February 28 & March 1, 2005 Assigned reading: Ch 11 pp354-384
advertisement

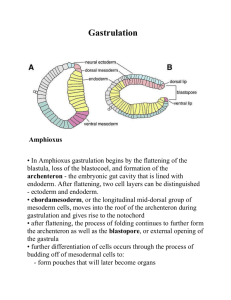
February 28 & March 1, 2005 Topic: Early development of chick and mammal Assigned reading: Ch 11 pp354-384 Minireview on studies by Gardner & Zernicka-Goetz Chick and mammalian eggs are dramatically different. Chicken eggs have a large yolk mass that is not penetrated by cleavage planes. Mammalian eggs, in contrast, have little yolk, and cleavage is holoblastic. However, by the gastrulation stage, chick and mammalian embryos look very similar. EARLY DEVELOPMENT IN BIRDS (chicken) CHICK CLEAVAGE Chick cleavage is “discoidal”: The cleavage planes are meroblastic: they do not proceed through the entire egg. The young embryo is formed in a small disc of cytoplasm at the animal pole. (see Figure 11.12) The early cleavage cells are continuous with each other, with the yolk at their bases (Fig. 11.12) In later cleavage stage embryogenesis, the "blastoderm" is 5 -6 cell layers thick. Figure 11.13 &11.14A In the center of the embryo, the deep cell layers die and are shed, leaving a disc one cell deep. The edges are still several layers deep. The center of the embryo appears relatively clear and is called the "area pellucida". The edge appears more dense and is called the "area opaca". At the interface between these two cell populations is the marginal zone. Turn the disc on its side (Fig 11.13) The space between the embryo and the yolk is called the subgerminal cavity (don’t confuse with blastocoel). Formation of the blastocoel: The "area pellucida" is one cell layer thick. That cell layer is called the epiblast. To form a blastocoel, the embryo must create a second layer, called the hypoblast. The cells that form the hypoblast come from two sources: Cells delaminate from the epiblast. Migration of deep cells from the posterior region. The epiblast and hypoblast are joined at the edges, and the space between them is the blastocoel. The chick embryo will be derived entirely from the epiblast. The cells of the hypoblast will contribute to extra-embryonic tissues. D/V AXIS FORMATION The dorsal-ventral axis is determined by the pH gradient between the albumin (egg white) above the disc (pH 9.5) and the subgerminal space (pH 6.5). This differential is created by active transport of ions and water across the cell layer as the subgerminal space is created. There is also a membrane potential difference, such that there is a higher concentration of positive ions in the subgerminal space than in the albumin. Albumin basic, negative Subgerminal space acidic, positive The dorsal side of the embryo is the side contacting the albumin, and ventral side is adjacent to the yolk. CHICK GASTRULATION Gastrulation initiates with the formation of the "primitive streak" or "primitive groove". The primitive streak is analogous to the amphibian blastopore. The primitive streak is first evident as a thickening of the epiblast at the posterior region of the embryo. See Figure 11.14. The visible "streak" is due to the ingression of endodermal precursors from the epiblast into the blastocoel. The top of the epiblast defines the dorsal side of the embryo. NOTE: Gastrulation in chick involves the movement of individual cells. Contrast with frogs, in which gastrulation involves movement of sheets of cells. Ingression of individual cells requires that cells lose affinity for their neighbors and initiate migration. The streak eventually extends 60 - 75% of the length of the area pellucida. The endodermal and mesodermal precursors form two populations. The deep cells displace the hypoblast anteriorly. These cells will contribute to endodermal structures. The mesodermal cells spread between the endoderm and the epiblast, and they will form the mesodermal structures. Figure 11.15 Regression of the primitive streak. As gastrulation completes in the anterior, further development proceeds, and the primitive streak moves posteriorly (back towards the blastopore). In avian (and mammalian) embryos, there is a gradient of developmental maturity, in which the anterior is more developmentally mature than the posterior. This means that the anterior initiates organogenesis before the posterior completes gastrulation. The ectoderm expands via epiboly to surround the entire yolk mass. Thus, the yolk becomes enclosed within the embryonic tissue. Cell division plays an important role in the expansion of the ectoderm. Henson's node is the organizer. The anterior end of the primitive streak (or primitive groove) is called Henson's node. Henson's node is functionally similar to the dorsal lip of the amphibian blastopore. The cells of Henson's node secrete inhibitors of TGF-beta ligands. The chick equivalent of the N.C. and the organizer (Koller’s sickle and Henson’s node) Experimentally, how would you identify the chick organizer? (See Figure 11.20) How would you identify the chick equivalent of the N.C.? The A-P axis of the chick is thought to be dictated by gravity as the embryo roles down the oviduct. The blastoderm is less dense than yolk and stays on top the egg. The more elevated region of the blastoderm become the posterior. See Figure 11.17 Koller’s sickle, which overlaps with the posterior marginal zone (Fig 11.19A) is equivalent to the amphibian Nieuwkoop Center, as it can induce gastrulation in neighboring tissue. Study question: Assume that you are a researcher who is trying to identify the molecules that mediate organizer activity in Henson’s node. You have several candidates that you would like to test. Describe an experiment that would allow you to test the hypothesis that a given molecule plays an important role in the function of Henson’s node. 1. correlative; is it expressed at the appropriate time & place. 2. loss-of-function: is it required? 3. gain-of-function: is it sufficient? Note: Any one experiment can support or refute the hypothesis, but multiple experiments are needed to build a strong case for a given hypothesis or model.








![Mes0derm induction and development of the ~]~ERSPECTIVES](http://s2.studylib.net/store/data/012930763_1-7ac31f9d236199dfbffb2f38a21e87e2-300x300.png)
