II. Heat
advertisement

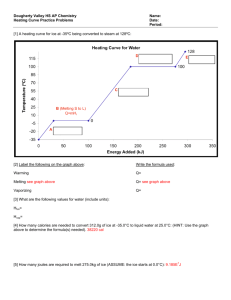
II. Heat 1. Heat and energy A little bit of history: • Old theories • Robert von Mayer (1814-1878) • James Joule (1818-1889) • Hermann von Helmholtz (1821-1894) 1 cal = 4.186 J Calorie: the amount of heat required to raise the temperature of 1 gram of water from 14.5 ◦C to 15.5 ◦C. 2. Specific heat dQ cmdT Q cmT 1 dQ c m dT cwater 1cal / g C 4190 J / kg K 1 cal = 4.186 J 2a. Heat capacity Q C T C cm dQ CdT dQ C dT 2. Calorimetry Q 0 ΔQ=cmΔT i Example: 200 g of water at the temperature of 80°C is placed into a 50 g glass at 20°C. What is the final temperature of the system. mw = 200g Tw = 80º C cw=1.0cal/(gCº) mg = 50g Tg = 20º C cg = 0.20cal/(gCº) T=Tf - ? Qi 0 Qw Qg 0 Qw cwmw (T Tw ) 0 Qg c g mg (T Tg ) 0 cw mw (T Tw ) cg mg (T Tg ) 0 T cw mwTw c g mg Tg c w mw c g m g ci mi (T T )i 0 c mT T c m i i i i i 3. Phase transitions. Q = ±mL ΔQ = ±ΔmL Latent heat: energy required for a material to change phase, even though its temperature is not changing. Example: 100 g of ice at the temperature of -40°C should be melted and heated to the temperature of 20°C. Calculate needed heat. m= 100g Ti = -40º C ci=0.5cal/(gCº) T = 20º C cw =1.0cal/(gCº) Tc = 0º C L = 80 cal/g ΔQ - ? Q c i m(Tc Ti ) mL c w m(T Tc ) mc i (Tc Ti ) L c w (T Tc ) Q 100 g * [0.5cal/(gC º ) * (0º C (40º C)) 80cal / g 1.0cal/(gC º ) * (20º C 0º C)] 100 g * [20 80 20]cal / g 12000cal Example: Ice and Water 200 g of ice at -40º C is mixed with 300 g of water at 20º C. mw = 300g What will happen with ice and water? mi = 200g Tw = 20º C Ti = -40º C ci=0.5cal/(gCº) cw =1.0cal/(gCº) Tc = 0º C L = 80 cal/g Qw cw mw (Tc Tw ) 1.0cal / gC 300 g 0 C 20 C 6000cal Qi ci m i (Tc Ti ) 0.5cal / gC 200 g 0 C 40 C 4000cal Qiw mL m 80 cal/g Qw Qi Qiw 6000cal 4000cal m 80 cal/g 0 m 25g T 0 C 25 g of ice will be melted The temperature of the mixture will be 0º C Example Energy is required for a material to change phase, even though its temperature is not changing. Avogadro number and number of moles One mole is the amount of substance that contains as many elementary entities as there are atoms in 12g of carbon -12 n N A 6.022 1023 molecules / mol N NA n - number of moles N - number of molecules NA - Avogadro number msample - total mass of sample m - mass of one molecule μ=M - molar (atomic) mass (“weight”) Molecular weight msample mN mnN A n mN A Molar specific heat (molar heat capacity) Q cM nT c M n cm sample dQ cM ndT cM c cM 1 dQ n dT Question A beaker contains 0.200 kg of water. The heat capacity of the water is ___ J/˚C. Assume: the specific heat of water is 4000 J/(kg·˚C). 1. 200 2. 3. 4. 400 600 800 Question The heat capacity of the water in a massless beaker is 800 J/˚C. The amount of heat needed to increase the temperature of the water from 15.0 ˚C to 30.0 ˚C is ___ kJ. 1. 4 2. 8 3. 12 4. 16 Question A liter of water is put in a sauce pan on a stove. In order to heat the water at 10 ˚C/minute requires a heating power of ___ Watts. (Ignore the heat capacity of the pan. Assume the heat capacity of a liter of water is 4200 J/˚C.) 1. 500 2. 700 3. 900 4. 1100 Question Block A: 1.0 kg, c = 400 J/(kg·˚C), initial temperature = 100 ˚C Block B: 2.0 kg, c = 200 J/(kg·˚C), initial temperature = 0 ˚C After the blocks are put into thermal contact and reach equilibrium, the final temperature is __ ˚C. 1. 2. 3. 4. 50 30 70 40 Example How much heat does it take to heat 1.00 kg of water ice from 10 ˚C up to 100 ˚C and boil the water away as steam? Given: cice 2.10 kJ/kg·°C cwater 4.19 kJ/kg·°C Lf 334 kJ/kg Lv 2256 kJ/kg 1. Heat the ice up to 0 °C Q1 mcice T (1 kg)(2.1 kJ/kg·°C)(10 °C) = 21.0 kJ. 2. Melt the ice at 0 °C Q2 mLf (1 kg)(334 kJ/kg) = 334 kJ 3. Heat the liquid water up to 100 °C Q3 mwater cwater T (1 kg)(4.19 kJ/kg·°C)(100 °C) = 419 kJ 4. Boil off the water into steam at 100 °C Q4 mLv (1 kg)(2256 kJ/kg) = 2256 kJ Total Q Q1 Q2 Q3 Q4 = 21 kJ + 334 kJ + 419 kJ + 2256 kJ = 774 kJ + 2256 kJ Q = 3030 kJ = 3.03 MJ Most (~ 3/4) of the heat is used in boiling off the water. Example A 10 g ice cube at 0 ˚C is put into 100 g of water at 20 ˚C. Does the ice cube melt completely? If not, the final temperature of the water + ice cube would be 0 ˚C. If so, what is the final temperature of the water? Lf 334 kJ/kg cwater 4.19 kJ/kg·°C The heat needed to be added to the ice to melt it is Qice mice Lf (0.010 kg)(334 kJ/kg) = 3.34 kJ. The heat given off by the liquid water when it is cooled to 0 °C is | Qwater | mwater c | T | (0.100 kg)(4.19 kJ/kg·°C)(20 °C) = 8.38 kJ. Thus there is more than enough thermal energy in the water to completely melt the ice. The heat absorbed by the ice to raise its temperature to the final temperature T2 is Qice mice Lf mice cwater (T2 Tice 1 ) mice Lf mice cwaterT2 . The heat absorbed by the water to decrease its temperature to the final temperature is Qwater mwater cwater (T2 Twater 1 ). The sum of these two heats adds to zero: mice Lf mice cwaterT2 mwater cwater (T2 Twater 1 ) 0. Solving for T2 gives m c T mice Lf T2 water water water 1 (mice mwater )cwater (0.100 kg)(4.19 kJ/kg·°C)(20 °C) 3.34 kJ = (0.010 kg + 0.100 kg)(4.19 kJ/kg·°C) = 10.9 °C .