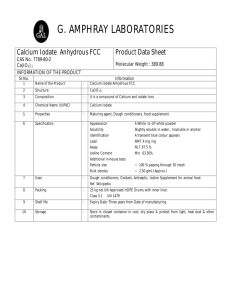
THE MARINE CHEMISTRY OF IODATE B.S., SUBMITTED IN PARTIAL FULFILLMENT
advertisement

THE MARINE CHEMISTRY OF IODATE by GEORGE TIN FUK WONG B.S., California State College, Los Angeles 1971 SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE the at MASSACHUSETTS INSTITUTE OF TECHNOLOGY June, 1973 Signature of Author. .... th Depart en to ofE a Planetary Sciences Feb. 21, 1973 Certified by.. .0-.....e.. .-- ry .,. ........ _----h-sis Kpervisor Accepted by. .......... .o............... Chairman, Departmental Committee on Graduate Students ACKNOWLEDGMENTS I wish to express my gratitude to Dr. Peter G. Brewer whose invaluable advice, guidance and made encouragement this research enjoyable and possible. I also want to thank Dr. John M. Edmond for his encouragement and for the instructive discussions on this study. I would also like to thank especially Mr. Roy Smith for his time and effort in collecting the samples for me during the Geosecs cruise. The helpful comments from Dr. Nelson Frew and from the fellow graduate students are also very much appreciated. -ii- TABLE OF CONTENTS Page . i Acknowledgments............................... ii Approval Page.... Table of .. 0 . . . . . . . . . . . Contents......................... . iii . List of Figures.... vi List of Tables................................ viii Abstract ... .. .......... ......... ......... . x . . 1 Chapter I. Introduction...... . . . . . . . . . . . . A. Purpose of the study.................... 1 B. Iodine in the sea....................... 1 C. Analytical methods for the analysis of iodine II. The in analytical 11 seawater.....................,. 23 method........................ A. The feasibility of a direct method...... 23 . . .. 24 B. Initial te t i. . . . . . . . . . . The Beer's Law region for the tri-iodide ion..................... 0 24 ii. Determination of the iodate concentration in a standard iodate solution using the standard addition method...............,.... -111- 27 iii. Experimental conditions ............ 27 iv. Theoretical interferences.......... v. The tri-iodide ion absorption curve. ............ ..... . ....... 29 32 C. The analyticalmethod................... 34 i. Instrumentation.................... 34 ii. Standard solutions................. 34 iii. Procedure for the analysis of iodate in seawater................. iv. The precision of the analytical methods ........ v. 43 Correlation between the addition method and the titration method.... vi. 38 50 Correlation between the addition and the Matthews and Riley's ,.. .... .... meth ds, ... .... ... 52 D. Attempts for developing an analytical method for the determination of iodide seawater........................ 55 storage................................ 58 in III. IV. Sample A. A qualitativestudy..................... 58 B. A quantitative study.................... 60 The distribution of iodate in the sea......... -iv- 63 A. Seasonal variations in the iodate content of the Woods Hole coastal surface water....... .......... ..... . 63 B. Diurnal variations in the iodate content in seawater..................... 66 C. The distribution of iodate in the Argentine Basin in the South Atlantic... V Conclusion.... VI. Future work . . . . . .............................. ...... * .. 0... ....... Refe .... enc ...s.. ... 97 ..... ... ... 99 # a 0 # .... ... ... 102 Appendix 1....................................0 a 00*0 Appendix 2. . . . . . . . . .. . . . . . 70 . . . 10 9 ll LIST OF FIGURES Page Figure I-1. Iodine profiles from the Pacific and their station positions.................. Figure 1-2. Variations in iodide-iodine and total iodine in the Pacific................... Figure 1-3. The concentration diagram of the iodideiodate couple at a pH of 8.1 and with a total iodine concentration of 0.4 Figure I-4. The analytical scheme for the analysis of iodide and iodate by co-precipitation... . . . . . . . . . . . . . . . . Figure II-1. The Beer's Law region of the tri-iodide ion absorption at 353 nm. in a 10 cm. 20 26 Figure 11-2. The tri-iodide ion absorption curve...... 33 Figure 11-3. The apparatus............................ 35 Figure 11-4. The analytical scheme for the analysis of iodate and total iodine............... 39 Figure 11-5. A sample plot for the photometric micro- Figure II-6. A sample plot for the determination of iodate in seawater by the addition Figure IV-1. Seasonal variations in the iodate content of the Woods Hole coastal surface Figure IV-2. 49 water.......................... Diurnal variations in the iodate content and the corresponding changes in the phosphate concentration of the Woods Hole coastal water.................,.. -vi- 65 68 Figure IV-3. Cruise track of R/V Knorr, Geosecs Cruise, Leg 5, and the locations of stations 60 and 61...................... 71 The nitrate, phosphate and silicate profiles of station 60................... 76 The oxygen, salinity and potential temperature profiles of station 60 with the water masses identified.............. 77 The nitrate and phosphate profiles of station 61 78 Figure IV-7. The oxygen profile of station 61......... 79 Figure IV-8. The salinity and potential temperature profiles of station 61 with the water Figure IV-4. Figure IV-5. Figure IV-6. masses identified..................... . 79 Figure IV-9., The iodate profile of station 60......... 81 Figure IV-10. The iodate profile of station 61......... 82 Figure IV-ll. The relationship between phosphate and iodate. .T........ ...... . ... ..... . Figure IV-12. The relationship between nitrate and iodate....**.....................,..... 86 87 Figure IV-13. The relationship between AOU! and iodate.,. 88 Figure IV-14. Vertical profile through the western basin of the Atlantic Ocean showing idealized water-mass distribution........ 91 -vii- LIST OF TABLES Page Table I-l. Some molecular properties of iodate and iodide. ........... Table 1-2. ...... . . .. * ... 5 Mean concentration of iodine in the surface water............................. Table 1-3. P/I ratio in some algae................... Table 1-4. Analytical methods for the analysis of iodine in seawater........................ 12 13 14 Results for testing the Beer's Law region of the tri-iodide ions at 353 nm. in a 10 cm. cell..... .. .............. . 25 Results for the determination of iodate concentration in a standard iodate solution.................................. 28 Table 11-3. Some possible interfering species......... 30 Table 11-4. Results for the determination of the precision of the addition method.......... 48 Data for determining the precision of the photometric micro-titration method.... 45 Table II-1. Table 11-2. Table 11-5. Table 11-6. Table 11-7. Correlation between the addition method and the titration method.................. 51 Correlation between the addition method and the Matthews and Riley's method..... 53 Table III-1. The qualitative effect of storage on the iodate content in seawater by various storage methods ....................... Table 111-2. A quantitative study on the effect of storage on the iodate content in seawater by controlling the storage temperature.... -viii- 59 61 Table IV-l. Seasonal variations in the iodate content of the Woods Hole coastal surface water.... 64 Table IV-2. Diurnal variations in the iodate content and the corresponding changes in the phosphate concentration of the Woods Hole coastal water.............................. 67 Table IV-3. Nutrient data from station 60.............. 72 Table IV-4. Nutrient data from station 61.............. 74 -ix- l ABSTRACT THE MARINE CHEMISTRY OF IODATE GEORGE TIN FUK WONG Submitted to the Department of Earth and Planetary Sciences on March 1, 1973 in partial fulfillment of the requirements for the degree of Master of Science. An analytical technique has been developed for the analysis of iodate in seawater. In this method, iodate is reduced to iodine by excess iodide in an acidic solution and the iodine thus formed is reacted with excess iodide to form the tri-iodide complex. The tri-iodide ion concentration is determined spectrophotometrically at 353 nm. by a series of additions of known amounts of a standard iodate solution. This method has a coefficient of variation of 2.7%. Alternatively, the tri-iodide ion concentration can be determined by photometric micro-titration with a standard thiosulfate solution with a coefficient of variation of 0.7%. A comparison of the results from these two techniques and from a standard method shows that they agree to within experimental uncertainties. The new method is faster than the standard method, is of comparable precision and can be performed on board ship for rapid routine analysis of iodate. Attempts to design a similar method for the analysis of iodide in seawater have so far been unsuccessful. Storage experiments show that seawater can be stored at low temperature for up to a month without significant alteration of its iodate content. Studies on the diurnal and seasonal variations of iodate are inconclusive. The iodate content in the surface water at the Woods Hole coast is only about one half of that in the South Atlantic surface water. The total iodine concentration is rather uniform in the sea. If iodate is consumed by the reduction of iodate to iodide by bacteria, this difference in iodate concentration may be due to the higher biological activity in coastal waters. Iodate profiles from samples of stations 60 and 61 of the Geosecs cruise, leg 5, in the Argentine Basin in the South Atlantic show a surface depletion, a maximum at around 1500m., followed by a minimum and then a gradual increase towards the bottom. The iodate profiles resemble the profiles of the nutrients nitrate and phosphate. Phosphate-iodate, nitrateiodate and AOJ-iodate graphs show linear relationships with correlation coefficients of 0.94, 0.95 and 0.88 respectively. The molar ratios from these plots are AOU:nitrate:phosphate: iodate = 2440:357:22.7:l or AOU:nitrate:phosphate = 108:16:1. This is close to the reported ratios of AOU:nitrate:phosphate = 138:16:1. From these ratios, it may be calculated that the recycled iodate is 7-8% of the total iodate and that 90% of the iodate is conservative, Thesis Supervisor: Peter G. Brewer Title: Associate Scientist Department of Chemistry Woods Hole Oceanographic Institution Woods Hole, Massachusetts -xi- CHAPTER I INTRODUCTION A. Purpose of the study The purpose of this study was to examine the marine chemistry of iodine in order to assess its involvement in the biological cycles of the ocean and its use as an oceanic tracer. The redox chemistry of iodine, and the previously reported variations in seawater showed the potential use of this system. As a first step, it was desirable to develop a rapid, precise and simple method for the analysis of iodate in seawater and then to use this method to carry out analyses of vertical profiles of iodate in the ocean. These data, together with observations of diurnal and seasonal variations should give important information on the iodine system in seawater. B. Iodine in the sea The presence of iodine-containing substances has been known to man for a long time. It is believed that a legendary Chinese emperor Shen-Nung, said to have lived in the third millenium B.C., mentioned seaweed as an efficacious remedy against goiter in his treatise on herbs and roots (Langer 1960). Iodine can be found everywhere in nature in low con-1- centrations, but elemental iodine was not discovered until the early nineteen century (Courtois 1813). Studies on the iodine system in the marine environment can be traced back to 1825 when Pfaff detected iodine in Baltic seawater, and later, it was also found in French coastal waters by Laurens in 1835 (Riley 1965a). Agreement among the early quantitative determinations of iodine varied widely from 0.017 ug/l (Macadam 1852) to 8000 ug/l (Marchand 1855). Winkler (1916) arrived at a more reasonable value of 38 ug/l. He also suggested that both iodide and iodate were present. The speciation of iodine in the sea has been a rather controversial subject. Sugawara (1957) supported Winkler's proposal because "iodate is the only possible iodine form in higher oxidation state at the hydrogen ion concentration of seawater". Shaw et al.(1957) suggested that hypoiodous acid (HIO) is the dominant oxidized species. His main reasons were: (1) the ratio of oxidized iodine to iodide is about unity, and this ratio can be obtained by assuming a thermodynamic equilibrium in the system of hypoiodous acid, iodide, atmospheric oxygen, water and hydrogen ions at the pH of seawater; (2) if air is equilibrated with seawater containing hypoiodous acid and iodide in equal amount, the iodine concentration observed in marine air will be observed, and, seawater will be able to lose iodine when exposed to iodine-free air as observed; -2- (3) the ratio of total iodine to chloride is higher in warm, partly enclosed sea areas and this can be explained by the effect of temperature upon the equilibria mentioned in (1) and (2) which results in an increase in the content of volatile iodine molecules at lower temperatures; and, (4) the disproportionation of hypoiodite is slow because it involves third order kinetics. Sugawara et al.(1958) performed experiments to verify his own proposal. He showed that: (1) there is no detectable amount of hypoiodite in seawater; (2) the system iodine, iodide, hypoiodite and iodate reaches a thermodynamic equilibrium quickly with iodide and iodate as the predominant components; (3) even in the presence of oxygen, the oxidation of iodide to a higher oxidation state is quite slow; and, (4) the ratio of oxidized iodine to iodide is not unity and it varies from one location to another. Johannesson (1958), in support of Sugawara, showed that: (1) the triiodide ion is not formed in seawater upon the addition of excess iodide unless the seawater is previously acidified. If hypoiodite is present, it will react with excess iodide without acidification; and, (2) the oxidized iodine concentration, estimated from the tri-iodide concentration, will be too large unless it is in the form of iodate. Shaw et al. (1958) finally accepted that the oxidized form of iodine is -3- iodate because of the experimental evidences of Sugawara (1958) and Johannesson (1958). It is presently generally accepted that iodine is present in seawater primarily as iodide and iodate and that the amount of organic iodine is negligible (Sugawara et al.1957). Iodine complexes are generally less stable than those of the other halides (Cotton et al.1962). Since chloride does not form significant amounts of complexes in seawater, it is expected that iodide will behave similarly. Some of the molecular properties of iodide and iodate are compiled in table I-1. The iodine concentration in seawater varies with location (Sugawara et al.1962) and with depth (Tsunogai et al.1971). The iodide concentration may vary from below detection limit to 0.2 uM (ca. 25 ug-I/1) while the range of iodate concentrations is between 0.2 uM (ca. 25 ug-I/1) and 0.4 uM (ca. 50 ug-I/l) (Tsunogai et al.1971). Thus, the total iodine concentration which is the sum of the two will be constant at ca. 0.4 uM. These deviations can best be shown in Tsunogai et al. (1971) and Tsunogai's (1971b) recent data in figures I-1 and 1-2. If iodate and iodide are the only important iodine species in the ocean, one may consider the thermodynamics of the iodate-iodide redox couple: -4- Table I-1. Some molecular properties of iodate and iodide*. Iodate Iodide Formula 103' I" Oxidation state +5 -*1 Radius of iodine atom (1) VI 1.03** VI 2.13** VIII 1.97*** Ionic structure Pyramidal I-0 bond length (A) 1.83 % double bond character 20% 0-bond orbital hybridization sp3 Partial molal volume at 25.3 250C (cm3/mole) * Data compiled from Cotton et al. 1962, ---. 36.22 Douglas et al. 1965, Rankama et al.1950, Whittaker et al.1970, Millero 1971, and Nightingale 1960. ** Co-ordination numberVI. * Co-ordination number VIII. -5- Figure I-1. Iodine profiles from the Pacific and their station positions. Tsunogai et al.1971, Tsunogai 1971b. Fig. I-la. The vertical distribution of iodine at 120 01'N, 158 0 02'E in the North Pacific where the depth is 5855m. The concentration of iodide(.), iodate (x) and total iodine (o) are indicated. Fig. I-lb. Vertical distribution of iodide-iodine and total iodine with salinity (.) and temperature (x) at the Tonga Trench (23011'S, 174 040'W), where the depth is 10200m. Fig. I-lc. Vertical distribution of iodide-iodine and total iodine with salinity (.) and temperature (x) at 60000s and 169 0 55'W in the Southern Ocean. Fig. I-lb Fig . I-la 3 0.4Jg at/L 0.2jpg at/lt 0.3 0.4 a - Total lide -2 km -3 -5 I T 10I 8 35 *. 35.5 20 *C 30 34.5 s -4 10 0.1 0.3 pgat/l 0.4 0.2 180 cO00O*** -40 dooooooo0000o0o* * KH-70-2 0 0 0o 0 0 00 0 000 e3 0 Oil a KH-70-l *20 0 KH69-4 . -10 *5 Oshoro ~--i S. @N 0 Se1-0 160*E 180i ,. 0 160'110 0 0 10 .10Os S 34 I I 00s 0-. 0 34.5 I 0It 20KH-68-4 * 30- 0 Ti: 0 180 6 40- 0 *. - Fig. I-ld Fig, I-lc -6- SIC 50'6- Figure 1-2. Variations in iodide-iodine and total iodine in the Pacific. Tsunogai et al.1971. Fig. I-2a. The concentration of iodide-iodine in the equatorial Pacific along 170 0W section. Circles and triangles in the figure show the sampling location, and depth and the ranges of concentration which are 0.025-0.05 (A), 0.15 (.) 0.05-0.10 (A), 0.10- and 0.15-0.21 (o) in the unit of ug-at/l. Fig. I-2b. The concentration of total iodine in the same station as Fig. I-2a. Solid circles show the concentration range of 0.36-0.39 ug-at/l and open circles show range of 0.39-0.42 ug-at/l. a S I a a oi N 40a o 00 8 o 0 0 0 c 0 00 0 0 c 0 0000 00 0 @00 00 tJ00o .000 "Do0 0 *0 G00 00o 000 0 4 0 a 0 0 a I 103- + 6H+ + 6e- = I' log K = 110..l + 3H2 0 (Bjerrum et al.1958). Therefore, K= [I-I 6 [103-] [H+] 6 [ej or, log |0O3~ = 6pH + 6pE - log K ---- (1) [I-] In normal seawater, pH = 8.1, and pE = 12.5 (Sillen 1961). Therefore, [103] = [I-] The equilibrium concentrations of both species can also be shown by a concentration diagram as in figure 1-3. Total iodine concentration is assumed to be 0.4 uM and the pH is fixed at 8.1. The concentration of each species at various pE can be calculated from equation (1). This diagram indicates that, at equilibrium, the amount of iodide in seawater is negligible. However, experimental findings show small but significant concentrations of iodide and this dis-equilibrium has been noted for some time (Goldberg 1963; Sillen 1961; Riley et al. 1971). Recently, Tsunogai et al.(1969) proposed that this dis-equilibrium is caused by biological activity of bacteria. They demonstrated that bacteria, that are capable of reducing nitrate to nitrite, can also reduce iodate to iodide enzyma-8- Figure 1-3. The concentration diagram of the iodide-iodate couple at a pH of 8.1 and with a total iodine concentration of 0.4 ug-at/l. -6- -7 -6 I I I LOG CONCENTRATION -5 I (LOG -~ I uM) -2 - I I 0, tri tically. The enzyme used may be nitrate reductase. Although the importance of iodine as a nutrient for marine plankton has not been firmly established, recent work has shown that marine plankton do take in and excrete iodine, mostly in the form of iodide (Kolehmainen et al.1969; Kuenzler 1969). It is also known that iodine is involved in the biological activity of some species and is essential for red algae, brown algae and mammals (Bowen 1966). The metabolism of iodine in the higher animals has been well documented (Gross 1962). For plants, a suitable amount of-iodine is necessary in order to provide a favorable growth stimulation (Hanson 1963). In all marine species, iodine can be found in their body tissues (Hanson 1963). If the iodate and iodide concentrations in the oceans are controlled mainly by biological activity, environmental changes that can vary the amount of biological activity will be reflected in the iodate-iodide ratio. These environmental changes may be seasonal variations, latitude changes, temperature changes, etc. Since biological activity is highest in surface waters, the greatest variations may be observed there. Some work has already been done in this direction in the Pacific (Tsunogai et al.19711 Tsunogai 1971b). It has been observed that the highest iodide concentration is in the sur- -10- face water. In the surface layer, the concentration of iodide is higher in warm water (that is, above 200C, average concentration = 0.1 ug-at/l) than in cold water (that is, less than 2000, average concentration = 0.03 ug-at/1). Among the warm waters, the highest iodide concentration is found in the equatorial zone (average concentration = 0.13 ug-at/1) where the biological activity is also high. Table 1-2 is a summary of their results. If iodine is being taken in by plankton, it may bear some relationship to the other nutrients (for example, phosphate and nitrate). From Vinogradov's (1953) data, one can see that the P/I ratio is approximately constant within a single species despite the crudeness of the data (see table 1-3). C. Analytical methods for the analysis of iodine in seawater The low abundance and the narrow range of concentration of iodate renders a precise and sensitive analytical method a necessity for a reliable quantitative study. Riley (1965) listed the available methods prior to 1965 (see table 1-4). In recent years, a few more methods or modifications of the older methods have been proposed. These methods are also listed and summarized in the same table. The analytical pro-11- Table 1-2. Mean concentration of iodine in the surface water. Tsunogai et al 1971. Location Depth Temp. No. of samples I" IO3' Total I 10-' ug-at/l 0C Lat. Long, m. 20N- 1700W 0 10 12±3 26*3 38±1 30 10 13&3 26*3 39t1 66 10. 14+3 25*5 39*1 100 10 13±4 27*3 40+1 Vari- 170 0W 0 >20 15 10±3 27±2 37t2 0 <20 13 2+1 34+1 36±l 0 >20 15 7±2 28+2 0 <20 3 3±2 31*3 34±1 40 2.5+2 38±3 41*2 ous Vari- 137- 0 ous 163 E Vari- 170 0W >1000 ous -12- Table 1-3. P/I ratio in some algae*. Iodine (I) % Dry Weight Phosphorus (P) I/P % Dry Weight, Ratio 0.27 o,44 60% " 0.20 0.44 45% -U 0022 0.44 50% Stem 0.13 0024 50% Lamina 0018 0.38 50% Nereocystis luetkeana 0.14 0.34 40% Stem 0.07 0.23 30% Leaf 0.1 0.37 30% Macrocystis pyrifera U - * Data from Vinogradov (1953). -13- Table I-4. Analytical methods for the analysis of iodine in seawater. Approx. conc. found (ug/1) Method of separation Method of deterlnination Reference 43 Iodate reduced with As2 O3 Photometric Reith 1930. Titrimetric Skopintsev + HCl, iodine liberated with nitrate and extracted with CC1 4 . 26-56 Oxidized to 103 MnO4, with and Mikhai- reduced the latter with C2042-, treated with lovskaya I- and acid, liberated 1933. iodine titrated. Direct method, depending on Photometric catalytic action of I- on Dubravcic 1955. reaction between Ce4+ and As3+, remaining Ce4+ determined. Total I I- = 7-57 AgOl, converted to 103- co-precipitated with -14- Photometric Sugawara with starch- et al. Table I-4. (continued) Approx. Method of separation conc, found (ug/l) Method ofdetermination Reference I03--I with Br. Reacted wit]h acid- iodine 1955, = 0-5 ified I-, liberated iodine color 1957. measured. Total I by reduction of IO" with S0 3 2 - followed by determination of I~ as described above. Total I IO3- reacted with iodide, Ampero- Barkley et =45-70 liberated iodine treated metric al. 1960a, I0 3-I with excess S20 3 2 -, titration 1960b. which was titrated with iodate. Total I by oxidation of I~ with Br to IO3" which was determined as described above. Total I also deter- Photometric mined by its catalytic with effect on the reaction of brucine Ce4+ with As 3+, Total I Catalytic effect of I~ on Photometric Voipio 1961, =60-118 reaction of Ce4+ with measurement Kappanna et -15- Table 1-4. (continued) Approxr., Method of separation cone. found (ur/1) Method of determination Reference of residual al.1962. As3+, Ce4 Modified Sugawara method, a. Photo- reduction of 103-0 with (NH2 )2 .H2SO. metric mea- et al. 1970. Matthews surement of tri-iodide ions. b. Photometric microtitration with S2032- I--I Improved Sugawara method. Photometric Tsunogai = 0-25 with starch et al.1971; 103--I =25-50 iodine Tsunogai color 1971a, 1971b. Total I 10 3- reacted with iodide- Photometric Schnepfe =55-90 starch mixture directly. with starch 1972. Total I by oxidation of I~ iodine -16.. Table 1-4. Approx. conc. found (ug/l) I03--I =37-64 (continued) Method of separation Method of determination with alkaline KMnO4 to 1O-, which was dete rmined color as described above. -17- Reference cedures of these methods usually involve the titration of liberated iodine or photometric procedures which may depend on: (1) the catalytic action of iodide on the oxidation of arsenic(III) by cerium(IV); (2) the color intensity of extracted iodine; or (3) the color intensit'y of the iodinestarch complex. Many of the procedures require initial concentration and separation steps or a control of the chlorinity. Some of these methods do not distinguish between the iodine oxidation states. Some others have a precision insufficient to detail the range of variations that are expected in the ocean. Partly because of these analytical difficulties, the amount of data obtained thus far on the distribution of iodide and iodate in the sea is very sparse. The data reported by different investigators are also quite different as summarized in table I-4. The more reliable data probably were obtained by Sugawara's (1955) method or its modifications (Tsunogai 1971a; Matthews et al.1970) which are capable of determining iodide and iodate separately. In this method, iodide is coprecipitated as silver iodide with a silver chloride carrier from several hundred milliliters of seawater. The precipitate is separated from the supernatant liquid and then treated with bromine water. Bromine oxidizes iodide to iodate and the excess -18- bromine can be removed by boiling. The iodate can then be converted to iodine with excess iodide-in an acidic solution. Tsunogai (1971a) determined iodine as the iodine-starch complex colorimetrically at 570 rm. and claimed a coefficient of variation of 3%. Matthews et al.(1970) converted iodine to the tri-iodide complex and determined it either spectrophotometrically at 353 nm. or by a photometric micro-titration with a standard thiosulfate solution. They claimed a coefficient of variation of 1% for the spectrophotometric method and 0.4% for the titration methpd. Iodate can be determined similarly after being reduced to iodide. A schematic description of the methods is given in figure 1-4. There are several drawbacks in using these methods. First, they are very timeconsuming. Only about three to four samples can be analyzed for both iodide and iodate in two to three days. Secondly, these methods also involve the use of bromine water, which is rather unpleasant to handle. Besides, the procedure is complicated and cannot be easily automated, or be done at sea. Consequently, these methods are probably not suitable if a large number of samples are to be analyzed. Thus, it would be desirable if a fast, simple and accurate analytical technique can be developed for the routine analysis of iodate and iodide. The Matthews' method is used in this study as a -19- Figure 1-4. The analytical scheme for the analysis of iodide and iodate by co-precipitation. -20- Soluble silver salt e.g. AgNO 3 Seawater Ag+ + I- = AgI Ag+ + Cl- = AgCl Ag+ + Br- = AgBr Supernatant liquid Precipitate (Ag, AgCl, Bromine water AgBr AgI + 3Br 2 + 3H2 0 = 4 2 I03' 103- + AgBr + 5Br- + 6H+ 'Supernatant Precipitate N2H.H2SO4 N2H liquid HSL 103I3 + 3(N 2 H4 .H2 SO4 ) = 21- + 3N2 + 6H20 + 3H2 SO4 ~I 103 AgCl, AgBr * Steps for removing possible interfering species, for example, BrO~. ** For the determination of 12, please see the next page. -21- Fig. 1-4. (continued) Tsunogai's Improved Sugawara Method Matthews and Riley's Method Photometric micro-titration Spectrophotometric Determination standard method for determining the reliability' of the new method. -22- CHAPTER II THE ANALYTICAL METHOD A. The feasibility of a direct method The ideal method will be a simple and rapid method that does not require pre-concentration steps and has a precision of ±3% or less. After a survey of the literature (table 1-4), it seems that a photometric method will be most promising. The feasibility of a direct method will depend on. (1) an absorbing species that can give-a detectable absorption at such a low concentration; (2) absorbances that obey Beer's Law in that concentration range; (3) the absence of other species in seawater that may interfere with the formation of the absorbing species; and, (4) the absence of other species that absorb at the same wavelength as the absorbing species. The most probable absorbing species that may be used seems to be the tri-iodide ion. Tri-iodide ion has been generated in seawater (Johannesson 1958) and it absorbs strongly at 353 nm. Moreover, for each iodate ion, three tri-iodide ions are formed according to the equation: 103~ + 81 313 + 6H+ + 3H2 0, and thus, the absorption will be amplified three fold . With -23- a 10 cm. cell, one may be able to detect a significant absorbance of the tri-iodide ions generated from seawater directly. B. Initial tests i. The Beer's Law region for the tri-iodide ion Known volumes of a standard 8 uM potassium iodate solution were put into 50 ml. volumetric flasks and 0.25 g. of potassium iodide and 1 ml. of 0.1 M sulfuric acid were added into each flask. The resulting solutions were diluted to volume with distilled water. Absorbances of the solutions were recorded at 353 nm. after fifteen minutes. The results are summarized in table II-1. The iodate concentration ranges from 0.16 uM to 0.80 uM which encompassed the iodate concentration range of 0.2 uM to 0.4 uM and total iodine concentration of 0.4 uM (Tsunogai et al.1971). The absorbance against iodate concentration plot (figure II-1) shows a linear relationship. This shows that the tri-iodide ion concentration generated from the iodate or total iodine in seawater will give a detectable absorbance using the presently available spectrophotometer, and, furthermore, the Beer's Law is obeyed in the iodate and total iodine concentration ranges in seawater. -24- Table II-1. Reaults for testing the Beer's Law region of the tri-iodide ions at 353 nm. in a 10 cm. cell. Volume of 8 uM KIO? used (mlJ Final Volume (ml) Final Iodate Conc. (uM) Absorbance 1 50 0.16 0.101 2 50 0.32 0.225 3 50 0.48 0-342 5 50 0.80 0.608 -25- Figure II-1. The Beer's Law region of the tri-iodide ion absorption at 353 rm. in a 10 cm. cell. 4 1 44 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 t~) IODATE CONCENTRATION (uM) I ii. Determination of the iodate concentration in a standard iodate solution using the standard addition method The procedure is the same as that for the determination of iodate in seawater which will be described later. Five trials were made. The results are tabdlated in table 11-2. The standard iodate solution is 0.2 uM or 25.4 ug-I/l which is close to or lower than the lower limit of the iodate concentration in seawater. A graph of average absorbance against the amount of iodate-iodine added is plotted. The iodate concentration thus determined is 26.2 ug-I/l and the deviation from the theoretical concentration is 3%. This experiment shows that this method is suitable for the determination of iodate at.low concentrations. Moreover, the deviation of 3% (which assumes that there is no uncertainty in the theoretical concentration) is within the goal of ±3% of coefficient of variation. iii. Experimental conditions The reactions involved in the generation of triiodide proceed smoothly at room temperature. No special experimental conditions are required. However, the excess iodide is liable to slow air oxidation. Therefore, absorbances must be recorded within a few hours of tri-iodide -27- Table 11-2. Results for the determination of iodate concentration in a standard iodate solution. Absorbance I II III IV V Average Absorbance 0.000 0.184 0.141 0.127 0.141 0.135 0.1456 1.015 0.274 0.266 0.264 0.275 0.265 0.2688 2.030 0.395 0.405 0.408 0.418 0.415 0.4082 3.045 0.520 0.531 0.539 0.543 0.559 0.5384 I02~-I1 added (ug) From the average absorbance against iodate-iodine added plot, y-intercept (I) = 0.138, and slope (S) = 0.132. Therefore, 103- = I x 25 = 26.2 ug-I/l. Concentration of the standard iodate solution is 0.2 uM or 25.4 ug-I/1. Thus, the deviation from the theoretical concentration is 26.2-25.4 x 100% = 3%. 25.4 -28- formation. iv, Theoretical interferences Possible chemical species that may interfere with the analytical procedure have also been examinea. The major reactions involved in the analytical scheme are :1) 103~ + 51~ + 6H+ = 312 + 3H2 0 2) 3I2 + 31 = 313 (in the presence of excess iodide) -3+ S4062- and 3) 13" + 2S 2 03 2- (for the titrimetric method only) Reducing agents that are capable of reducing 12 or 13 to'I~ will decrease the iodate concentration found and oxidizing agents that can oxidize the excess iodide to iodine will increase it. Other species that can react with thiosulfate will also increase the iodate concentration in the titrimetric method. A survey of the more probable interfering species and the reactions that they may be involved in are listed in table 11-3. The data were obtained from Jacobson (1949). Of course, there are still other chemical species that can react with iodide or iodine, but they are not present in the ocean, for example, bromine and hydrazine. There are several -29- Table 11-3. Some possible interfering species. Conc. of interfering species in seawater (ug-at/l) Interfering species Reaction* Reaction* conditions Cu 2+ 2Cu 2 + + 4I" = Cu2 I2 + 12 none <0.05 N02 2N0 2 - + 21 presence 0.01*** + 4H+ = 2NO + 12 + 2H 0 2 ** of mineral acid MnO4"' 2MnO4 + I + H2 0 boiled negligible** very acid- negligible** ic condi- present as tion solid Fe(OH)3 saturated negligible** 2Mn0 2 + IOf-+ 20H~ Fe 3 + H2 S 2Fe3+ + 21" = 2Fe 2+ + 12 H 2S + I2 = S + 2H+ + 2I~ with H 2S So3 2 - So3 2- + 12 + H2 0 + 2H+ SO42- + 21 S20 3 2S 2 032- + 31-= acidic negli- solution gible** none negligible** S 4 0 6 2 ~ + 312S 2 0 3 2- + 12 = none S406 2- + 21" H3 AsO 4 H 3 AsO4 + 21 + 2H+ = strong- 0.04**** Table 11-3. (continued) Interfering species Reaction* Reaction* conditions H3 As04 HAs0 2 + 12 + 2H2 0 mineral acid * Jacobson 1949 ** Goldberg 1963 *** **** Wada et al.1972 Johnson et al. 1972 Conc. of interfering species in seawater (ug-at/1) reasons that make the effect from the possible interfering species negligible. Firstly, their concentrations in the open oceans are low when compared to that of iodate. The average concentrations of these interfering species in the oceans are also listed in table 11-3 if they are available. Secondly, the reactions involved in the analysis of iodate are rapid and do not require extreme reaction conditions (for example, heating) as some of these interfering reactions do. Thus, all interfering reactions that are slow or require specific reaction conditions will have negligible effect. Thirdly, for each iodate ion, three tri-iodide ions are formed. This means that the contribution of iodate to the absorbance is being amplified three-fold. The interfering species do not have this amplifying effect. Thus, their interfering effect is reduced by a factor of three. v. The tri-iodide ion absorption curve The absorption curves of the tri-iodide ions developed from a sample of seawater and from a pure iodate solution of comparable concentration (0.2 uM) (figure 11-2) are identical. Both exhibit maxima at the same wavelengths. A prominent maximum is observed at 3500 A (350 nm.). The ratio of the intensities of corresponding maxima is also the same. No -32- Figure 11-2. The tri-iodide ion absorption curve. A. Developed from a 0.2 uM standard potassium iodate solution. Solid line. B. Developed from seawater. Broken line. Chart speed l 5 in./min. Scanning speed : 25 I/sec. .Path length 1 10 cm. X is the point at which the light source was switched from visible to UV. 2500A INTENSITY SCALE FOR CURVE A 0.0 .05 1 .1 1.0 .2 1.5 2.0 WOW 00I 0.0.00-l0 N 3500A 500A I - .4 0.0 INTENSITY SCALE FOR CURVE B 1 I .5 I 1.0 1.5 shoulder or extra baseline noise can be observed in the seawater curve. This shows that there is no interfering species in seawater which absorb at around the wavelength of 350 nm. C. The analytical method i. Instrumentation Absorbances are recorded by a Beckman DU quartz spectrophotometer light source fitted with a Gilford Model 2220 adapter and a Gilford Model 222 photometer and regulated power supply. For the standard addition method, absorbances are measured with a matched pair of 10 cm. cells. For the photometric micro-titration, the housing for the cells was modified as shown in figure II-3a. The titration cell is a modified version of the standard 10 cm. cell (as shown in figure II-3b). A Gilmont No. 7846 Buret with a maximum capacity of 2.0 ml. and divisions of 0.002 ml. is used for the titration. The Luer joint is fitted with teflon adapters from Rainin Instrument. A teflon tube is used to connect the buret to the cell (see figure II-3c). ii. Standard solutions The following standard solutions were prepared prior Figure 11-3. The apparatus. Fig. II-3a. The titration cell. Fig. II-3b. The modified housing for the titration cell. Fig. II-3c. The titration buret. -35Fig. II-3b Fig. II-3a COVER I / ,- I TO DETECT? 1CYR. .1 TEFLON ADAPTOR MICRO-BTJPET TEFLON DELIVERY TUBING Fig, II-3c IT IG -- CTPI 'j, to the analysis :- 1. 5% potassium iodide solution Five grams of potassium iodide crystals were weighed out and traferred into a 100 ml. volumetric flask. The crystals were dissolved in a small volume of distilled water and then diluted to volume. 2. Standard potassium iodate solutions 2.14 g. (0.01 mole) of potassium iodate crystals were weighed out and transferred into a 500 ml. volumetric flask. The crystals were dissolved in a small volume of distilled water and then diluted to volume to form a 20,000 uM stock solution. Iodate solutions with concentrations of 2,000 uM, 200 uM, 20 uM, 8 uM, 4 uM, 0.4 uM and 0.2 uM were prepared from this stock solution by successive dilutions. 3. 0.1 M sulfuric acid 25 mi. of concentrated sulfuric acid (36N) was added slowly and in small increments to 400 ml. of distilled water with intermittent swirling and cooling in an ice-water slush bath. After all the sulfuric acid had been added and the solution cooled, another twenty five milliliters of distilled water was added to the solution in the same manner. The resulting solution is approximately 1 M. 50 mi. of the 1 M sulfuric acid was pipetted into a 500 mi. volumetric flask -36- and diluted to volume to make a 0.1 M sulfuric acid solution. 4. Approximately 100 uM sodium thiosulfate solution (Kolthoff et al 1969) About 3 1. of distilled water was boiled for 30 minutes and then cooled back to room temperature. 2.48 g. (0.01 mole) of sodium thiosulfate pentahydrate .was.weighed out and transferred into a 100U ml. volumetric flask. About 0.1 g. of sodium carbonate was weighed out and added to it. The solids were dissolved in a small volume of the freshly boiled water and then diluted to volume. The resulting solution was about 10,000 uM. Thiosulfate solutions with concentrations of 1000 uM and 100 uM were prepared from this stock solution by successive dilutions. About 0.05 g. of sodium carbonate was dissolved in every 500 ml. of solution. The freshly boiled water was used for the preparation of all these thiosulfate solutions. The thiosulfate solution was allowed to stand overnight before standardization. The standardization procedure is the same as the photometric micro-titration method for the determination of iodate in seawater except that a standard 0.4 um iodate solution is used instead of seawater. The thiosulfate solution should be standardized just before the the titrations are performed. -37- iii. Procedure for the analysis of iodate in seawater Allow the stored samples to warm up to room temperature just before the analysis. Shake the sample bottle vigorously in order to ensure thorough mixing. Filter the sample through a Whatman 40 ashless filter paper with suction. After that, the sample can be analyzed by one of the routes shown in figure 11-4. 1. The standard addition method Pipette 40 ml. of seawater into each of a set of four 50 ml. volumetric flasks. Mark the flasks 0, 1, 2 and 3 and pipette 0, 1, 2, and 3 ml. of an 8 uM potassium iodate solution (1.015 ug 103~-I/ml) into each flask accordingly. Add 0.25 g. of potassium iodide, as solid crystals or as a 5 ml. aliquot of a 5% (w/v) potassium iodide solution, and 1 ml. of 0.1 M sulfuric acid to each flask and then dilute the solutions to volume. Shake the flasks and then record the absorbance of each solution in a 10 cm. cell at 353 nm. after fifteen minutes with seawater as the reference. Repeat this procedure two more times for each sample. Plot the average absorbance against the amount of iodate-iodine added. The slope (S) and the intercept (I) of the graph can be determined either graphically or by a linear regression calculation. The iodate concentration in ug-I/1 is given by: -38- Figure 11-4. The analytical scheme for the analysis of iodate and total iodine. -39- Total Iodine Iodate IO3~ I~ + Oxidizing agent > Photometric microtitration Addition method 103 03~ = x 25. one can use the relationship of 1 uM-I = 126.9 ug-I/l to convert the result to uM-I. 2. The photometric micro-titration method Pipette 30 ml. of the filtered seawater into the cell and add to it 0.25 g. of potassium iodide crystals, 1 ml. of 0.1 M sulfuric acid and distilled water until the final volume is 38±1 ml. Put the titration cell into the spectrophotometer and insert the delibery tube of the buret into the cell. Using seawater as the reference, read the absorbance at 353 nm. after about ten to fifteen minutes of stirring and at one minute intervals thereafter until a constant absorbance is reached. Balance the spectrophotometer before each reading using the seawater reference. Then, add the standardized thiosulfate solution from the buret at known volume intervals of 0.1 or 0.2 ml. After each addition of the thiosulfate solution, stir the solution for one minute and then read the absorbance at half to one minute intervals until a constant absorbance is attained. Again, the spectrophotometer is balanced before each reading. The end-point is reached when no further decrease in absorbance is observed upon further additions of thiosulfate solution. Take at least five points before and after the end-point. Plot (Vi+AV)A against AV -40- (where Vi is the initial volume, AV is the volume of thiosulfate added and A is the absorbance). The end-point is the point where the two lines intersect. A plot of (Vi+AV)A against AV is used instead of A against AV in order to take into account the dilution effect caused by the titrant and make the resulting graph linear. This relationship is developed as follows. Definition of symbols: A - absorbance at any point of the titration. AO - initial absorbance before any thiosulfate solution is added. a - molar extinction coefficient. b - path length. C - concentration of tri-iodide ions at any point of the titration. Ci - initial concentration of tri-iodide ions. CT - concentration of the thiosulfate solution. Vi- AV initial volume. - total volume of thiosulfate solution added up to that point. VE.P. - volume of thiosulfate solution required to reach the end-point. A0 , a, b, Cj, CT and V- are constants. Vi and CT are known quantities. The reaction that is involved in the titration is: 1 3 + 2S2 03 2- = 31 + S406 2 Therefore, prior to the addition of any thiosulfate solution, Ao = abCi, and the amount of tri-iodide ions =ViCi. After V ml. of the thiosulfate solution is added, amount of thiosulfate added = AVCT, tri-iodide ions consumed = tAVCTl tri-iodide ions remained = (ViCi - WAVCT), and therefore, the concentration of tri-iodide ions = (ViCi - tAVCT)/(Vi + LV). The resulting absorbance is ab(ViCi - khVCT) A= (Vi + AV) ,or (Vi + AV)A = (-abCT)AV + (abViCi). Since a, b, Ci, CT and Vi are all constants, therefore, a plot of (Vi + AV)A against AV will be a straight line. At the end-point, ViCi = bVE.P.CT. Therefore, A = 0 or some constant value if there is a residual absorbance due to improper alignment of the cells or other mechanical imperfection. One can therefore locate the end-point at the intersection of the two straight lines. The corresponding AV value will be the volume of thiosulfate solution needed to reach the -42- end-point. Ci can be calculated from VE.P. because VE.P.CT 2Vi 4 Since the tri-iodide ions are formed by the reaction IO~ + 81~ + 6H+ = 313- + 3H20 -, therefore, Ci [10 3 3 = VE.P.CT 6Vi * Figure 11-5 is a sample plot of (Vi+AV)A against AV for a titration. A against AV plots deviate from linearity near the end-point while the (Vi+AV)A against AV plot does not exhibit such a deviation. The initial volume, Vi, does not have to be known to better than ±1 or 2 ml. The cell is calibrated so that Vi will not vary for more than 1 ml. The effect on the end-point is not detectable for a difference of 2 ml. in Vi. iv. The precision of the analytical methods One sample set of results is shown here for each of the two methods - the addition method and the titration method. The seawater was collected by the Woods Hole coast. 1. The addition method The analysis was repeated five times and the average absorbances of the five trials were used for the absorbance -43- Figure 11-5. A sample plot for the photometric microtitration. I 0' 0 T p V (,V + TA) I 2' 0o -.44- Table 11-5. Data for determining the precision of the photometric micro-titration method. Vi = 39 + 1 ml CT= 67.6 um (ml) A (Vi+AV)A 0.00 0.184 7.176 0.10 0.20 0.124 4.861 0.30 (Vi+Av)A III A (Vi+AV)A 0.183 6.954 0.186 7.068 0.152 5.791 0.159 6.058 0.123 4.699 0.126 4.813 0.092 3.524 0.096 3.677 .068 2.611 0.040 1.540 0.40 0.064 2.522 m.65 2.496 0.45 0.055 2.170 0.052 1.999 0.038 1.463 0.50 0.55 0.029 1.147 0.029 1.119 0.60 0.029 1.147 0.029 1.119 0.029 1.119 0.80 0.029 1.154 0.030 1.164 0.032 1.242 1.00 0.029 1.160 0.029 1.131 0.032 1.248 1.30 0.028 1.128 0.029 1.140 0.032 1.258 1.50 0.028 1.134 0.029 1.148 0.032 1.267 0.031 1.237 0.032 1.277 1.60 --- 1.90 VEP I03f 0.530 ml 0.534 ml 0.528 ml 0.199 uM 25.3 ug/1 0.201 um 0.198 uM 25.5 25.2 -45- ug/1 ug/1 Table 11-5. (continued) IO3' VEP x 67.6 Vi x6 The average iodate concentration is 25.3 ug-I/1 ± 0.7% -46- against ug iodate-iodine added plot. The results are summarized in table 11-4 and the plot is shown in figure 11-6. Using a linear regression fit, the iodate concentration was found to be 27.2 ug-I/1 and the standard deviation is 2.7%. Thus, the precision of this method is acceptable for the study of the distribution of iodate in seawater. Using this method, about six samples can be analyzed in a day if, for each sample, three replica are analyzed. This will be about twice as fast as the co-precipitation method. The procedure involved is simple enough so that the automation of this method with minor modifications in the procedure seems very promising. 2. The titration method The analysis was repeated three times for a single sample of seawater. The results are summarized in table 11-5. The iodate concentration was found to be 25.3 ug-I/1 with a coefficient of variation of 0.7f. The lines of the titration curved were fitted by eye. From the sample plot (figure 11-5), it can be seen that the data points fall exactly into two straight lines of different slopes. These two lines intersect at a sharp angle and consequently, the uncertainty in the end-point will be small. It is hoped that a computer program will be developed eventually to handle the line -47- Table 11-4. Results for the determination of the precision of the addition method. 103-I added (ug) Absorbance I II III IV V Average Absorbance 0.000 0.135 0.127 0.117 0.123 0.127 0.1258 1.015 0.245 0.243 0.244 0.244 0.294 0.254 2.030 0.371 0.360 0.361 0.365 0.368 0.365 3.045 0.480 0.474 0.482 0.517 0.488 0.4882 The data was fitted to a straight line by a linear regression program. From the program, slope (S) = 0.118 and the y-intercept = 0.1285. Therefore, x 25 = 27.2 ug-I/l. I03- = The standard deviation is 2.7f. -48- Figure 11-6. A sample plot for the determination of iodate in seawater by the addition method. 0.0 3.0 2.0 1.0 IODATE -IODINE ADDED (ug) 4.0 fitting task. This will eliminate prejudice from the investigator and also allow the calculation of confidence limits for the end-points. Using this method, eight to ten samples a day can be analyzed. The procedure is-simple and the amount of chemical manipulations is minimal making it possible for on board analysis and automation. Also, the volume of sample required is small (only about one fifteenth of the volume needed for the co-precipitation method) making it possible for routine analysis. v. Correlation between the addition method and the titration method A sample seawater was analyzed three times by the titration method and twice by the addition method. The average iodate concentration is 25.8 ug 103~-1/1 ±2%. (The results are summarized in table 11-6.) The difference of 2% from the mean is within the experimental uncertainties of these two methods and, thus, it may be concluded that both methods give comparable results. -50- Table 11-6. Correlation between the addition method and the titration method. The results were obtained from the same sample of seawater. Titration method Number of replica Addition method 3 2 25.3 26.3 Average iodate conc. - (ug If--I/l) Average iodate concentration from the two methods is 25.8 ug 103'-/1 ± 2% vi. Correlation between the addition and the Matthews and Riley's method. Samples of seawater were analyzed by both the addition method and the Matthews' method. Iodate was analyzed by both methods for one sample. For the other samples, iodide was analyzed by the Matthews' method and iodate was analyzed by the addition method. The results are summarized in table 11-7. In the one case where the iodate concentration has been determined by both methods, the results differ by 5% from the mean (27.5 ug-I/1 +1.5 ug-I/l). This is within the uncertainties of the methods. The iodate concentration determined by the addition method in the surface water at Woods Hole is found to be lowwhen compared to open ocean surface water, at 0.2 uM. (In open ocean water, it ranges from 0.3 to 0.4 uM.) The corresponding iodide concentrations found by the co-precipitation method are all high and close to 0.2 uM. The total iodine concentration obtained by adding the corresponding iodide and iodate concentrations from these two methods are all around 0.4 uM. Since total iodine is rather uniform in the oceans and in the Pacific, the total iodine has been reported to be 0.4 uM (Tsunogai et al 1971), it shows that the iodate and iodide values are probably -52- Table 11-7. Correlation between the addition method and the Matthews and Riley's method. Addition Method Iodate (ug-I/1) Matthews' Method Iodate Iodide (ug-I/1) (ug-I/1) 26.0 29.0 Total Iodine* (ug-I/1) 25.4 26.6 52.0 30.0 25.4 55.4 29.2 22.6 51.8 22.8 50.6 23.2 49.6 27.8 --- 26.4 * Total iodine is the sum of the iodide and iodate concen- trations from the two different methods. -53- acceptable, otherwise, the total iodine concentration will not be close to that in the average ocean water. The amount of data from the co-precipitation method is small because of its technical difficulties and the time required for each analysis. The method involves the use of bromine and the complete removal of bromine was found -to be difficult. Other workers have shared this same experience (Liss, private communication). Any trace of residual bromine can catalyse the oxidation of iodide to iodine. Since excess iodide is used to generate the tri-iodide complex, this excess iodide may be oxidized and give absurdly high iodate or iodide concentrations. This flaw was detected in a rather late stage of this research and the procedure was subsequently modified to give the results summarized here. In the original Matthews' method, the first batch of excess bromine was removed by boiling and the second batch by bubbling nitrogen through and adding phenol to the solution. In the modified . procedure, in both instances, bromine was removed by boiling for at least one hour. One question that may arise is whether the addition of reagents (for example, the addition of acid in the production of the tri-iodide ions) will alter the iodine composition in the sample by side reactions. The smoothness of -54- the graphs for both methods and the agreement among the addition, titration and Matthews' methods suggest that the interference is probably negligibly small. This comparative study of the addition method and the co-precipitation method shows that the accuracy of the addition method is acceptable. Since the titration method yields results comparable to those of the addition method, its accuracy should also be acceptable. Summing up all these studies, one may conclude that the new methods can give highly reproducible results that are comparable to the values obtained from the co-precipitation method. D. Attempts for developing an analytical method for the determination of iodide in seawater In the original scheme (figure II-4), it was anticipated that iodide can be oxidized to iodate, and then the total iodine concentration determined by the methods used for the determination of iodate. The difference between the total iodine and iodate concentrations will be the concentration of iodide in the sample. Several oxidizing agents have been attempted but none of them seems satisfactory. 1. Sodium perchlorate Excess sodium perchlorate was added to each 40 ml. -55- uM of a 0.2 uM potassium iodate and 0.2,potassium iodide solution in a 50 ml. volumetric flask. The solution was heated in a hot-water bath. Total iodine was determined using the addition method after the heating. The iodide concentration obtained was low. At first, incomplete oxidation was suspected. The oxidation step was studied intensively. Kinetic experiments on reaction time and reaction temperature were performed. An optimal condition of heating at 7500 for one and a half hours was obtained. However, even under optimal conditions, the iodide concentration found was still low, and only 20%O con- version was apparently achieved. This is probably because perchlorate can only oxidize iodide to iodine. So, instead of I- > 3 -- 3 > 312 >313-, one has 2I~ > 12 13* Consequently, the amplification effect of the direct method is lost. Iodide concentrations in seawater are much lower than iodate concentrations, and, at times, may even drop to zero. The loss of sensitivity by a factor of six is significant and as a result, this route was abandoned. 2. Bromine water Bromine water can oxidize iodide to iodate. However, the complete removal of bromine is difficult. As stated earlier, any trace of residual bromine can catalyse the oxidation of the excess iodide and give absurdly high results. -56- The toxicity of bromine also makes the use of bromine as the oxidizing agent undesirable. 3. Alkaline permanganate solution Iodide can be oxidized to iodate by boiling it with an alkaline permanganate solution. Again, the complete removal of the excess permanganate without side effects on later steps is difficult. Moreover, manganese dioxide is formed in the process. The removal of manganese dioxide by filtration will cause some loss in sample solution and also greatly increase the amount of manual manipulation. -57- CHAPTER III SAMPLE STORAGE Effect of storage on the iodate content of seawater A. A qualitative study About 40 1. of seawater was collected and about one half of it was filtered. The iodate content in it was determined by the addition method. Ten one-liter glass erlenmeyer flasks and ten polyethylene bottles were used for sample storage. About 2 1. of the unfiltered seawater was put into each of five glass flasks and five polyethylene bottles and about 2 1. of the filtered seawater was put into each of the remaining bottles. Each sample in each set of five bottles was stored under different conditions and all of them were analyzed for iodate again after eight days. The qualitative results are summarized in table III-1. In all cases when chemicals are added, the absorbances are all much higher than would be observed for a non-spiked sample. This may be cause by a change in the iodate concentration or by the absorption of the added chemical at 353 nm. as in the case of mercuric chloride. Both sets of bottles give similar observations showing that either glass or polyethylene bottles can be used. The filtered sample and the -58- Table III-1. The qualitative effect of storage on the iodate' content in seawater by various storage methods. Storage Conditions Temperature Chemical Added 50C None No observable change. Room temperature None No observable change. Room temperature Hl High absorbance. Room temperature Toluene High absorbance. Room temperature HgCl 2 Very high absorbance. Effect of Storage -59- unfiltered samples also give comparable results. From this qualitative study, it seems that a possible way for storage is by temperature control. B. A quantitative study The samples were all stored in polyethylene bottles and the seawater was not filtered before storage. Three storing temperatures were used: room temperature, 500 (refrigerator temperature), and frozen (freezer temperature). The results are tabulated in table 111-2. This study shows that seawater can be stored for up to four to five weeks at low temperature with negligible effect on the iodate concentration. Storing at room temperature will cause the iodate concentration to increase slowly. This is probably due to the oxidation of iodide to iodate as predicted by the thermodynamic equilibrium calculation upon the death of the bacteria. Frozen samples give comparable results after being re-frozen and being stored for another week or two. The frozen samples were all from the Southern Atlantic. They all have concentrations comparable to those reported for the Pacific (Tsunogai 1971b; Tsunogai et al.1971). This shows that the iodate concentrations probably have not been changed during storage. -6o- Table 111-2. A quantitative study on the effect of storage on the iodate content in seawater by controlling the storage temperature. Storing Temperature Room temperature 50C 50 C Frozen sample Frozen sample Frozen sample Time of Storage (day) Iodate Concentration (ug-I/l) 0 27.8 9 26.8 35 41.1 0 27.8 9 28.4 36 29.4 0 26.0 7 26.4 18 25.0 16 59.2 28 56.9 16 59.6 28 61.2 16 62.7 26 61.6 Table 111-2. (continued) Storing Temperature Frozen sample Time of Storage (day) Iodate Concentration (ug-I/l) 11 60.5 26 60.3 -62- CHAPTER IV THE DISTRIBUTION OF IODATE IN THE SEA A. Seasonal variations in the iodate content of the Woods Hole coastal surface water Surface seawater samples were collected between 0900 and 1200 off Woods Hole at intervals of a few days from July to November. Water was collected with a 20 1. polyethylene bottle and was brought back to the laboratory for immediate analysis of iodate. The results are shown in table IV-l. A plot of iodate concentration against date of sample collection is shown in figure IV-l. This study on the seasonal variations of iodate concentrations in the surface water is far from conclusive. First, the spring algal bloom was missed. The most pronounced change should be observed in this period. Secondly, the location for sample collection was so close to shore that tidal effect may carry seawater from greater depths with higher iodate concentration to the surface and cause changes in iodate concentration. The tidal effect may be eliminated if the sampling site is a little further away from the shore. Alternatively, a large tank filled with surface water may be used to approximate the surface ocean and more control on the exp.erimental conditions will be obtained. Under normal cir- -63- Table IV-l. Seasona1~variations in the iodate content of the Woods Hole coastal surface water. Date of Collection Iodate Concentration (ug-I/1) July 13, 1972. 27.2 Aug. 9, 1972. 27.8 Sept. 18, 1972. 30.2 Sept. 25, 1972. 26.0 Oct. 2, 1972. 24.7 Oct. 3, 1972.. 25.4 Oct. 9, 1972. 27.8 Oct. 10, 1972. 26.4 Nov. 7, 1972. 26.3 Nov. 21, 1972. 25.7 -64- Figure IV-1. Seasonal variations in the iodate content of the Woods Hole coastal surface water. 0 - O Q00 ci C4 20 0~ ~~ 30 IODATE CONCENTRATION (ug-I/1) 40 cumstances, if iodate is being reduced by bacteria to iodide, iodate concentrations should be inversely proportional to the amount of biological activity. A maximum change of 5 ug-I/1 (or 15-20%) of iodate has been observed in the period of July 13 to Nov. 21, 1972. There was a su'dden dip in iodate concentration from Sept. 18 to Oct. 3. The red tide, which is an indicator for high biological activity, was observed in the coastal water off Northern Massachusetts in Sept. 14 (Boston Globe 1972). This dip may be related to this increase in biological activity. However, I must emphasize that this conclusion is a highly speculative one. More data will be needed before a firm seasonal trend of iodate concentration variations can be established. B. Diurnal variations in the iodate content in seawater Surface seawater samples were collected in the same manner and at the same place as mentioned in the previous section on Oct. 9 and Oct. 10, 1972 at various time of the day. Iodate was analyzed by the addition method immediately after collection. Phosphate was analyzed by the method of Murphy et al.(19 6 2). The results are shown in table IV-2. A graph of phosphate and iodate concentrations against time of the day is shown in figure IV-2. -66- Table IV-2. Diurnal variations in the iodate content and the corresponding changes in the phosphate concentration of the Woods Hole coastal surface water. Date of Collection Oct. 9, 1972 Oct. 10, Time of Collection Iodate Concentration (ug-I/l) Phosphate Concentration (ug-P/l) 0925 27.8 19.6 1257 30.0 18.8 1625 29.2 17.9 0730 26.4 19.1 1972. -67- Figure IV-2. Diurnal variations in the iodate content and the corresponding changes in the phosphate concentration of the Woods Hole coastal water. A. The iodate concentration vs. time of day curve. B. The phosphate concentration vs. time of day curve. -99- 0 H> '-3 H nOH H- 0- 20 15 IODATE CONC. \ 25 I I / / / I / (ug-P/1) (ug-I/1) 'A PHOSPHATE CONC. / / I I I. The study on the diurnal variations is also inconclusive although some trends have been observed. The problems of the sampling site still exist. Moreover, the samples were obtained in October when the biological activity was probably low. Consequently, the change in iodate concentration was small. The experiment was performed on a very cold, windy day. Wind stress might have brought deeper water with higher iodate concentration to the surface. The tidal effect might also have changed the iodate concentration. Bad weather also prevented sampling at night. These disturbing factors caused by the tide and the wind again may be avoided by doing better controlled experiments with a tank. The phosphate data are just what one would expect f6r a nutrient. Phosphate concentration is inversely proportional to the amount of biological activity and a decrease in the concentration was observed in the daytime while high concentration was observed in both mornings. The iodate concentrations also changed but the variations observed in this study cannot be easily explained by biological activity alone. -69- C. The distribution of iodate in the Argentine Basin in the South Atlantic Samples were collected from the Geosecs cruise, leg. 5 (Nov. 4 to Nov. 28, 1972) at station 60 and 61 on Nov. 2223 and Nov. 24-25 respectively. Locations of the stations are shown in figure IV-3. Samples were frozen in polyethylene bottles. They arrived at the laboratory at Woods Hole on December 5 and were analyzed for iodate immediately by the titration method. The standard thiosulfate solution was standardized just prior to the analysis. The iodate results are summarized in table IV-3 and table IV-4. Other hydrographic data are listed in appendix 1 and appendix 2. They were prepared by Broe5cer et al.(1972b). The physical oceanography of the sampling locations for stations 60 and 61 has been thoroughly studied (Fuglister 1960; Ewing et al.1971; LePichon et al.1971a, 1971b). Different water masses can be identified from the salinity, potential temperature, oxygen and nutrient profiles (figures IV-4, IV-5, Iv-6, IV-7 and IV-8) of these stations using the well-documented characteristics of the different water masses in the world oceans (Defant 1961; Sverdrup et al.1942; von Arx 1962). By comparing these published data with those obtained from station 60 and 61, one can distinguish five -70- Figure IV-3. Cruise track of R/V Knorr, Geosecs Cruise, Leg 5, and the locations of stations 60 and 61. I I __________I 4. -BR- - ; ?-B-A7Z-- L 20- I'' i~Y ~7! ', I 30 p 40* TRACK -OF R*/V KNORR GEOSECS LEG 5 A LARGE VOLUME STATIONS * REGULAR STATIONS o STD STATIONS * SPECIAL STATIONS (SEE STATION LISTING) Table IV-3. Nutrient data from Station 60. Depth m I3uM Ip uM P04- NO 3 ~ uM uM uM -2.4 0.09 0.0 -4.2 0.10 0.0 Iox Iox AOU uM 103% 2 0.387 0.388 -0.001 0 65 0.392 0.394 -0.002 -1 126 0.448 0.441 0.007 2 18.1 0.25 2.1 244 0.448 0.434 0.014 3 33.8 0,,46 5.3 353 0.448 0.430 0.018 4 44.7 0.65 8.4 533 0.458 0.428 0.030 7 72.8 1.23 17.8 729 0.482 0.454 0.028 6 69.2 1.70 25.1 1036 0.482 0.442 0.040 8 97.6 2.02 30.2 1261 0.474 0.422 0.052 11 125.9 2.16 32.2 1483 0.474 0.415 0.059 12 144.2 2.17 32.4 1576 0.478 0.418 0.060 13 145.3 2.12 31.9 1723 0.458 0.400 0.058 13 140;.4 2.02 30.4 2270 0.459 0.421 0.038 8 91.6 1.46 22.6 2867 0.458 0.425 0.033 7 80.2 1.36 21.0 3212 0,465 0.431 0.034 7 84.0 1.40 21.7 3419 0.474 0.431 0.043 9 104.7 1.66 24.9 3725 0,471 0.419 0.052 11 127.4 2.00 29.6 4016 0.473 0.418 0.055 12 133.7 2.19 32.5 4197 0.475 0.421 0.054 11 132.9 2.22 32.4 -72- Table IV-3. (continued) Depth 03 m um uM uM 4223 0.474 0.420 0.054 4238 o.478 4267 0.476 --- a 4297 0.474 --- 4323 0.489 4346 0.474 4367 0.475 4388 0.495 4405 0.481 Ip 'p - Iox Iox I0' 11 AOU NO 3 ~ uM uM uM 133.0 2.21 32.6 2.23 Preformed iodate. Iox - Iodate of oxidative origin. -73- 2.23 32.4 2.23 32.4 Table IV-4. Nutrient data for Station 61. Depth I03' Ip Iox IO3- Iox uM uM uM AOU 3 P0 4 ~ NO3 " uM uM 5 0.403 o.403 0.000 0 -o.4 0012 0.0 148 o.425 0.413 0.012 3 28.1 0.41 4.6 268 0.420 0.412 0.018 2 18.9 0041 4.4 426 0.451 0.432 0.019 4 45.7 0.81 10.7 535 0.422 0*410 0.012 3 28.3 0.78 10.2 596 0.451 0.432 0.019 4 46.4 1.13 15.7 692 0.466 0.441 0.025 5 60.9 1.61 22.9 792 0.471 0.447 0.024 5 59.2 1.68 24.7 996 0.485 0.454 0.031 6 75.6 1.87 27.9 1307 o.490 o.442 0.048 10 117.6 2.14 31.8 1386 0.498 o.446 0.052 10 127.1 2.14 32.6 1868 0.482 0.423 0.059 12 145.0 2.06 31.0 2346 0.471 0.426 0.045 10 109.1 1.64 25.0 2825 0.470 0.432 0.038 8 93.9 1.48 22.7 3165 0.485 0.437 0.038 10 118.2 1.74 26.2 3362 0.471 0.419 0.052 11 126.3 1.88 28.1 3496 0.478 3728 0.488 0.433 0.055 11 135.4 2.08 30.7 4096 0.475 0.419 0.056 12 135.8 2.18 32.2 -74- Table IV-4. (continued) Depth 03 m um 4647 0.493 4704 0.486 4752 0.487 4804 0.486 4827 0.482 4844 0.475 4864 0.478 4883 0.477 4904 0.475 4924 0.508 4926 0.485 'p IOx Iox AOU P04 3~ N0 3 ~ uM uM 2 21 32.6 2.21 32,6 03' uM 0.439 um 0.054 u%4 11 132.8 ---- 0,454 0.054 Ip - Preformed iodate. Iox - 132.4 Iodate of oxidative origin. -75- Figure IV-4. The nitrate, phosphate and silicate profiles of station 60. . - Silicate. x - Phosphate. + - Nitrate. -760 0 50 100 10 20 3 NITRATE x 44,t X + 4 x + x + xx C), - O 0X 0+ + X . - -X4 x 44 + x 44 - C- uM uM PHOSPHATE uM 2 1 0 SILICATE + + Figure IV-5. The oxygen, salinity and potential temperature profiles of station 60 with the water masses identified. SACW - South Atlantic Central Water. AAIW - Antarctic Intermediate Water. NADW - North Atlantic Deep Water. AABW - Antarctic Bottom Water. . - Salinity. x - Oxygen. + - Potential temperature. -770 10 5 t 1 1 POTENTIAL TEMPERATURE 35.5 SACW 1*1+ + AAI +1 x C> C) * 4 C) CoI CD C4. C) + 0 C+ t 4I AAB~l 0C 260 0.2 UiM 240 35.0 34.5 34.2 20 220 200 180 15 36.0 S% Figure IV-6. The nitrate and phosphate profiles of station 61. x - Phosphate. + - Nitrate. 0 30 20 10 + -783 PO 4~ uM 2 0 NO 3 t + 4. x+ K x. * x~t 2 C4 + + *1 N C- C4 x E-i C1 c+ + K C 1-+ X + x + ++ O + uM Figure IV-7 and Figure IV-8. The oxygen, salinity and potential temperature profiles of station 61 with the water masses identified. SACW - South Atlantic Central Water. AAIW - Antarctic Interediate Water. NADW - North Atlantic Deep Water. AABW - Antarctic Bottom Water. - Salinity. x - Oxygen. + - Potential temperature. 15 POT.' TEMP. oc I I1 I I 200 180 240 220 260 I 02 UM L 34.2 36.0 35.5 35.0 34.5 +' S.A. AA4. 0> -x NADW x I, / K Ic AA BWII different water masses, namely, the surface water from 0 150 M., the South Atlantic Central Water from 150 - 600 m., the Antarctic Intermediate Water from 600 - 2000 m., the North Atlantic Deep Water from 2000 - 4200 m., and the Antarctic Bottom Water from 4200 m. to the bottom. The iodate profiles (figures IV-9 and IV-10) from both stations have some common features. There is a significant depletion in the surface layer from 0 - 120 m., a minor maximum between 200 - 500 m., a broad maximum from 1000 - 1800 m., and then a minimum from 2000 - 2800 m. From then on, the iodate concentration fluctuates. However, there is a trend of gradual increase until about a few hundred meters from the bottom. The iodate concentrations close tt the bottom fluctuate wildly. By matching the inflexion points observed in the iodate profiles and those in the T-S diagram, it can be seen that they do occur at roughly the same depths. It is possible that iodate may be characteristic of the different water masses. However, it would be unwise to generalize such a proposal to the world oceans by two profiles only. Much more data will be needed from various oceans before such a proposal can be firmly established. The nutrient-like behavior of iodate can be observed when the iodate profiles are compared to those of the other -80- Figure IV-9. The iodate profile of station 60. -81- /ODATE 40 .35 2- - GEOSECS STATION 3- 4- 5- 60 (p M) .45 .50 Figure IV-10. The iodate profile of station 61. -82- /ODATE 0.35 0 0.4 (pM) 0.45 0.5 l 2- GEOSECS STATION 61 3- 4- 1 7 0 nutrients. In station 60, they all have a surface depletion, a maximum around 1000m., followed by a mid-depth minimum and then a gradual increase towards the bottom. Even the minor changes in the other nutrients are reflected in the iodate profile. The slight inflexion at about 160 m. in the phosphate, nitrate and silicate profiles is recorded as a jump in the iodate profile. In station 61, a similar trend is observed. The two minor maxima at the surface around 150 m. and 450 m. in the other nutrients are much amplified in the iodate profile. These observations show that iodate does approximate the behavior of the other nutrients. In order to study the nutrient-like behavior of iodate in greater detail, calculations were made to see if some kind of relationship similar to the Redfield ratios exists between iodate and the other nutrients. Redfield (1963) proposed that organisms assimilate nutrients at the surface water, and, upon their death, they sink and the nutrients are regenerated via oxidation by bacterial activity. Consequently, the amount of oxygen used for this oxidation process may have a stoichiometric relationship with the other nutrients. Apparent oxygen utilization (AOU) has been defined as: AOU = 02' - 02 (Richards 1965) where 02' is the solubility of oxygen at the observed temperature and salinity -83- under an atmosphere saturated with water vapor and a total pressure of 760 Torr. 02 is the observed oxygen content in that sample of seawater. 02' can be derived from the data published by Weiss (1970) and Green et al.(1967). The AOU values calculated are shown in table IV-3 and table IV-4. A number of investigators (Park 1967; Redfield et al.1963) have shown that the nutrient parameters - phosphate, nitrate, silicate and AOU - exhibit linear relationships. From these graphs, the following equation is derived to describe the process for the regeneration of.nutrients: (CH2 0)l06(NH 3 ) 1 6 H3 PO4 + 13802 = 106c02 + 122H2 0 + 16HNO 3 + H3 PO (Richards 1965b). This model assumes that:- (1) seawater is saturated with oxygen at the surface; (2) the average composition of organisms is constant so that the utilization of oxygen for the regeneration of nitrogen and phosphorus compounds from the tissues by bacteria takes place in statistically definite and fixed ratio; (3) no oceanic mixing is taking place; (4) the rates of regeneration of the nutrients are the same; and (5) either no uptake of nutrients is taking place, or else, it must take place as the reverse reaction of the one proposed for the regeneration of the nutrients. Most of these assumptions are at best approximate. Surface water may be -84- supersaturated or undersaturated with oxygen, and oceanic mixing is definitely taking place. Riley (1951) and Grill et al.(1964) suggested that phosphate may be preferentially regenerated. Preferential uptake of nutrients may also occur. Consequently, this model is only a crude approximation of the real ocean and is used as an initial step towards the understanding of the nutrient cycle. In this study, iodate is plotted against phosphate, nitrate and AOU. The data from both stations are combined as shown in figures IV-ll, IV-12 and IV-13. The lines are fitted by a linear regression program from these data points. Some of the data points from the surface water are omitted in the computation because many of the assumptions of this model are violated in the surface water. Vigorous mixing processes, active nutrient uptake and penetration of oxygen from the atmosphere are all occuring in the surface water to the extent that the model is no longer valid. The solid lines on the figures are all drawn from the slopes and intercepts given by the program. The broken lines serves as demarcations for a 3% deviation from the projected lines. The correlation coefficient of phosphate to iodate is 0.94 and the ratio of phosphate to iodate is 22,7:1. The correlation coefficient of nitrate to iodate is 0.95 while nitrate : iodate = 357 : 1. -85- Figure IV-11. The relationship between phosphate and iodate. . Data from station 60. x Data from station 61. 4 0 Cn 0S XX X PHOSPHA TE (,uM Figure IV-12. The relationship between nitrate and iodate. . Data from station 60. x Data from station 61. -87- 40 */* 30 0/ / 14J / 0/ 20 k / 10 x x 0 0.35 0.45 0.40 /ODAT ( M) 0.50 Figure IV-13. The relationship between AOU and iodate. . Data from station 60. x Data from station 61. 150 .. 0o0 / / 50 / 7 7. / / x Xe x X JX / 0 0.35 0.40 /ODATE 045 (p M) 0.50 x Lastly, the correlation coefficient of AOU to iodate is 0.88 and AOU:iodate = 2440:1. From these three ratios, AOU: nitrateaphosphate = 108:16:1 which is close to the generally accepted value of 138:16:1 (Richards 1965b). The phosphate-iodate and nitrate-iodate plots have higher correlation coefficients than the AOU-iodate plot. This is probably due to the much higher uncertainties in the AOU values. Phosphate and nitrate can all be determined by standard methods directly to within 1-2% (Murphy et al.1962; Morris et al.1963) while the AOU values are calculated from the difference of two oxygen solubility values. This increase in the amount of data manipulation will increase the uncertainties in the AOU values. Furthermore, some of the assumptions in the AOU concept mentioned earlier are probably not met and this will also increase the uncertainties. Greater precision in the iodate and AOU values will be required for better correlations between the two. The data points from the surface water in the AOUiodate plot fall away from the projected line. It implies that the preformed iodate is less than it should be. This may be due to the reduction of iodate to iodide by bacteria (Tsunogai et al,1969) giving rise to a net loss in iodate. From the AOU-iodate ratio of 2440:1 obtained from -89- this study, iodate can be divided into its generic components - preformed iodate (Ip) and iodate of oxidative origin (Iox). These results are also summarized in tables IV-3 and IV-4, The amount of regenerated iodate is small throughout the entire depth. It is usually around 10% of the total iodate while the remaining 90% of the iodate is preformed, implying that only a few percent of the iodate is recycled in the sea. One can also estimate the amount of regenerated iodate from oxygen consumption data and using the currently accepted oceanic circulation model (Broeiger 1961). Most numbers used in this estimation are only approximate and may vary by a factor of two to three, and, in addition to this, there ar'e other assumptions made in this calculation. Thus, it can only be regarded as a first approximation to the real situation. To start with, one can try to follow the presently accepted path of transport of a parcel of water from the Noth Atlantic surface to the deep South Atlantic (Broeker et al,1961; Broecker 1963). It is shown schematically in figure IV-14. The parcel of water sinks at about 60N at the Greenland Sea and then travels southwards as the North Atlantic Deep Water (NADW) and eventually reaches our point of observation at station 60 and station 61 at 300S. -90- Iodate is Figure IV-14. Vertical profile through the western basin of the Atlantic Ocean showing idealized water-mass distribution. SACW - South Atlantic Central Water. AAIW - Antarctic Intermediate Water. NADW - North Atlantic Deep Water. AABW - Antarctic Bottom Water. NACW - North Atlantic Central Water. -91- I ~- I (~#/wl/ ill N4JJWI 448W I I 'ows LA TI JDE 3oes I assimilated in the North Atlantic surface water by organisms and as the water travels downwards and southwards, it ages and the dead organisms are oxidized and consequently iodate will be regenerated. Thus, there should be a gradual increase in iodate concentration. This process assumes that the parcel is a sealed parcel of seawater so that there is no exchange with other parcels of seawater by mixing. It further assumes that the gradient of oxygen consumption with latitude is small. This has been shown to be close to the real situation by Riley (1951) in his study on the variations of the calculated rates of oxygen consumption with latitude. The oxygen consumption data were calculated using an advection-diffusion model (Riley 1951; Wyrtki 1962; Craig 1971). The assumptions in this model are:- (1) the distribution of oxygen is constant with time which means that a steady state has been reached; (2) the horizontal gradient of oxygen concentrations is negligible relative to the vertical gradient; and, (3) the ratio of the eddy diffusivity coefficient to the vertical advection velocity is constant with depth. In view of the large number of assumptions made, I must emphasize again that the calculation can only produce -92- an.approximate picture of the real situation. The calculation is shown below. The oxygen comsumption data can be taken from the average of the values reported by Craig (1971), Wyrtki (1962) and Riley (1951). Therefore, oxygen consumption in the North Atlantic Deep Water = 1.3 x 10~3 ml/l-yr = 58 x 10-3 uM/kg-yr = AOU. AOU:IO ~ = 2440:1 (from this work). Iodate production is 2 0 uM/kg-yr = 2.38 x 10-5 um/kg-yr. Assuming that the average of the surface iodate concentrations in stations 60 and 61 of this work is similar to the iodate concentration in the North Atlantic surface water (therefore, surface iodate concentration = 0.395 uM), and that the residence time of the North Atlantic Deep Water is about 570 years (Craig 1971), then, the percent increase in iodate (that is, the amount of regenerated iodate) in the North Atlantic Deep Water can be calculated to be: Iox _ -Itotal 2.28 x 10-5 x 570 x 100% 0.395 + (2.38 x 10-5 x 570 0.395 0 -93- - 3%. This result can be compared to what was observed in stations 60 and 61. Stations 60 and 61 were located at around 30 0S in the Atlantic. North Atlantic Deep Water had been identified earlier in this discussion from T-S diagrams and it was located at about 2000 to 3000 meters. Iodate of oxidative origin (Io ) has also been calculated. In station 60, at 2867 m., Iox OX = 0.033 0.458 Itotal x 100 % = 7%. In station 61, at 2825 m., I____ 'ox ,total = 0.038 0.470 x 100 % = 8%. These results agree well with the circulation model proposed (Broeer et al.1961; Broeer 1963) and similar observations on nitrate and phosphate (Broedker 1971; BroE5er et al.1972a, 1972b; Spencer et al 1972a, 1972b; Craig et al 1972). A water parcel in the North Atlantic surface water has relatively low iodate content with negligible amount of regenerated iodate. As it sinks and travels towards the South Atlantic, the regenerated iodate content increases. The discrepancy between the observed and the calculated values is probably due to the deviations from the assumptions in this calculation. -94- The mechanism for the uptake of iodine by marine organisms is still largely unknown. Literature (Shaw 1962) gives a very confusing picture. It is possible that organisms take up iodate because it has a structure similar to nitrate. This failure of some organisms to distinguish between similar chemical species has been reported in the arsenate-phosphate pair (Harvey 1960). Arsenate has been found to take the place of a portion of the phosphate required for growth by the fresh-water algae Stichococcus flaccidus (Comere 1909). At the same time, as mentioned earlier, bacteria that are capable of reducing nitrate can take in iodate and reduce it to iodide. A large number of algae have the ability to reduce nitrate using the enzyme nitrate-reductase. So, it is possible that these algae may assimilate iodate too. The average surface iodate concentration in the open ocean in the South Atlantic is about 0.395 uM while the surface iodate concentration in the Woods Hole coastal water is only around 0.2 uM. This difference may be due to the difference in the amount of biological activity if iodide is produced from the reduction of iodate by bacteria. Coastal waters have higher biological activity, hence, more iodate is being converted to iodide. This is also reflected in the high iodide concentrations observed. The total iodine con- -95- centration in coastal waters is about 0.4 uMv. This is the amount that was observed in the open Pacific (Tsunogai 1971b). Thus, the difference in the iodate concentration between the open ocean waters and the coastal waters should probably just reflect thke difference in the amount of biological activity rather than a gross depletion of iodine close to the shore. -96- CHAPTER V CONCLUSION From the various studies that have been undertaken, the new analytical technique for iodate has been shown to be suitable for fast routine analysis of iodate. The analytical method is self-consistent. The addition method has a coefficient of variation of 2.7% while the titration method is precise to within 1%. The comparative study of these methods with the co-precipitation method shows that they agree to within experimental uncertainties. The storage experiments show that seawater can be stored at low temperature for up to a month without significant change in its iodate content. The study on the distribution of iodate in the ocean shows that the iodate concentration is significantly lower in the surface coastal waters than in the surface water in the open ocean probably as a result of higher biological activity. A depletion of iodate in the surface water of the ocean was also observed. This has been observed by Tsunogai et al. (1971b) in the Pacific. The iodate profiles and the profiles of the other nutrients - phosphate and nitrate - all have similar features. The Redfield-type -97- ratios derived from the iodate data are AOU:NO 3 :P0 4 3 - :103 = 2440:357:22.7:1 or AOU:NO3~:P0 4 3- = 108:16:1. This agrees well with the established ratio of AOU:N:P = 138:l6:1 (Richards 1965b). The amount of recycled iodate is small. It amounts to about 10f of the total iodate. From the simple model calculation, it is shown that the iodate of oxidative origin increases as the water mass becomes older and moves away from the surface where biological activity is high. These observations agree with the theory (Tsunogai et al.1969) that the reduction of iodate is caused by biological activity and that iodate exhibits nutrient-like behaviors. -98- CHAPTER VI FUTURE WORK The diurnal and seasonal variations studies are inconclusive and will have to be repeated in a more favorable season and under better controlled experimental conditions. The use of a tank as previously suggested may be one way to overcome the existing problems. The analytical method for the analysis of iodide has not been successfully developed yet. A rapid and precise analytical technique will be necessary in order to use iodide as a tracer. The iodide concentration has been observed to increase towards the water-sediment interface by Tsunogai (1971b) and he proposed that iodide may be diffusing from the sediment into the ocean. More data will be needed in order to confirm this observation. In conjunction to this project, kinetic data for the iodide-iodate couple will also be needed for a better understanding of the redox chemistry of this couple in seawater. If all these data are available, models may be set up to use iodide as a tracer to study the importance of the sediment-seawater exchange processes. In an anoxic basin, pH = 7.6 and pE = -4 (Spencer et -99- al.1971) assuming that pE is poised by the couple S04 2 + 9H+ + 8e~ = HS + 4H 2 0. For the iodate-iodide couple, = 6pH + 6pE log Then, [I031 - logK. -88.5 [I-] This means that if thermodynamic equilibrium is reached, iodine should be present solely as iodide in an anoxic basin. Thus far, there is no data on iodine in anoxic basins to confirm this hypothesis. If iodine is present in the form of iodide in anoxic waters, it may be used as a tracer to study the rates of exchange between anoxic water and aerated water. This exchange may be an outflow from an anoxic basin or an upward advection of anoxic water in polluted estuaries. The penetration of anoxic water into aerated water may lead to changes in the nutrient budget which may affect marine life. The techniques for the analysis of iodate and iodide in anoxic water will also have to be developed since the theoretically interfering species shown in table 11-3 may become significant in anoxic water. The large excess of iodine in the marine atmosphere has been known for some time (Duce et al.1963, 1965; Moyers -100- et al.1972) and has been attributed to the photo-oxidation of iodide (Miyake et al.1963). With the present analytical techniques available, it will be possible to study the proposed process quantitatively. The photochemistry in the sea, other than photosynthesis, has been largely ignored and this study can serve as an initial step towards a better understanding on the importance of this kind of chemistry in the sea. -101- REFERENCES Barkley, R. A. and T. G. Thompson (1960a). Determination of chemically combined iodine in sea water by amperometric and catalytic methods. Anal. Chem., 32, 154. Barkley, R. A. and T. G. Thompson (1960b). The total iodine and iodate-iodine content of sea-water. Deep-Sea Res., y, 24. Bjerrum, J., G. Schwarzenbach and L. G. Sillen (1958). 'Stability Constants, Part II (Inorganic Ligands)'. Chem. Soc. Spec. Publ. 7. Boston Globe (1972). Sept. 15 - Sept. 29. Bowen, H. J. M. (1966). 'Trace Elements in Biochemistry'. Academic Press, New York. Broe cer, W. S., R. D. Gerard, M. Ewing and B. C. Heezen (1961). Geochemistry and physics of ocean circulation. Ins'Oceanography', (Sears, ed.). AAAS. Broecer, W. (1963). Radioisotopes and large scale oceanic mixing. In: 'The Sea', (Hill, ed.), 2. Intersci. Publ., New York. Bro er, W. S. (1971). A kinetic model for the chemical composition of sea water. Quarternary Res., 1, 188. Broecer, W. S., A. E. Bainbridge and P. M. Kroopnick (1972a). Geosecs preliminary report, Leg 3, Reykjavik to Bridgetown, Sept.4 - Oct. 1, 1972. Scripps Inst. Oceanogr. Bro, er, W. S., T. Takahashi and A. W. Mantyla (1972b). Geosecs preliminary report, Leg 5, Recife to Buenos Aires, Nov. 4 - Nov. 28, 1972. Scripps Inst. Oceanogr. Comere, J. (1909). Action of arsenate on the growth of algae. Bull. Soc. Bot. Fr., d6, 147. Cotton, F. A. and G. Wilkinson (1962). 'Advanced Inorganic Chemistry'. J. Wiley Sons, New York. -102- Courtois, B. (1813). Decouverte d'ure substance nouvelle (iode) das vareck. Ann. Chim. (Phys.) 1, 88, 304. Craig, H. (1971). The deep metabolism : oxygen consumption in abyssal ocean water. J. Geophys. Res., 76, 5078. Craig, H., K. Park and R. Weiss (1972). Geosecs preliminary report, Leg 4, Bridgetown to Recife, Oct. 9 - Oct. 31, 1972. Scripps Inst. Oceanogr. Defant, A. (1961). 'Physical Oceanography', 1. Pergamon Press, New York. Douglas, B. E. and D. H. McDaniel (1965). 'Concepts and Models of Inorganic Chemistry'. Ginn Co., Waltham. Dubravcic, M. (1955). Determination of iodine in natural waters. Analyst, 80, 295. Duce, R. A., J. T. Wasson, J. W. Winchester and F. Burns (1963). Atmospheric iodine, bromine and chlorine. J. Geophys. Res., 68, 3943. Duce, R. A., J. W. Winchester and T. W. Van Nahl (1965). Iodine, bromine and chlorine in the Hawaiian marine atmosphere. J. Geophys. Res., L0, 1775. Ewing, M., S. L. Eittriem, J. I. Ewing and X. Le Pichon (1971). Sediment transport and distribution in the Argentine Basin. 3. Nepheloid layers and processes of sedimentation, Phys, Chem. Earth, 8, 49. Fuglister, F. C. (1960). Atlantic Ocean atlas of temperature and salinity profiles and data from the International Geophysical Year of 1957 - 1958. Woods Hole Oceanogr. Inst. Goldberg, E. D. (1963). The ocean as a chemical system. In : 'The Sea', 2. Intersci. Publ., New York. Green, E. J. and D. E. Carritt (1967). New table for oxygen saturation of seawater. J. Mar. Res., 2j, 140. Grill, E. V. and F. A. Richards (1964). Nutrient regeneration from phytoplankton decomposing in sea water. J. Mar. Res., 22, 51. Gross, J. (1962). Iodine and bromine. In : 'Mineral Metabolism, -103- An Advance Treatise', (Comar and Bronner, ed.). Academic Press, New York. Hanson, W. C. (1963). Iodine in the environment. In : 'Radioecology', (Schultz and Klement, ed.). Rheinhold. Harvey, H. W. (1960). 'The Chemistry and Fertility of Sea Waters'. Cambridge Uni. Press, Lond.on. Jacobson, C. A. (1949). 'Encyclopedia of Chemical Reactions', 1 - 1. Rheinhold, New York. Johannesson, J. K. (1958). Oxidized iodine in seawater. Nature, 182, 251. Johnson, D. L. and M. E. Q. Pilson (1972). Arsenate in the Weatern North Atlantic and adjacent regions. J. Mar. Res., .20, 1. Kappanna, A. N., G. T. Gadre, H'. M. Bhavnagary and J. M. Joshi (1962). Minor constituents of Indian sea-water. Curr. Sci., 31, 273. Kolehmeinen, S.,S. Takatolo and J. K. Miettinen (1969). A tracer experiment with 1-131 in an oligotrophic l'ake. In : 'Symposium in Radioecology', (Nelson and Evan, ed.). USAEC, Oak Ridge. Kolthoff, I. M., E. B. Sandell, E. J. Meehan and S. Bruckenstein (1969). 'Quantitative Chemical Analysis'. Macmillan Co., Toronto. Kuenzler, E. J. (1969). Elimination of iodine, cobalt, iron and zinc by marine zooplankton. In : 'Symposium in Radioecology', (Nelson and Evan, ed.). USAEC, Oak Ridge. Langer, P. (1960). History of goitre. In t 'Epidemic Goitre'. World Health Organization Tongr., 44, 9. Le Pichon, X., S. L. Eittreim and W. J. Ludwig (1971a).Sediment transport and distribution in the Argentine Basin. 1. Atlantic bottoa current passage through the Falkland Fracture Zone. Phys. Chem. Earth, 8, 1. Le Pichon, X., M. Ewing and M. Truchay (1971b). Sediment transport and distribution in the Argentine Basin. 2. Antarctic bottom current passage into the Brazilian Basin. Phys. Chem. Earth, 8, 29. Liss, P. (1972). Private comm. Macadam, S. (1852). Chem. Gaz., 10, 281. Marchand, E. (1855). Mem. Acad. Med. Paris, 19, 121, Matthews, A. D. and J. P. Riley (1970). A study of Sugawara's method for determination of iodine in seawater. Anal. Chim. Acta, 31, 295. Millero, F. J. (1971). The molal volumes of electrolytes. Chem. Rev., 1, 147. Miyake, Y. and S. Tsunogai (1963). Evaporation of iodine from the ocean. J. Geophys. Res., 68, 3989. Morris, A. W. and J. P. Riley (1963). The determination of nitrate in sea water. Anal. Chim. Acta, 2L, 272. Moyers, J. L. and R. A. Duce (1972). Gaseous and particulate iodine in the marine atmosphere. J. Geophys. Res., 71, 5229. Murphy, J. and J. P. Riley (1962). A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta, 27, 31. Nightingale, E. R. (1960). Role of multiple bonding in electron transfer reactions. J. Phys. Chem., 6_, 162. Park. K. (1967). Nutrient regeneration and preformed nutrients off Oregon. Limnol. Oceanogr., 12, 353. Rankama, K. and T. G. Sahama (1950). Chicago Press, Chicago. 'Geochemistry'. Uni. Redfield, A. C., B. H. Ketchum and F. A. Richards (1963). The influence of organisms on the composition of seawater. In:.'The Sea', (Hill, ed.), 2. Intersci. Publ., New York. Reith, J, F. (1930). Der Jogdehalt von Meerwasser. Rec. Trav. Chim. Pays-Bar., 42, 142. Richards, F. A. (1965a). Dissolved gases other than carbon dioxide. In : 'Chemical Oceanography', (Riley and -105- Skirrow, ed.), 1. Academic Press, New york. Richards, F. A. (1965b). Anoxic basins and fjords. In : 'Chemical Oceanography', (Riley and Skirrow, ed.), 1, 611. Academic Press, New York. Riley, G. A. (1951). Oxygen, phosphate and nitrate in the Atlantic Ocean. Bull. Bing. Oceanogr. Bull., XIII, 1. Riley, J. P. (1965a). Historical introduction. In : 'Chemical Oceanography', (Riley and Skirrow, ed.), 1, 1. Academic Press, New York. Riley, J..P. (1965). Analytical chemistry of sea water. In : 'Chemical Oceanography', (Riley and Skirrow, ed.), 2, , 295. Academic Press, New York. Riley, J. P. and R. Chester (1971). 'Introduction to Marine Chemistry'. Academic Press, New York. Schnepfe, M. N. (1972). Determination of total iodine and iodate in sea water and in various evaporites. Anal. Chim. Acta, 58, 83. Shaw, T. I. and L. H. N. Cooper (1957). The state of iodine in seawater. Nature, 180, 250. Shaw, T. I. and L. H. N. Cooper (1958). Oxidized iodine in seawater. Nature, 182, 251. Shaw, T. I. (1962). Halogens. In : 'Physiology and Biochemistry of Algae', (Lewin, ed.). Academic Press, New York. Sillen, L. G. (1961). The physical chemistry of seawater. In : 'Oceanography', (Sears, ed.). AAAS. Skopintsev, B. A. and L. A. Michailovskaya (1933). Iodine content of White Sea water. Trud. Inst. Oceanogr. Moscow, 3, 79. Spencer, D. W. and P. G. Brewer (1971). Vertical advection diffusion and redox potentials as controls on the distribution of manganese and other trace metals dissolved in waters of the Black Sea, J. Geophys. Res., 26, 5877. Spencer, D. W., A. E. Bainbridge and J. M. Edmond (1972a). -106- Geosecs preliminary report, Leg. 1, Woods Hole to Reykjavik, July 18 - Aug. 7, 1972. Scripps Inst. Oceanogr. Spencer, D. W., J. M. Edmond and R. F. Weiss (1972b). Geosecs preliminary report, Leg 2, Reykjavik to Reykjavik, Aug. 12 - Aug. 30, 1972. Scripps Inst. Oceanogr. Sugawara, K. (1957). The distribution of some minor bioelements in Western Pacific waters. In : 'Proc. Regional Symp. Phys. Oceanogr., Tokyo, 1955'. UNESCO. Sugawara, K., T. Kayama and K. Terada (1955). A new method of spectrophotometric determination of iodine in natural water. Bull.Chem. Soc., Japan, 28, 494. Sugawara, K. and K. Terada (1957). Iodine distribution in the Western Pacific Ocean. J. Earth Sci., $, 81. Sugawara, K. and K. Terada (1958). Oxidized iodine in seawater. Naturejl82, 250. Sugawara, K., K. Terada, S. Kanamori, N. Kanamori and S. Okabe (1962). On different distribution of calcium, strontium, iodine, arsenic and molybdenum in the Northwestern Pacific, Indian and Antarctic Oceans' J. Earth.Sci., 10, 24. Sverdrup, H. U., M. W. Johnson and R. H. Fleming (1942). 'The Oceans, Their Physics, Chemistry and General Biology'. Prentice-Hall, Inc., New Jersey. Tsunogai, S. and T. Sase (1969). Formation of iodide-iodine in the ocean. Deep-Sea Res., 16, 489. Tsunogai, S. (1971a). Determination of iodine in seawater by an improved Sugawara method. Anal. Chim. Acta, 55, 444. Tsunogai, S. (1971b). Iodine in the deep water of the ocean. Deep-Sea Res., 18, 913. Tsunogai, S. and T. Henmi (1971). Iodine in the surface water of the ocean. J. Ocean. Soc. Jap., 2Z, 67. Vinogradov, A. D. (1953). 'The Elementary Composition of Marine Organisms'. Sears Found. Mar. Red., Yale Uni., New Haven. -107- Voipio, A. (1961). The iodine content of Barents Sea water. Rapp. Cons. Explor. Mer., 149, 38. Von Arx, W. S. (1962).'An Introduction to Physical Oceanography'. Addison-Wesley Publ. Co., Mass. Wada, E. and A. Hattori (1972). Nitrate distribution and nitrate reduction in deep sea waters. Deep- Sea Res., 12, 123. Weiss, R. F. (1970). The solubility of nitrogen, oxygen and argon in water and seawater. Deep-Sea Res., 17, 721. Whittaker, E. J. W. and R. Muntus (1970). Ionic radii for use in geochemistry. Geochim. et Cosmochim. Acta, 31, 945. Winkler, L. H. (1916). De Jodid- und Jodate- iongehalt des Meereswassers. Z. Angew. Chim., 29, 205. Wyrtki, K. (1962). The oxygen minima in relation to oceanic circulation. Deep-Sea Res., 2, 11. -108- Appendix 1. Hydrographic data of station 60. 0" Xz O C 0U > SVv >N D Q (V . eV Q. NO M tn 9- %0 0 C 0 t. Je4 a.J U) C ( N# ^ * ) O (NJ : ca 4O 0 . ) 0 0 co (V0DC.-4 (N C: O V .. 3 V (V. D 0' D( N 3C4 t-( k 0. (V 4 CO C) n 0.4 C v ^m N O ) 4 C% "N OnD ene)W) co 4 00 C2 k D - P- J'.4 co 0 - 4r) a3 ci N (V C) 3-.4 InIt-t--) ' O N) C) . Nre . t- o -D - - I)t)e)--')W K)4. -0& (VNNNV(V CD -4 'A 00.4.4 C 0 30.C)% fN ) N) N(V( c-i (j (Vj " (V (V f N CZ) D ( . e N l C%0O .4 .4 3re&. - C3 0 3 PO fo4 ... - ' (V . U- \ CIN 9 W 5) O 0' ' W K to Lr N (V r e4 r C% N Iu(V O4 D(IN 1 VI0000 IU < n n CD N( (VC4N rvN"'D-4 no o- e43- -e) . 'D (VN'O N co C, (VcVVcVN A A ) 'A41.-I ~003-Jr ODU)4)%-"-r--- 'J- rO ... :2 tK) co -I 3 90 f3 40% e 90In 4-4 90 t-in O2# 4fl.42 0- 3- -4 -4 fn W) In CO 0% 4)N r1N) N 0 ) 0 N c i1 4).. 4 0 (V (V 0 r- CDc0 o > N M0 0 4 D N) ID % 3 (V cu Co. N ( Co0% V %b O C C0 4 C ' (VO I- C(D )1 it N aiko In ') .N) to to N)lN (V cv (V (V (V 0' 0 0 r j (VK) co to .4 .... 1c 0 0. CO 0, d) %D N e O\ 20 CO ve * * * 4 K) fq % 0-4 *c * . . W) N (V . en 0 3-3 a 3C re) _4 CA I OQ CU CY 0 Co 0N NC o 3V r- c- o0 C) 0Z -C 0.43-4 0C .N) .4.1- ,o Ln %o cv fu cl 0 f ) fn 4T r- 01 Ors Ln %D O SC) - . to eN)03O) N N% to N If .4 4C C0C% %0 QD %0 4"4W4 W4 40 .3. D N 3D 4) (N -4 N) "N) 4 )4 N ) N e)f)Na- (I N 04 (V( C o . r .4 CN 4 -a . N) v 4 4"-4 -4 -4 (VN( (V (V (V a%0 - D n d) . -4-4 IN -O) (le 4 . (N i NN 7 ) 4 3 14- C4 Z e)')N))" 4 4 ,A u- 4) .4 tY to CJ1- " EV V( i V(N M (V V N) ( ) %3 D .0D %DIr4 D O4q v i 0 "4 V4 -4 -q 44 D en % & 0i:4)0 (V t- oC>0.4.4 0 ( . 0C0 -a% 'A I- .4 0 0 '-k ' C) (IVoN (V r- ru- r c) e~-.Y -1 ~QC- O ~ . fnco %Dca a . 4)0 00 '0 ,4 Ca %D 0 a% 4) 6(D r4 0' (O0 O N 4(V0 % to P C% al M l. o 0 e (V N. ) 4n (V (V (V ( (V - .. q 0 -Z I- CtrW 0 ) o) PO -' kol.~ 4eY 0%ul N . C :1, -. . t- N N)"cN CD co a, N a 0000 CD .) 10 N) (VNV V 0.-. .11-03-01- 0 23-33 3 "D PCNl C-DC_ C;c Cr- (V t- 90 N) N) 1- 7' 0.41-w 000(O e4 00 1 .4O . .404) .40. .4 0 .4OQOO 4 -4 0 C0 '30 c roN V%;cMa coCDco co a, 1- c- 3- Cm 3, 30o %D% g 0c r O10 PO C43 .4-13- 1- c.4 ) CD ~ o ~ 00 4cvC" O 0O .40 . N) (V v -4 N 0 Ntn vccu re 03 " W) 1 - tN N 4 . .4.4 V e) e4 N) mt-a, L40- Z D0 V4 W4 "4 co _r K) kD to Wo e-I O _r %D t0 4 Ij p10 :: r0 - (VN -10D4.4 N r)O r - 4r Q C N Z U = cO N "I Z n 44 4 . - -4 N N N N N K PC) to NNNNCV .- 1-1 (V (V 1-01-1- -N) N inW) (V cmto 3-3N N) No \D .4 N) 3 C N r10 C NN N 0 CD -4 4 9N30 CD % ; ;0.4 c NNV .4e n 4 .C1 .4 D -4 4t a,494-4 a,a*q4 4 o en 4 C %D -109-0 %D%0 = ~ . N I, N ( V N- ) -444 00 ) Z V)-K) r- ) _r C% 4 (V cN v N (V to (V (V c -W)II nN)(V to '30 -nT = N) N) (. C N) N) N 3- 3a% On0. ON- N 4 fy t 0- O (V'3 ~~ 0W)4 ~ q -4nt N .. f-7' 3- (V :2. (V .4 . N c) N C) o ) PC K)r)c tt e C .n C% r- to . 4 3.4 311-4 r. " .0 N u (V _r.e .1 - 43- o3-J &C~l I- D O D .v to4)01. . . 00 CY e (V r- 0. 4.-4 '4in o) ' 3 N i ( N 7V-4 -4-4 tOtO) N-) K) e-l) N V) " Nr NNjV N4 (N N VV( N 1V c 03-00 0 CN 'i cN (V(V((V NV NeCru(V N N(Vcrj 3" z ,D . e . N t-4 3 N r-r- .4. hl . 0 r0 7% lN:00N N N r- 4 z ".4.4" 01 p- 0 rN) cLnt N o4) KO N) . -: O - V3-\V0N) "n N t .) (VN)K) -- D LO n 14 0 i-- C>. in .1- Ot s (4 (V (V ( CO zi -4 (V N ) 0301 N).ej\ 0.41 .4' 04) z (V0 , 4 ) 0.4 n C% .) NNNNN 0r C: rer (v (VJ N1 (V-) co a1) C' (V -( 4 1- 0.4 Nco- .. O 4 . N (V (V en - K) N) .- v 1-0 (V C) (V 0 (V (Vj f 10 . W) En p 00o C% q . co Z" * 0 c 3 0 0 to -q f) N NV %O " NN O- toi . to -IN) ) 3- 3.4 (V 03 . N' '3 Nto to 00e4 .. 4. . (V a, 3l- W) .001-0't 0.4 (V IY (V N) N) 0D-0' C% cc1 a 0 a C co e4 LO (V C% * eon to O N w ko ) V) n O z3 a- 10 % CDV4iLO ... C% 9to cD 44)3-e - O3-1 .40' 304) '34) 4 .4.4.O (V .N \00 '34) (V 0 V . r4"4.-4 .4 %0D %U %D 10 -4 D P It t 1 10 r cr(a- N co %D %DW) -r " 4 0D fV0u'3D% _ D It.2 t& %D Z . '0 3.3f- 1-t -r inrLo \or N) "V-40 0,c (v C v (V (V K) 0 . 0 o N0 .4 T4 .1 .4 0N) L-%.4 t14 .-N e1) 3ID I- P- a~ -N) !v fu NW) N a,1 - Ln) v,. q= cNM o C) to K) n tN) 'n : 1- 3> 0 3 . .4 . N) N) N) N) N 3 N) N)4)N N)"0\O (V CO %.a N 13 eN) N)).4 .1-N en Cto o ZI, O zr a% -r a% W-) N(VOIDC, 0' -41C(\o %D An CD 00%cu Ln CDfC% o 1 n . . .4) c; -4n q4 _rCw cyO CD -4eD in~ % . 3l r- 3. -rt 3-011 4 'o 0 i- to (V -D ro -r CV 0.4 'VN) NNNNN %4n00 to %D o . .. 4 (VNr) k -0 o r _ .-A .4-1NI)C e . .e (V (V (V (V (Vi i3-' :I, ) N 0 (l PO .2C3 O O3 0 0 N) C) fcj (V1 (V -V (Vi 103D c N4 N) 1- 1DI a r U' ID ) ( -D NNNNN c~ (V (V Li cot Z. * O U - x 0e C K0:001-0 NNNN e N -4r- " 44n j -r C' _0 C> V Ov % N0c kf D V (v N 1% D- 0 e e 000OO00 CO00.ed.4 C e 0 00 C) N Sa' N CI CD NNN Z3 S SN 0 - Li 0 <n (-V LoI C- On-:ID ;tn 0W V) OD 43 OD -4 %D4 O4 4)0 f- D O tt n N 1-7%Q Z, %D 2 r- t- 0 Z, 0S4- (V 3-\ 0c 0(V 4 cu U.7LiC 0% a' 4 W ) 01.-10 0 . . . . " m3 . . .* In 'n .0 0 -n C,- K) ) Itn T) In ) LO a' en D .4 - 0 -YC) 0 0 0 0 _E N < 0 Q Q. Li enU N o 0 t X 04&)3- .. 4., .:1 ui CD C5 C5 C3 C :D Li L...~itoa 0 C3 -r ( CP - DATA' REPORT H/V Kf'ORR POsItION 32 31.05 STATION -CAST 60 1 60 0 60 1 60 60 8 8 60 8 60 60 60 60 60 8 1 60 60 42 UTTLE DEPTH M NUPBER 21 4125 16 4197 22 4223 17 4238 18 4267 19 4297 GEOSECS IEG V FIRST CAST 1330 22 NOV 72 0.0W TEMP DEG C 0.1v1 00125 0.130 0.113 0.105 06102 POT TEMP SALINITY SIGMA DLG C THETA 0/00 -0.120 34.672 27.076 -0.102 34,669 27.076 -0.100 34.670 27.877 -0.198 34.667 27.875 -0.209 34.672 27.OO -0,215 34,669 27.870 4323 4324 4346 4367 4308 0.099 -0,221 34.666 8 8 20 23 21 22 23 0.101 0.102 -0.219 -0.220 8 1 24 24 4405 4414 8 SIGMA z 46.971 47.299 47.412 47.482 47.617 47*745 SIGMA 4 46.130 46.130 46.138 46.139 46.144 46.143 0.1,03 0.106 27.876 27.077 27.870 27.878 27.876 47.860 47. 8b6 34.669 -0.222 34.669 -0.221 34.666 48.057 48.149 46.142 46-.143 46.144 46.144 46.142 0.109 0.111 -0.220 34,671 -0.219 34,667 27.080 27.876 40.226 48.261 46.145 46.142 ********* 34.668 47.966 STATION NIS DEPIH 4424 OXYGEN SIL P04 UM/KG UM/KG UM/KG 225 122.6 2.20 226 2.22 126.3 226 124.1 2.21 60(CONT) CORR.M -1403 ALK(T) C02(T) CO2(6C)PRESS UM/KG U EQ/KG UM/KG UIM/KG 0.13AR 2357 2234 32.35 2253 4194 32.44 4268 2356 32.64 2244 2237 42J4 431u 4340 4370 226 127.2. 125.4 2.23 2.21 4397 32.44 32.74 2355 2248 2247 4398 4421 4442 44b4 225 126.5 125,4 2.23 2.21 PRELIMINARY DATA ONLY.....NOT FOR PUBLICATION********** 32,44 32.64 2357 17 2241 DEC 2246 72 4481 4490 PAGE 2 Appendix 2. Hydrographic data of station 61, DATA R/V KNIORR POSITION 36 0.0S 45 SIGMA 2 26.093 26.308 26.736 27.139 27,346 STGMA OXYGEN SIL P04 NO3 4 UM/KG UP/KG UM/KG UM /KG 42.752 0 .00 241 0.12 1.1 240 42.047 0.11 0.00 1.1 43.129 226 1.4 0.30 2.83 43.326 224 4.68 1.7 0.41 229 1.7 43.369 0.40 4.10 27.537 27.748 27,909 28.243 20.641 43.404 235 143.424 43. 4 64 12.040 35.140 26.541 26.553 26.577 26,616 26,725 235 i28 215 224 6 7 n 9 10 224 260 317 363 426 14,900 14.600 14.327 13.365 12.106 14.865 35.661 14,758 35.646 14,278 35.540 61 5 61 61 61 61 5 5 5 5 11 12 15 16 17 405 535 596 647 692 12.181 11.612 12.114 11.540 9.223 7.009 6.202 35.242 35.13, 34.720 34.433 34.336 26.791 2u.817 26.903 27.012 27,043 23.972 29.226 29.618 29.990 30.244 43.867 43.941 44.227 61 61 61 61 61 5 5 5 5 5 18 746 19 20 21 22 5.240 792 847 894 996 34.246 34.249 34.254 34.232 34.224 27.097 27.132 27.162 27.101 27.229 30.563 30,818 31.105 31.350 31.802 44.003 44.866 4.385 3.C55 5.176 4.889 4.648 4.313 3,779 23 24 1 2 3 1145 1307 1306 1533 1700 3.439 2.913 2.776 2.671 2.764 3.354 2.819 2.677 2.561 2.637 34.265 34,325 34.364 34.457 34.572 27.302 2 1.398 27.441 27.525 27.610 4 5 6 7 8 18661 2020 21A6 2346 2504 2.832 2.077 2.899 2.690 34.655 2.719 34.720 3.01b 2,939 2.824 34.044 2.733 34.850 27.672 27.720 27.766' 27,011 27.030 36.369 37.153 37.905 38.660 39.406 45.703 45.737 45.765 9 10 11 12 15 2665 2825 2921 3165 3362 2,010 2.750 2.655 2.117 1.697 2.590 2.522 2.405 1.861 1.431 27.849 27.069 27.881 27,665 27,065 40,159 40.901 41.619 42.474 43,398 45.798 234 45.025. 241 245 45.049 222 45.893 218 45,942 15 16 17 18 19 3497 3545 3720 5912 4096 1,402 1.3b6 1.045 0,700 0.451 1,207 27.069 44,021 44,249 45.098 45.954 45.971 45.9b3 0,405 34.698 27.868 27.860 27,069 0.144 34o683 27.071 46,805 61 61 61 61 9.293 7, 07*, 6.267 4.956 4.718 . SALINIT '?ISIGMA 0/00 WHTA 35.82i 26.067 35,931 26.161 35,783 26.354 35.641 26.478 35.628 26.500 5 5 5 5 5 61 61 61 61 61 STATION GEOSECS LEG V FIRST CAST 1454 24 NOV 72 0,0W STATION BOTTLE DEPTH TEMP POT TEMP -CAST M DEG C DEG C NUPBER 61 5 1 17.436 -5 17.435 61 5 51 17.392 2 17,303 86 61 5 3 16.114 16.100 61 5 15.104 15.001 4 140 61 5 5 188 14.929 14.099 REPoRT 13.312 35.326 2.726 34.776 34,066 34,004 34,086 34.12 34,774 34,760 1.090 34.743 0.759 34.722 43.599 43.809 44.539 44.648 244 244 240 242 247 263 6 52 87 150 190 2046 2032 2046 2059 2064 2067 2044 2043 2061 2052 226 271 9.08 10.74 2331 2330 2342 2319 2305 0.70 9.08 2311 0.78 10.25 20662 1.13 1.45 1,61 15.73 21.01 22.97 2306 2205 2274 11.0 13.1 15.6 17.1 22.2 1.63 1.6B 23.66 24.73 1.75 1.78 1.87 25.71 26.39 27.96 1.7 1.7 2.0 0.39 3.1 0.70 0.81 3.6 3.1 3.4 5.6 8.7 10.6 0.41 0.!50 4.19 4.49 6.05 45.075 251 32.657 33.518 33.930 45.191 233 216 208 31.2 43.2 2.02 2.14 30.11 31.87 48.5 2.14 34.695 45.491 35.501 45,564 194 107 57.3 60.9 2.22 2,15 32.65 33.33 189 198 207 60.4 58.0 56.0 2.06 1.04 224 45.0 229 1. 6*4 45.1 1.59 46.1 42.9 1.56 1.40 1.45 23.84 70.5 06.9 1.74 1.08 26.23 28,14 96.4 145.618 45.66b 46.021 46.063 217 215 217 46.095 220 2347 2347 2343 1995 2004 2047 2037 2023 44.920 44.973 45.397 ALK(T) CO2(T) CU2(GC)PRLSS U EG/KG UM/KG UM/KG 0.-AR 2023 2022 2041 2055 2051 259 254 256 45.340 NIS 61 UEPTH 4933 CORR.M 43 . 2 1083 31.08 28.03 27.75 25.01 24.23 22,76 22.37 1.98 29.61 2.08 30.78 116.0 122.0 2.15 2.18 31,66 32,25 320 367 430 490 2272 2108 2050 2068 2062 2081 2096 2269 2271 2272 2103 2116 2125 2126 2132 2093 2092 2118 2122 2131 2265 , 2155 2151 2165 2166 2215 2223 1157 1321 1401 2211 2211 2206 1o90 2053 2272 2274 2296 2306 2313 2u7 2016 2094 2179 2196 2205 32.25 106.6 ********* PRELIMINARY DATA ONLY.....NOT FOR PUBLICATION********** 2329 2332 2322 -2214 540 602 b53 699 753 800 903 100b 155U 1728 2325 2205 2321 2188 2326 2173 2101 2537 2324 2171 2337 2204 2701 2064 3023 3211 2344 2219 2190 2173 2172 2211 2224 2347 2221 2233 2242 23 3 2359 2237 2242 2246 22 U 17 DEC 72 2213 2376 3413 3550 3630 3737 3975 4164 PAGE 1 DATA REPORT R/V POSITION KNOnR 36 45 0.OS STATION COTTLE DEPTH tiur'OLR M4 -CAST 20 4273 61 1 21 4463 61 1 61 1 22 4647 16 4704 61 8 17 4753 61 8 61 8 18 4784 GEOSECS LEG V FIRST CAST 1454 24 NOV 72 0.0W POT TEMP TEMP orG C DEG C -0.017 0.305 0.209 -0.130 -0.188 0.171 0.157 -0.200 -0.215 0.155 -0.224 0.152 SALINITY SIGMA 0/00 IIETA 34.676 27.074 34.671 27.875 34.668 27.076 34.667 27.876 34.665 27.875 34.667 27.077 SIGMA z 47.634 48. 467 49.285 49.536 49.749 49.970 34.666 34.665 34.655 34.674 34.672 27.076 27.075 27,067 27.882 27.881 50.071 50.078 0.155 -0.224 -0.223 -0.225 -0.228 -0.231 0 .15; 0.160 0.160 -0.231 34.672 -0.231 34.670 -0.231 34.674 27.881 27.879 27.883 50.421 50.509 50.520 4825 4e27 4644 4865 4885 0.155 0.156 .0.156 4905 4924 4926 0.156 ********* 50.143 50.240 50.332 DEPTH SIL SIGMA OXYGEN UM/KG UM/KG 4. 46.116 222 124.4 46.131 225 125.3 46.138J 226 126.3 46.140 46.140 46.143 46.142 46.141 46.134 46.149 46.148 IJIs STATION P04 lv03 UM/ K6 UM/KG 2.19 2.19 2.20 32.35 32.44 32.54 61(CCUNT) 4933 CURR.M ALK(T) C02(T) C02(GC)PRESS U EQ/KG Ui/KG UM/KG D.UAR 2355 2240 2249 4351 2356 2245 2247 d247 4768 4836 4691 226 127.2 2.21 32.64 2358 2254 226 127.3 2.21 32.64 2361 2237 4913 4915 49-32 4953 4973 46.140 46.146 4541 473U 4994 2247 46.150 PRELIMINARY DATA ONLY.....NOT FOR PUBLICATION********** 5015 5017 17 DEC 72 PAGE 2