2005).
Dresselhaus, M. S. Optical Properties of Solids. 1-253 (2001). at
<http://web.mit.edu/course/6/6.732/www/texts.html>
Blatt, F. J. Physics of electronic conduction in solids. (1968).
Palik, E. D. Handbookof Optical Constants ofSolids. (Academic Press, 1997).
Hamaguchi. Basic Semiconductor Physics. (Springer, 2004).
80
63.
64.
65.
66.
67.
Pankove, J. I.Physics. Optical Processes in Semiconductors (DoverBooks on
Physics). (Dover Publications, 2010).
Zawadzki, W. Electron transport phenomena in small-gap semiconductors.
Advances in physics 23, 435-522 (1974).
PHILIPP, H. R. & Ehrenreich, H. Optical Properties of Semiconductors. Physical
Review 129, 1550-& (1963).
Ioffe.ru - Bandstructureand carrierconcentrationof Indium Arsenide (InAs). at
<http://www.ioffe.ru/SVA/NSM/Semicond/InAs/bandstr.html>
Kane, E. Band structure of indium antimonide. JournalofPhysics and Chemistry
of Solids (1957).
81