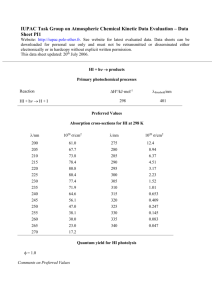
Coursepack for Air Environment Workshop in the Chemical Sciences May 24, 2006
advertisement

Coursepack for Air Environment Workshop in the Chemical Sciences May 24, 2006 Instructor Theodore S. Dibble Associate Professor of Chemistry SUNY-Environmental Science and Forestry Syracuse, NY 13210 tsdibble@syr.edu (315) 470-6596 Contents: A. Outline B. Reference works C. Selected pages from JPL Data Evaluation #14 D. Data useful in teaching kinetics calculations E. Data on climate change F. Comparing the stratospheric chemistry of the halogens G Heterogeneous chemical kinetics H. Concentration data for the troposphere I. Lifetimes with respect to OH, O3, and NO3 J. Degradation pathways of alkenes, oxygenates, aromatics K. Tropospheric photochemistry A. OUTLINE I. II. Ozone: The Good, the Bad, and the Ugly Atmospheric Structure and Circulation Temperature defines the structure of the atmosphere Pressure vs. altitude - the barometric law does not apply below ~130 km ! Atmospheric composition and units Vertical mixing times in the global atmosphere Global circulation patterns Horizontal mixing times Exercise: Distribution of CF2Cl2 and CO Spectrometry and Photochemistry Absorption cross-sections Spectrometry of ozone Solar flux versus wavelength, altitude, and Solar Zenith Angle Quantum yields Photolysis rate constants Activity: Calculating photolysis rates with Excel Kinetics: The atmosphere is not in equilibrium Pseudo-first order approximation – Cl + O3 Steady-state approximation – ClO example Ozone loss from CFCs Catalytic cycles and storage Activity: Calculating the fate of atomic chlorine [Comparing the stratospheric chemistry of the halogens] Chemistry of radical families are coupled 3rd order kinetics – practical Activity: Calculating the fate of ClO CFCs and CFC substitutes [Degradation pathways] Ozone Hole Ozone hole – observations Dark and Heterogeneous chemistry [Heterogeneous Kinetics] Tropospheric Ozone Photostationary state relationship CO and CH4 Lifetimes of selected VOCs Degradation pathways of alkanes [alkenes, oxygenates] [Relevant photochemistry] Propagation and termination reactions Activity: Steady state concentration of C3H7, C3H7OO and C3H7O radicals Meteorology and air pollution Global climate change Window regions and saturation Activity: Saturation at a single wavelength III. IV. V. VI. VII. IX. B. REFERENCE WORKS Textbooks Finlayson-Pitts, B. J. and Pitts, J. N., Jr. Chemistry of the Upper and Lower Atmosphere, 2000. Jacob, D. Introduction to Atmospheric Chemistry Seinfeld, J. H. and Pandis, S. N. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change 1998 (2nd edition available August 2006) Wayne, R. P. Chemistry of Atmospheres, 3rd edition, 2000. Warneck, P. Chemistry of the Natural Atmosphere Key References Atkinson, R. Gas Phase Tropospheric Chemistry of Organic Compounds, J. Phys. Chem. Reference Data Monograph 2, 1994. Or, for a less detailed update, see: Atkinson, R. and Arey, J. "Gas-phase tropospheric chemistry of biogenic volatile organic compounds: a review" Atmospheric environment; 37(2, suppl.): S197-S219, 2003. Houghton, J. Global Warming: The Complete Briefing. Sander, S. P., Chemical Kinetics and Photochemical Data for Use in Stratospheric Modeling, #14, JPL Publication 02-25, 2002. http://jpldataeval.jpl.nasa.gov/download.html Other References Science, Vol. 276, 16 May 1997. Special issue on Tropospheric Processes. Faraday Discussions #100, 1995. Faraday Discussions #130, 2005. Atmospheric Sciences: Entering the twenty-first century (National Research Council, 1998. http://fermat.nap.edu/catalog/6021.html Decade-to-Century-Scale Climate Variability and Change: a Science Strategy. National Research Council, 1998. http://fermat.nap.edu/catalog/6129.html Rethinking the Ozone Problem in Urban and Regional Air Pollution. National Research Council, 1991. http://fermat.nap.edu/catalog/1889.html Section B page 1 of 2 Atkinson, R. Kinetics and Mechanisms of the Gas Phase Reactions of Hydroxyl Radical with Organic Compounds, J. Phys. Chem. Reference Data Monograph 1, 1989. Barker, J. R., Ed. Progress and Problems in Atmospheric Chemistry, 1995. Chameides, W. L. Biogeochemical Cycles Hansen, L. D. and Eatough, D. J. Organic Chemistry of the Atmosphere Hester, R. E., Air Pollution and Health Lave, L. B. Air Pollution and Health Newman, L. Measurement Challenges in Atmospheric Chemistry. Okabe, H. Photochemistry of Small Molecules, 1978. Selected Data Sources on the Web http://www.esf.edu/chemistry/dibble/AtmosChemCalc.htm http://www.esf.edu/chemistry/dibble/links.htm#Atmospheric Ozone maps http://exp-studies.tor.ec.gc.ca/e/ozone/Curr_allmap_g.htm http://toms.gsfc.nasa.gov/ozone/ozone.html http://lap.physics.auth.gr/ozonemaps2/ Kinetics http://kinetics.nist.gov/index.php http://www.iupac-kinetic.ch.cam.ac.uk/ NIST Database IUPAC Evaluations Documents by W. P. L. Carter on smog chemistry http://pah.cert.ucr.edu/~carter/bycarter.htm C. SELECTED PAGES FROM CHEMICAL KINETICS AND PHOTOCHEMICAL DATA FOR USE IN STRATOSPHERIC MODELING. #14. JPL PUBLICATION 02-25, 2002. Table of Contents Example of Table 1 - Bimolecular Rate Constants Example of Table 2 - Termolecular Rate Constants Thermodynamic Tables TABLE OF CONTENTS INTRODUCTION viii I.1 Basis of the Recommendations...................................................................................................................ix I.2 Scope of the Evaluation..............................................................................................................................ix I.3 Format of the Evaluation .............................................................................................................................x I.4 Computer Access.........................................................................................................................................x I.5 Data Formats ...............................................................................................................................................x I.6 Units ............................................................................................................................................................x I.7 Noteworthy Changes in This Evaluation .....................................................................................................x I.8 Acknowledgements ................................................................................................................................. xiii I.9 References ............................................................................................................................................... xiii SECTION 1. BIMOLECULAR REACTIONS 1-1 1.1 Introduction ............................................................................................................................................. 1-1 1.2 Uncertainty Estimates.............................................................................................................................. 1-2 1.3 Notes to Table 1..................................................................................................................................... 1-31 1.4 References ............................................................................................................................................. 1-93 SECTION 2. TERMOLECULAR REACTIONS 2-1 2.1 Introduction................................................................................................................................................2-1 2.2 Low–Pressure-Limiting Rate Constant, k ox ( T ) ..................................................................................... 2-1 2.3 2.4 2.5 2.6 2.7 2.8 Temperature Dependence of Low–Pressure Limiting Rate Constants: Tn ............................................... 2-2 High-Pressure-Limit Rate Constants, k∞(T) ............................................................................................ 2-2 Temperature Dependence of High-Pressure-Limiting Rate Constants: Tm .............................................. 2-3 Uncertainty Estimates.............................................................................................................................. 2-3 Notes to Table 2....................................................................................................................................... 2-8 References ...............................................................................................................................................2-16 SECTION 3. EQUILIBRIUM CONSTANTS 3-1 3.1 Format ..................................................................................................................................................... 3-1 3.2 Definitions ............................................................................................................................................... 3-1 3.3 Notes to Table 3....................................................................................................................................... 3-3 3.4 References ............................................................................................................................................... 3-5 SECTION 4. PHOTOCHEMICAL DATA 4-1 4.1 Format and Error Estimates ..................................................................................................................... 4-3 4.2 Halocarbon Absorption Cross Sections and Quantum Yields ................................................................. 4-3 4.3 References ........................................................................................................................................... 4-102 SECTION 5. HETEROGENEOUS CHEMISTRY ................................................................................................... 5-1 5.1 Introduction ............................................................................................................................................. 5-1 5.2 Surface Types—Acid/Water, Liquids, and Solids................................................................................... 5-2 5.3 Surface Types—Soot and Alumina ......................................................................................................... 5-2 5.4 Surface Composition and Morphology.................................................................................................... 5-3 5.5 Surface Porosity....................................................................................................................................... 5-4 5.6 Temperature Dependences of Parameters................................................................................................ 5-4 5.7 Solubility Limitations .............................................................................................................................. 5-4 5.8 Data Organization.................................................................................................................................... 5-4 5.9 Parameter Definitions .............................................................................................................................. 5-5 5.10 Mass Accommodation Coefficients for Surfaces Other Than Soot ......................................................... 5-8 5.11 Notes to Table 5-1 ................................................................................................................................... 5-9 5.12 Gas/Surface Reaction Probabilities for Surfaces Other Than Soot........................................................ 5-16 5.13 Notes to Table 5-2 ................................................................................................................................. 5-19 iv 5.14 5.15 5.16 5.17 5.18 5.19 5.20 Soot Surface Uptake Coefficients.......................................................................................................... 5-32 Notes to Table 5-3 ................................................................................................................................. 5-32 Henry’s Law Constants for Pure Water................................................................................................. 5-35 Notes to Table 5-4 ................................................................................................................................. 5-36 Henry’s Law Constants for Acids.......................................................................................................... 5-40 Notes to Table 5-5 ................................................................................................................................. 5-40 References ............................................................................................................................................. 5-44 APPENDIX A. THERMODYNAMIC PARAMETERS ..........................................................................................A-1 A.1 Gas-phase entropy and enthalpy values...................................................................................................A-1 A.1 References ...............................................................................................................................................A-8 TABLES Table I-1. Editions of this Publication......................................................................................................................... viii Table I-2. Panel Members and their Major Responsibilities for the Current Evaluation............................................. viii Table 1-1. Rate Constants for Second-Order Reactions...............................................................................................1-5 Table 2–1. Rate Constants for Termolecular Reactions...............................................................................................2-4 Table 3-1. Equilibrium Constants.............................................................................................................................. 3-2 Table 4-1. Photochemical Reactions ......................................................................................................................... 4-4 Table 4-2. Combined Uncertainties for Cross Sections and Quantum Yields........................................................... 4-6 Table 4-3. Absorption Cross Sections of O2 Between 205 and 240 nm .................................................................... 4-7 Table 4-4. Absorption Cross Sections of O3 at 273 K ............................................................................................... 4-8 Table 4-5. Parameters for the Calculation of O(1D) Quantum Yields ....................................................................... 4-9 Table 4-6. Absorption Cross Sections of HO2 ......................................................................................................... 4-10 Table 4-7. Absorption Cross Sections of H2O Vapor .............................................................................................. 4-11 Table 4-8. Absorption Cross Sections of H2O2 Vapor............................................................................................. 4-11 Table 4-9. Mathematical Expression for Absorption Cross Sections of H2O2 as a Function of Temperature......... 4-12 Table 4-10. Absorption Cross Sections of NO2 ....................................................................................................... 4-13 Table 4-11. Quantum Yields for NO2 Photolysis .................................................................................................... 4-14 Table 4-12. Absorption Cross Sections of NO3 at 298 K ........................................................................................ 4-16 Table 4-13. Mathematical Expression for Absorption Cross Sections of N2O as a Function of Temperature* ....... 4-16 Table 4-14. Absorption Cross Sections of N2O at 298 K ........................................................................................ 4-17 Table 4-15. Absorption Cross Sections of N2O5...................................................................................................... 4-18 Table 4-16. Absorption Cross Sections of HONO .................................................................................................. 4-19 Table 4-17. Absorption Cross Sections and Temperature Coefficients of HNO3 Vapor......................................... 4-20 Table 4-18. Absorption Cross Sections of HO2NO2 Vapor ..................................................................................... 4-20 Table 4-19. Absorption Cross Sections and Quantum Yields for Photolysis of CH2O ........................................... 4-21 Table 4-20. Absorption Cross Sections of CH3O2, C2H5O2, and CH3C(O)O2 ......................................................... 4-22 Table 4-21. Absorption Cross Sections of CH3OOH............................................................................................... 4-23 Table 4-22. Absorption Cross Sections of PAN ...................................................................................................... 4-25 Table 4-23. Absorption Cross Sections of FNO ...................................................................................................... 4-26 Table 4-24. Absorption Cross Sections of CCl2O, CClFO, and CF2O at 298 K...................................................... 4-27 Table 4-25. Absorption Cross Sections of Cl2 ......................................................................................................... 4-28 Table 4-26. Absorption Cross Sections of ClOO .................................................................................................... 4-29 Table 4-27. Absorption Cross Sections of OClO at the Band Peaks ....................................................................... 4-30 Table 4-28. Absorption Cross Sections of Cl2O ...................................................................................................... 4-32 Table 4-29. Absorption Cross Sections of ClOOCl at 200–250 K .......................................................................... 4-33 Table 4-30. Absorption Cross Sections of Cl2O3 ..................................................................................................... 4-34 Table 4-31. Absorption Cross Sections of Cl2O4 ..................................................................................................... 4-34 Table 4-32. Absorption Cross Sections of Cl2O6 ..................................................................................................... 4-34 v Table 1 - page 8 A-Factora E/R k(298 K)a O2(1Σ) + O3 → products 2.2×10–11 0 2.2×10–11 O2(1Σ) + H2O → products – – O2(1Σ) + N → products – O2(1Σ) + N2 → products O2(1Σ) + CO2 → products g Notes 1.2 200 A63 5.4×10–12 1.3 – A64 – <10–13 – – A65 2.1×10–15 0 2.1×10–15 1.2 200 A66 4.2×10–13 0 4.2×10–13 1.2 200 A67 O + OH → O2 + H 2.2×10–11 –120 3.3×10–11 1.2 100 B1 O + HO2 → OH + O2 3.0×10–11 –200 5.9×10–11 1.1 50 B2 O + H2O2 → OH + HO2 1.4×10–12 2000 1.7×10–15 2.0 1000 B3 → HO2 H + O2 M (See Table 2-1) H + O3 → OH + O2 1.4×10–10 470 2.9×10–11 1.25 200 B4 H + HO2 → products 8.1×10–11 0 8.1×10–11 1.3 100 B5 OH + O3 → HO2 + O2 1.7×10–12 940 7.3×10–14 1.2 80 B6 OH + H2 → H2O+ H 5.5×10–12 2000 6.7×10–15 1.1 100 B7 OH + HD → products 5.0×10–12 2130 4.0×10–15 1.2 200 B8 OH + OH → H2O + O 4.2×10–12 240 1.9×10–12 1.4 240 B9 Reaction f(298 K)b HO× Reactions M → H2O2 (See Table 2-1) OH + HO2 → H2O + O2 4.8×10–11 –250 1.1×10–10 1.3 100 B10 OH + H2O2 → H2O+ HO2 2.9×10–12 160 1.7×10–12 1.15 50 B11 HO2 + O3 → OH + 2O2 1.0×10–14 490 1.9×10–15 1.15 +160 –80 B12 HO2 + HO2 → H2O2 + O2 2.3×10–13 –600 1.7×10–12 1.3 200 B13 1.7×10–33[M] –1000 4.9×10–32[M] 1.3 400 B13 –180 1.0×10–11 1.1 50 C1 M → H2O2 + O2 NO× Reactions M → NO2 O + NO (See Table 2-1) O + NO2 → NO + O2 5.6×10–12 1-8 Table 2–1. Rate Constants for Termolecular Reactions Low-Pressure Limita ko(T) = ko300 (T/300)–n n ko300 Reaction High-Pressure Limitb k∞(T) = k∞300 (T/300)–m m k∞300 f g Notes 1.1 50 A1 Ox Reactions O + O2 M (6.0) (–34) 2.4 – – (3.5±3.0) (–37) 2.0 0.6±0.6 – – A2 → HO2 (5.7±0.5) (–32) 1.6±0.5 (7.5±4.0) (–11) 0±1.0 B1 M (6.9) (–31) 1.0 (2.6) (–11) 0 (9.0±2.0) (–31) 1.5±0.3 (3.0±1.0) (–11) 0±1.0 (2.5) (–31) 1.8 (2.2) (–11) 0.7 (7.0±1.0) (–31) 2.6±0.3 (3.6±1.0) (–11) 0.1±0.5 (2.0) (–30) 3.0 (2.5) (–11) 0 M (1.8±0.3) (–31) 3.2±0.4 (4.7±1.0) (–12) 1.4±1.4 M (2.0) (–30) 4.4 (1.4) (–12) 0.7 → O3 O(1D) Reactions M → N2 O O(1D) + N2 HOx Reactions H + O2 M → H2 O 2 OH + OH 1.5 100 B2 NOx Reactions O + NO M → NO2 M O + NO2 → NO3 OH + NO → HONO M M → HONO2 (See Note) OH + NO2 HO2 + NO2 → HO2NO2 NO2 + NO3 → N2 O 5 NO3 M → NO + O2 C1 1.3 100 C2 C3 1.3 100 C4 C5 1.2 100 See Note C6 C7 Hydrocarbon Reactions CH3 + O2 M (4.5±1.5) (–31) 3.0±1.0 (1.8±0.2) (–12) 1.7±1.7 D1 M (1.5±1.0) (–28) 3.0±1.0 (8.0±1.0) (–12) 0±1.0 D2 M (5.5±2.0) (–30) 0.0±0.2 (8.3±1.0) (–13) 2 –2±1 D3 M (1.0±0.6) (–28) 0.8±2.0 (8.8±0.9) (–12) 0 0±2 D4 (1.4±0.5) (–29) 3.8±1.0 (3.6±1.6) (–11) 0.6±1.0 D5 M (5.3) (–29) 4.4 (1.9) (–11) 1.8 M (2.8±1.0) (–27) 4.0±2.0 (5.0±1.0) (–11) 1.0±1.0 → CH3O2 C2 H5 + O 2 → C2 H5 O 2 OH + C2H2 → HOCHCH OH + C2H4 → HOCH2CH2 CH3O + NO → CH3ONO M CH3O + NO2 → CH3ONO2 C2H5O + NO → C2H5ONO 2-4 1.1 0 D6 D7 APPENDIX A. THERMODYNAMIC PARAMETERS Table of Contents APPENDIX A. THERMODYNAMIC PARAMETERS ..........................................................................................A-1 A.1 Gas-phase entropy and enthalpy values..................................................................................................A-1 A.1 References ..............................................................................................................................................A-8 Tables Table A-1. Gas-phase entropy and enthalpy values for selected species at 298.15 K and 100 kPa. .........................A-1 A.1 Gas-phase entropy and enthalpy values Table A-1 lists selected entropy and enthalpy of formation values at 298 K for a number of atmospheric species. As much as possible, the values were taken from primary evaluations, that is, evaluations that develop a recommended value from the original studies. Otherwise, the values were selected from the original literature, which is referenced in the table. Often, the enthalpy of formation and the entropy values are taken from different sources, usually due to a more recent value for the enthalpy of formation. The cited error limits are from the original references and therefore reflect often widely varying criteria. Some enthalpy values were corrected slightly to reflect the value of a reference compound selected for this table; these are indicated. Values that are calculated or estimated are also indicated in the table. Table A-1. Gas-phase entropy and enthalpy values for selected species at 298.15 K and 100 kPa. SPECIES H H2 O(3P) O(1D) O2 O2(1∆g) O2(1Σg+) O3 OH HO2 H2O H2O2 N(4S) N2 NH NH2 NH3 NH2OH NH2NO2 NO N2O NO2 NO3 N2O3 N2O4 N2O5 HNO ∆Hf(298 K) kJ mol–1 217.998±0.006 0.00 249.18±0.10 438.05±0.1 0.00 94.29±0.01 156.96±0.01 141.8±2 37.20±0.38 13.8±3.3 –241.826±0.040 –135.88±0.22 472.68±0.40 0.00 357±1 186±1 –45.94±0.35 –40.2±9.2 –26±10 91.29±0.17 81.6±0.5 34.19±0.5 73.7±1.4 86.6±1 11.1±1 13.3±1.5 107.1±2.5 ∆Hf(298 K) kcal mol–1 52.103±.001 0.00 59.56±0.02 104.70±0.03 0.00 22.54±0.01 37.51±0.01 33.9±0.5 8.89±0.09 3.3±0.8 –57.798±0.010 –32.48±0.05 112.973±0.10 0.00 85.3±0.3 44.5±0.3 –10.98±0.08 –9.6±2.2 –6.2±3 21.82±0.04 19.50±0.12 8.17±0.1 17.6±0.3 20.7±0.3 2.65±0.25 3.18±0.36 25.6±0.6 A-1 S(298 K) J K–1 mol–1 114.717±0.002 130.680±0.003 161.059±0.003 S(298 K) cal K–1 mol–1 27.418±.0.001 31.233±0.001 38.194±0.001 205.152±0.005 49.033±0.001 239.01 183.74 229.1 188.835±0.010 234.52 153.301±0.003 191.609±0.004 181.25±0.04 194.71±0.05 192.77±0.05 236.18 268.54 210.76 220.01 240.17 258.4±1.0 314.74 340.45 355.7±7 57.12 43.91 54.76 45.133±.002 56.05 36.640±0.001 45.796±0.001 43.32±0.01 46.54±0.01 46.07±0.01 56.45 64.18 50.37 52.58 57.40 61.76±0.24 75.22 81.37 85.01±2 Referencea, b, c [28] [28] [28] [70] [28] [36] [36] [33] [33,87] [35,58] [28] [33] [28] [28] [4] [4] [28] [5] [33] [5,22] [33] [33] [1,29] [33] [33] [33] [5] SPECIES HONO HONO2 HO2NO HO2NO2 C CH CH2(3B1) CH2(1A1) CH3 CH4 CN HCN C2N2 CH2NH2 CH3NH2 CH2NO NH2CO NCO HNCO CO CO2 HCO CH2O HCOO C(O)OH HC(O)OH CH3O CH3O2 CH2OH CH3OH CH3OOH CH2NO2 CH3NO2 CH3ONO CH3ONO2 C2H C2H2 C2H2OH C2H3 C2H4 C2H5 C2H6 CH2CN CH3CN CH2CO CH3CO CH2CHO CH3CHO CH3CH2O (CHO)2 ∆Hf(298 K) kJ mol–1 –78.45±0.8 –133.9±0.6 –23.8 –53.1±2.5 716.68±0.45 597.37±1.3 390.4±0.8 428.0±0.8 146.65±0.29 –74.48±0.41 440±5 132±4 309.1±0.8 149±8 –23.4±1.0 157±4 –15.1±4 151±14 –104±12 –110.53±0.17 –393.51±0.13 44.15±0.43 –108.7±0.05 127 –193 –378.8±0.5 17.15±3.8 9.0±5.1 –11.5±1.3 –201.0±0.6 –139.0±8.1 147.3 –74.3±0.6 –64.0 –122.2±4.3 565.3±2.9 227.4±0.8 121±11 299±5 52.4±0.5 120.9±1.7 –83.85±0.29 252.6±4 74.04±0.37 –49.58±0.88 –10.0±1.2 10.5±9.2 –166.1±0.5 –15.5±3.3 –212±0.8 ∆Hf(298 K) kcal mol–1 –18.75±0.2 –32.0±0.1 –5.7 –12.7±0.6 171.29±0.11 142.77±0.3 93.31±0.2 102.3±0.2 35.05±0.07 –17.80±0.10 105±1 31.5±1 73.9±0.2 35.6±2 –5.6±0.3 37.5±1 –3.6±1 36±3 – 24.8±2.8 –26.42±0.04 –94.05±0.03 10.55±0.10 –25.98±0.01 30 –45 –90.54±0.1 4.1±0.9 2.15±1.2 –2.75±0.31 –48.04±0.14 –33.2±1.9 35.2 –17.8±0.2 –15.3 –29.2±1.1 135.1±0.7 54.35±0.2 28.9±2.6 71.5±.1 12.52±0.12 28.9±0.4 –20.04±0.07 60.4±1.0 17.70±0.09 –11.85±0.21 –2.4±0.3 2.5±2.2 39.7±0.1 –3.7±0.8 –50.7±0.2 A-2 S(298 K) J K–1 mol–1 254.07 266.88 274 294±3 158.100±0.001 183.04 194.90 S(298 K) cal K–1 mol–1 60.72 63.78 65.6 70.3±0.7 37.787±0.001 43.75 46.58 193.96 186.38 202.64 201.82 242.20 46.36 44.55 48.43 48.24 57.89 242.89 58.05 232.38 237.97±0.8 197.660±0.004 213.785±0.010 224.34 218.76 244.7 251.6 248.87 232.86 55.54 56.9±0.2 47.242±0.001 51.096±0.002 53.62 52.28 58.5 60.1 59.48 55.655 244.170±0.018 239.865 58.358±0.004 57.329 272.48 275.2 284.3 301.9 209.73 200.93 65.12 65.8 67.95 72.15 50.13 48.02 219.316 250.52 229.162 52.418 59.88 54.771 245.12±0.8 58.59±0.2 263.95 63.09 Referencea, b, c [33] [33] [61], calc. [84] [28] [33] [88] [39] [33,88] [31,82] [33] [33] [33] [62], corr. [31,79] [96], calc. [96], calc. [75], corr., [33] [97], corr.,[102] [28] [28] [8], corr., [33] [33] [106], calc. [106] [33,106] [11,33] [46] [41] [33] [46] [31] [31,79] [98] [79,98] [11,33] [33] [32] [99] [33] [11,33] [33,82] [52] [2,102] [88] [11] [11] [31,79] [11] [30] SPECIES C2H5O C2H5O2 C2H5OOH CH2CH2OH CH3CHOH C2H5OH CH3COO CH2C(O)OH CH3C(O)O CH3C(O)OH CH3C(O)O2 CH3C(O)O2NO2 HOCH2COOH CH3OCH2 CH3OCH3 CH2(OH)CH2OH CH3OOCH3 (HOCO)2 C3H5 C3H6 n–C3H7 i–C3H7 i–C3H7O2 C3H8 C2H5CHO CH3COCH3 n-C4H10 (CH3COO)2 F F2 HF HOF FO FOF OFO FOO FOOF FONO FNO FNO2 FONO2 CF CHF CF2 CF3 CF4 CHF3 CHF2 CH2F2 CH2F ∆Hf(298 K) kJ mol–1 –17.2 –27.4±9.9 –175.4±12.9 –31±7 –63.7±4 –234.8±0.5 –190 –243 –192.5 –432.8±0.5 –154.4 –240.1 –583±10 –13.0±4 –184.1±0.5 –392.2±4.0 –125.5±5.0 –731.8±2.0 166.1±4.3 20.0±0.7 100±2 86.6±2.0 –65.4±11.3 –104.68±0.50 –185.6±0.8 –217.1±0.7 -125.65±0.67 –500±10 79.38±0.30 0.00 –273.30±0.70 –98.3±4.2 109±10 24.5±2 380±20 25.4±2 19.2±2.0 67 –65.7 –79 10±2 244.1±10 143.1±12 –184±8 –465.7±2.1 –933.20±0.75 –692.9±2.1 –239±4 –452.7±0.8 –32±8 ∆Hf(298 K) kcal mol–1 –4.1 –6.6±2.4 –41.9±3.1 –7.5±1.7 –15.2±1 –56.12±0.12 –45 58 –46.0 –103.4±0.1 –36.9 –57.4 –139±3 –3.1±1 –44.0±0.1 93.7±1.0 –30.0±1.2 –174.9±0.5 39.7±1.0 4.78±0.2 24±0.5 20.7±0.5 –15.6±2.7 –25.02±0.12 44.4±0.2 51.9±0.2 -30.03±0.16 –120±3 18.94±0.07 0.00 –65.32±0.17 –23.5±1.0 26±3 5.86±0.5 90.8±5 6.07±0.5 4.59±0.5 16 –15.70 –19.0 2.5±0.5 58.3±2.4 34.2±3.0 –44.0±2 –111.3±0.5 –223.04±0.18 –165.6±0.5 –57.1±1.0 –108.2±0.2 –7.6±2 A-3 S(298 K) J K–1 mol–1 S(298 K) cal K–1 mol–1 281.622 284.9 238.4 67.309 68.1 57.0 332.67 79.51 318.6±5.0 76.1±1.2 267.34 303.81 63.90 72.61 320.6±5.0 248±15 266.6 76.6±1.2 59.3±3.6 63.72 281±5 67.2±1.2 270.20 304.51 295.46 309.91 390.7±6.0 158.751±0.004 202.791±0.005 173.799±0.003 226.77±0.21 216.40±0.3 247.46±0.4 251±1 259.5±0.2 277.2±0.2 64.58 70.62 74.07 93.4±1.4 37.942±0.001 48.468±0.001 41.539±0.001 54.20±0.05 51.72±0.07 59.14±0.1 60.0±0.3 62.02±0.05 66.25±0.05 248.0 277.1 290 213.03±0.04 234.87 240.83±0.04 264.56 261.454 259.67 258.50 246.59 236.52 59.27 66.24 70 50.92±0.01 56.14 57.56±0.01 63.23 62.49 62.06 61.78 58.94 56.53 Referencea, b, c [62] [46] [46] [32] [62] [33] [106], calc. [106] [63], calc. [18,79] [63], calc. [63], calc. [30] [62], corr. [31,79] [31,79] [30] [30] [94] [17,79] [99] [95] [46] [16,82] [31,79] [31,79] [31,82] [30] [28] [28] [28] [22] [21] [21] [21], calc. [21] [21] [6], est [98] [98] [22], est. [22,33] [33,83] [22,83] [33,89] [28] [33,89] [81] [85] [81] SPECIES CH3F FCO CHFO CF2O CF3O CF2O2 CF3O2 CF3OH CF3OOCF3 CF3OF CH2CH2F CH3CHF CH3CH2F CH2FCH2F CH2FCHF CH2FCHF2 CHF2CHF2 CH2CF3 CH3CF3 CHF2CH2 CH3CF2 CH3CHF2 CHFCF3 CH2FCF3 CF2CF3 CHF2CF3 C2F6 Cl Cl2 HCl ClO ClOO OClO ClO3 ClClO ClOCl ClOOCl ClClO2 ClOClO Cl2O3 HOCl ClNO ClNO2 cis–ClONO trans–ClONO ClO2NO ClONO2 FCl CHCl CCl2 ∆Hf(298 K) kJ mol–1 –238±8 –161.2±8.1 –383±7 –607.9±7.1 –624±8 –427±6 -612.5±15.4 –911±8 –1434±11 –724±8 59.4±8 –70.3±8 –277.4±4.2 –432±25 235.5 –665±4 –860±24 –517.1±5 –745.6±1.7 –277 –302.5±8.4 –500.1±6.3 –697 –896±8 –891±5 –1105±5 –1344.3±3.4 121.301±0.008 0.00 –92.31±0.10 101.63v0.1 98.0v4 94.6v1.2 194±12 90±30 81.3±1.8 127.6±2.9 154.2 175.5 150±6 –74.8±1.2 52.7±0.5 12.5±1.0 –64.4±6.3 75.3±6.3 102 22.9±2.0 –55.70±0.31 326±8 230±8 ∆Hf(298 K) kcal mol–1 –56.8±2 –38.5±2.0 –91.6±1.7 –145.3±1.7 –149±2 –102±1.5 146±4 –218±2 –343±3 –173±2 –14.2±2 –16.8±2 –66.3±1 –103.2±6 56.28 –158.9±1 –205.6±5.7 –123.6±1.2 –178.2±0.4 –66.3 –72.3±2 –119.7±1.5 –166.5 –214.1±2 –213±1.3 –264±1.1 –321.3±0.8 28.992±0.002 0.00 –22.06±0.02 24.29±0.03 23.4±1 22.6±0.3 46±3 22±7 19.4±0.4 30.5±0.7 36.9 41.9 35.8±1.5 –17.9±0.3 12.6±0.1 3.0±0.3 15.4±1.5 18.0±1.5 24.3 5.5±0.5 13.31±0.07 78.0±2.0 55.0±2.0 A-4 S(298 K) J K–1 mol–1 222.78 S(298 K) cal K–1 mol–1 53.246 246.82 258.97 58.99 61.89 279.7 274.0 265.1 66.86 65.48 63.4 293.3 70.11 320.3 306.8 287.3 297.8 290.3 282.4 326.2 316.2 76.6 73.32 68.67 71.17 69.39 67.50 77.97 75.58 333.7 331.8 165.190±0.004 223.081±0.010 186.902±0.005 225.07±0.5 269.32±0.5 256.84±0.1 270.75±0.5 278.8±2.0 79.76 79.30 39.481±0.001 53.318±0.002 44.671±0.001 53.79±0.12 64.37±0.1 61.39±0.03 64.71±0.1 66.6±0.5 301.0±5.0 294±2 309±2 390±20 236.50±0.42 261.58 272.23 71.9±1.2 70.3±0.5 73.9±0.5 94±5 56.52±0.10 62.52 65.06 316 302.38 217.94 234.88 265.03 75.5 72.27 52.09 56.85 63.34 Referencea, b, c [85], H est. [44] [91],calc., [33] [91],calc., [33] [91], calc. [48], calc. [56] [91] [91] [91] [66],[25], calc. [66], [26], calc. [59], est. [33] [42] [27] [51], corr. [64], corr. [31] [25,104] [23] [25], calc. [80], [26], S calc [23] [27], H corr. [23], H est. [105] [23] [23,89] [28] [28] [28] [22] [22] [22,72] [22] [22] [34] [22,72] [55],calc., [22] [55],calc., [22] [14] [22,34] [33] [33] [54], calc. [54], calc. [61], calc. [3] [33] [33,83] [33,83] SPECIES CCl3 CCl3OH CCl3O CCl3O2 CCl4 CHCl3 CHCl2 CHCl2O2 CH2Cl CH2ClO2 CH2Cl2 CH3Cl ClCO CHClO CCl2O CHFCl CH2FCl CFCl CFCl2 CFCl3 CF2Cl2 CF3Cl CHFCl2 CHF2Cl CF2Cl CFClO CH2ClCOOH C2H3Cl CH3CHFCl CH2CF2Cl CH3CF2Cl C2Cl4 1,1–C2H2Cl2 Z–1,2–C2H2Cl2 E–1,2–C2H2Cl2 C2HCl3 CH2CCl3 1,1,1–C2H3Cl3 1,1,2–C2H3Cl3 1,1,1,2–C2H2Cl4 1,1,2,2–C2H2Cl4 C2HCl5 CH3CCl2 CH3CCl2O2 CH3CHCl2 CH2CH2Cl CH3CHCl CH3CH2Cl C2Cl6 Br ∆Hf(298 K) kJ mol–1 71.1±2.5 –293±20 –43.5±20 –20.9±8.9 –95.6±2.5 –102.9±2.5 89.0±3.0 –17±7 117.3±3.1 –4v11 –95.1±2.5 –81.9±0.6 –24.9±4.2 –164±20 –220.9 –61±10 –264±8 31v13 –89.1±10.0 –285.3 –494.1 –709.2±2.9 –285±9 –484.8 –279±8 –429±20 –427.6±1.0 22±3 –313.4±2.6 –318 –536.2±5.2 –18.8±4 2.4±2.0 –3±2 –0.5±2.0 –19.1±3.0 71.5±8 –144.6±2.0 –148.0±4.0 –152.3±2.4 –156.7±3.5 –155.9±4.3 42.5±1.7 –69.7±4 –130.6±3.0 93.0±2.4 76.5±1.6 –112.1±0.7 –142±4 111.870±12 ∆Hf(298 K) kcal mol–1 17.0±0.6 –70.0±5 –10.4±5 –5.0±2.1 –22.8±0.6 –24.6±0.6 21.3±0.7 –4!2 28.0±0.7 –1±3 –22.8±0.6 –19.6±0.2 –5.9±1.0 –38±5 –52.8 –14.5±2.4 –63.2±2 7.4±3.2 –21.3±2.4 –68.2 –118.1 –169.5±0.7 –68.1±2.1 –115.6 –66.7±2 –103±5 –102.2±0.2 5.3±0.7 –74.9±0.6 –75.9 –128.2±1.2 –4.5±1 0.6±0.5 –0.7±0.5 –0.1±0.5 –.6±0.7 17.1±2 –34.6±0.5 –35.4±0.9 –36.4±0.6 –37.5±0.8 –37.3±1.0 10.2±0.4 –16.7±1 –31.2±0.7 22.2±0.6 18.2±0.4 –26.8±0.2 –34.0±1 26.74±0.03 A-5 S(298 K) J K–1 mol–1 303.24 S(298 K) cal K–1 mol–1 72.47 309.90 295.51 280±7 74.069 70.63 66.9±2 271±7 64.5±2 270.31 227.15 266.0 259.07 283.8 64.606 54.290 63.6 61.92 67.82 264.3 259.032 63.17 61.91 309.9 300.7 285.2 293.0 280.8 74.06 71.87 68.16 70.04 67.11 276.70 325.9±5.0 66.13 77.9±1.2 322.08 307.1 341.03 76.98 73.41 81.51 325.20 77.72 320.03 76.488 288±5 68.8±1.1 305.05 271±7 279±6 275.78 398.62 175.018±0.004 72.908 64.8±2 66.7±1.4 65.913 95.27 41.830±0.001 Referencea, b, c [37] [90], calc. [90], calc [46] [38,85][60] [60,85] [92] [92] [92] [92] [60,85] [60,85] [22,57] [33], H est, [98] [100] [24,100], H est. [33,83] [100] [24], corr. [24], corr. [24,89] [24], H est. [24], H est. [68] [33] [30] [60] [47] [77] [47,77] [33,38] [60] [60] [60] [31,78] [86] [15,47,60] [60] [60] [60] [60] [92] [45], corr. [15,47] [93] [92] [15,60] [33,38] [28] Br2(g) ∆Hf(298 K) kJ mol–1 30.91±0.11 ∆Hf(298 K) kcal mol–1 7.39±0.03 HBr Br2O HOBr BrO –36.29±0.16 106.2±2.5 –60.5±1.1 126.2±1.7 –8.67±0.04 25.4±0.6 –14.5±0.3 30.2±0.4 OBrO 163.9±4.4 BrOO SPECIES S(298 K) J K–1 mol–1 S(298 K) cal K–1 mol–1 Referencea, b, c 245.468±0.005 58.668±0.001 [28] 198.700±0.004 47.490±0.001 232.97±0.1 55.681±0.023 [28] [34] [34] [19,103] 39.2±1.1 271±2 64.8±0.5 [19,43], est. 108±40 26±10 289±3 69.1±0.7 [19] BrO3 221±50 53±12 285±2 68.1±0.5 [19], est. BrOBr 107.6±3.5 25.7±0.8 290.8±2 69.50±0.48 BrBrO 168±20 40±5 313±2 74.8±0.5 [19], est. 273.66±0.8 65.41±0.2 228.985 240.046 54.729 57.372 330.67 79.03 334.57 294 245.85±0.25 80.0 70.23 58.76±0.06 287.3±0.4 68.66±0.09 358.06 337.0±5.0 180.787±0.004 85.6 80.5±1.2 43.209±0.001 [102] [53], calc. [53], calc. [53], calc. [76] [33] [33] [100] [13]calc.,[33] [100] [33] [13], calc. [49] [10] [67] corr. [47,50] [69] [47] [89] [13,33] [30] [28] BrNO Z–BrONO E–BrONO BrNO2 BrONO2 BrF BrCl CH2Br CHBr3 CHBr2 CBr3 CH2Br2 CH3Br CH2CH2Br CH3CHBr CH3CH2Br CH3CBr2 CH3CBr2H CF3Br CBr4 CH2BrCOOH I [19] 82.17±0.8 71.9 88.3 45.2 42.3±6.3 –58.9±1.0 14.79±0.16 169±4 23.8±4.5 188.9 235±25 –11.1±5.0 –37.7±1.5 135.6±6.7 127±4 –61.5±1.0 140.2±5.4 26.7±1.9 –641.4±2.3 83.9±3.4 –383.5±3.1 106.76±0.04 19.64±0.2 17.19 21.1 10.8 10.1±1.5 –14.08±0.3 3.53±0.04 40.4±1.0 5.7±1.1 45.0±2.2 56±6 –2.7±1.2 –9.02±0.36 32.4±1.6 30.4±1 –14.7±0.3 33.5±1.3 6.4±0.5 –153.3±0.5 20.0±0.8 –91.7±0.7 25.52±0.01 I2 62.42±0.08 14.92±0.02 260.687±0.005 62.306±0.001 [28] HI 26.50±0.10 6.33±0.03 206.590±0.004 49.376±0.001 [28] HOI –69.6±5.4 –16.6±1.3 255.0±0.1 60.95±0.03 [12,34] IO OIO IOO IO3 IOI IIO IOOI IIOO IOIO OIIO INO 115.9±5.0 77±15 96.6±15 242±50 92.4±15 134.1±15 156.8±15 103.0±15 124.2±15 224.0±15 121±4 27.7±1.2 18±4 23±4 58±12 22.1±4 32.1±4 37.5±4 24.6v4 29.7±4 53.5±4 29.0±1 239.6±0.1 279.9 308.4 293±4 306.5 317.8 337.0 339.9 349.7 356.3 282.8±4 57.27±0.03 66.9 73.7 70.0±1.0 73.3 76.0 80.5 81.2 83.6 85.2 67.6±1 [9,34] [65], calc. [65], calc. [20], est. [65] [65] [65], calc. [65], calc. [65], calc. [65], calc. [101] A-6 SPECIES INO2 ICl IBr CH3I CH2I2 CF3I CH3CH2I S ∆Hf(298 K) kJ mol–1 60.2±4 17.506±0.105 40.88±0.08 13.76±0.12 118.4±0.1 –586.2±2.1 –7.5±0.9 277.17±0.15 ∆Hf(298 K) kcal mol–1 14.4±1 4.184±0.025 9.77±0.02 3.29±0.03 28.30±0.03 –140.1±0.5 –1.79±0.2 66.25±0.04 S(298 K) J K–1 mol–1 S(298 K) cal K–1 mol–1 Referencea, b, c 294±6 427.567 258.95 253.70±0.25 70.3±1.5 102.191 61.89 60.635±0.06 [101] [22] [22] [49] 309.41±1.34 307.78 295.52±0.42 73.95±0.32 73.56 70.63±0.10 [49] [33,89] [47,50] 167.829±0.006 40.112±0.002 [28] [28] S2 128.6±0.3 30.74±.07 228.167±0.010 54.533±0.003 HS 195.55 46.74 [74], corr., [33] 142.80±2.85 34.13±0.68 [28] H2S –20.6±0.5 –4.92±0.12 205.81±0.05 49.19±0.01 SO 221.94 53.04 [33] 4.78±0.25 1.14±0.06 [28] SO2 –296.81±0.20 –70.94±0.05 248.223±0.050 59.327±0.012 SO3 256.541 61.315 [33] –395.9±0.7 –94.62±0.17 HSO [7] –6.1±2.9 –1.5±0.7 H2SO4 299.282 71.530 [33] –733±2 –175.2±0.5 CS 210.55 50.32 [33] 279.775±0.75 66.87±0.18 CS2 237.882 56.855 [33] 116.7±1.0 27.9±0.2 [71] CS2OH 110.5±4.6 26.4±1.1 321±20 77±5 CH3S [73], corr. 125.0±1.8 29.87±0.44 CH3SH 255.14 60.98 [31,79] –22.9±0.7 –5.47±0.17 CH2SCH3 [40] 136.8±5.9 32.7±1.4 CH3SCH3 285.96 68.35 [31,79] –37.4±0.6 –8.94±0.2 CH3SSCH3 336.80 80.50 [31,79] –24.7±1.0 –5.9±0.3 OCS 231.644 55.36 [33] –141.7±2 –33.9±0.5 Notes: a. Error limits are estimates from the original references. b. If two references are given for a substance, the first refers to the enthalpy value while the second to the entropy. c. The terms “calc” and “est” indicate that the value is calculated or estimated. The term “corr” indicates that an enthalpy value has been adjusted to reflect the value chosen in this table for a reference substance. A-7 References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. Abramowitz, S. and M. W. Chase, 1991, Pure App. Chem., 63, 1449-1454. An, X. W. and M. Mansson, 1983, J. Chem. Thermo., 15, 287-293. Anderson, L. C. and D. W. Fahey, 1990, J. Phys. Chem., 94, 644-652. Anderson, W. R., 1989, J. Phys. Chem., 93, 530-536. Anderson, W. R., 1999, Comb. Flame, 117, 394-403. Atkinson, R., D. L. Baulch, R. A. Cox, R. F. Hampson, J. A. Kerr, M. J. Rossi and J. Troe, 1997, J. Phys. Chem. Ref. Data, 26, 521-1011. Balucani, N., P. Casavecchia, D. Stranges and G. G. Volpi, 1993, Chem. Phys. Lett., 211, 469472. Becerra, R., I. W. Carpenter and R. Walsh, 1997, J. Phys. Chem., 101, 4185-4190. Bedjanian, Y., G. Le Bras and G. Poulet, 1997, J. Phys. Chem. A, 101, 4088-4096. Bedjanian, Y., G. Poulet and G. Le Bras, 1999, J. Phys. Chem. A, 103, 4026-4033. Berkowitz, J., G. B. Ellison and D. Gutman, 1994, J. Phys. Chem., 98, 2744-2765. Berry, R., J. Yuan, A. Misra and P. Marshall, 1998, J. Phys. Chem. A, 102, 5182-5188. Bickerton, J., M. E. M. da Piedade and G. Pilcher, 1984, J. Chem. Thermo., 16, 661-668. Burkholder, J. B., R. K. Talukdar, A. R. Ravishankara and S. Solomon, 1993, J. Geophys. Res., 98, 22937-22948. Chao, J., A. S. Rodgers, R. C. Wilhoit and B. J. Zwolinski, 1974, J. Phys. Chem. Ref. Data, 3, 141-162. Chao, J., R. C. Wilhoit and B. J. Zwolinski, 1973, J. Phys. Chem. Ref. Data, 2, 427-437. Chao, J. and B. J. Zwolinski, 1975, J. Phys. Chem. Ref. Data, 4, 251-261. Chao, J. and B. J. Zwolinski, 1978, J. Phys. Chem. Ref. Data, 7, 363-377. Chase, M. W., 1996, J. Phys. Chem. Ref. Data, 25, 1069-1111. Chase, M. W., 1996, J. Phys. Chem. Ref. Data, 25, 1297-1340. Chase, M. W., 1996, J. Phys. Chem. Ref. Data, 25, 551-603. Chase, M. W., 1998, J. Phys. Chem. Ref. Data, Monograph 9. Chen, S. S., A. S. Rodgers, J. Chao, R. C. Wilhoit and B. J. Zwolinski, 1975, J. Phys. Chem. Ref. Data, 4, 441-456. Chen, S. S., R. C. Wilhoit and B. J. Zwolinski, 1976, J. Phys. Chem. Ref. Data, 5, 571-580. Chen, Y., A. Rauk and E. Tschuikow-Roux, 1990, J. Chem. Phys., 93, 6620-6629. Chen, Y., A. Rauk and E. Tschuikow-Roux, 1990, J. Chem. Phys., 93, 1187-1195. Chen, Y., A. Rauk and E. Tschuikow-Roux, 1991, J. Chem. Phys., 94, 7299-7310. Cox, J. D., D. D. Wagman and V. A. Medvedev CODATA Key Values for Thermodynamics; Hemisphere Publishing Corp.: New York, 1989. Davis, H. F., B. Kim, H. S. Johnston and Y. T. Lee, 1993, J. Phys. Chem., 97, 2172-2180. Dorofeeva, O., V. P. Novikov and D. B. Neumann, 2001, J. Phys. Chem. Ref. Data, 30, 475-513. Frenkel, M., G. J. Kabo, K. N. Marsh, G. N. Roganov and R. C. Wilhoit Thermodynamics of organic compounds in the gas state; Thermodynamics Research Center: College Station, TX, 1994; Vol. I. Fulle, D., H. F. Hamann, H. Hippler and C. P. Jänsch, 1997, Ber. Bunsenges. Phys. Chem., 101, 1433-1442. Gurvich, L. V., I. V. Veyts and C. B. Alcock Thermodynamic Properties of Individual Substances,, Fourth ed.; Hemisphere Publishing Corp.: New York, 1991; Vol. 2. Hassanzadeh, P. and K. K. Irikura, 1997, J. Phys. Chem. A, 101, 1580-1587. Hills, A. J. and C. J. Howard, 1984, J. Chem. Phys., 81, 4458-4465. Constants of Diatomic Molecules; Huber, K. P. and G. Herzberg, Eds.; National Institute of Standards and Technology, 1998. Hudgens, J. W., R. D. Johnson, R. S. Timonen, J. A. Seetula and D. Gutman, 1991, J. Phys. Chem., 95, 4400-4405. Huybrechts, G., M. Marmon and B. Van Mele, 1996, Int. J. Chem. Kinet., 28, 27-36. Jacox, M. E. Vibrational and Electronic Energy Levels of Polyatomic Transient Molecules; National Institute of Standards and Technology, 1998. Jefferson, A., J. M. Nicovich and P. H. Wine, 1994, J. Phys. Chem., 98, 7128-7135. A-8 41. 42. 43. 44. 45. 46. 47. 48. 49. 50. 51. 52. 53. 54. 55. 56. 57. 58. 59. 60. 61. 62. 63. 64. 65. 66. 67. 68. 69. 70. 71. 72. 73. 74. 75. 76. 77. 78. 79. 80. 81. 82. 83. 84. 85. 86. 87. Johnson, R. D. and J. W. Hudgens, 1996, J. Phys. Chem., 100, 19874-19890. Kerr, J. A. and D. M. Timlin, 1971, Int. J. Chem. Kinet., 3, 427-441. Klemm, R. B., R. P. Thorn, L. J. Stief, T. J. Buckley and R. D. Johnson, 2001, J. Phys. Chem. A, 105, 1638-1642. Knyazev, V. D., A. Bencsura and I. R. Slagle, 1997, J. Phys. Chem. A, 101, 849-852. Knyazev, V. D., A. Bencsura and I. R. Slagle, 1998, J. Phys. Chem. A, 102, 1760-1769. Knyazev, V. D. and I. R. Slagle, 1998, J. Phys. Chem. A, 102, 1770-1778. Kolesov, V. P. and T. S. Papina, 1983, Russ. Chem. Rev., 52, 425-439. Kraka, E., Z. Konkoli, D. Cremer, J. Fowler and H. F. Schaefer, 1996, J. Amer. Chem. Soc., 118, 10595-10608. Kudchadker, S. A. and A. P. Kudchadker, 1975, J. Chem. Phys. Ref. Data, 4, 457-470. Kudchadker, S. A. and A. P. Kudchadker, 1979, J. Phys. Chem. Ref. Data, 8, 519-526. Lacher, J. R. and H. A. Skinner, 1968, J. Chem. Soc. A, 1034-1038. Lafleur, R. D., B. Szatary and T. Baer, 2000, J. Phys. Chem. A, 104, 1450-1455. Lee, J. H., R. B. Timmons and L. J. Stief, 1976, J. Chem. Phys., 64, 300-305. Lee, T. J., 1994, J. Phys. Chem., 98, 111-115. Li, W. K. and C. Y. Ng, 1997, J. Phys. Chem. A, 101, 113-115. Lightfoot, P. D., R. A. Cox, J. N. Crowley, M. Destriau, G. D. Hayman, M. E. Jenkin, G. K. Moortgat and F. Zabel, 1992, Atmos. Environ., 26A, 1805-1961. Lim, K. P. and J. V. Michael, 1994, J. Phys. Chem., 98, 211-215. Litorja, M. and B. Ruscic, 1998, J. Electron.Spec. Rel. Phenom., 97, 131-146. Luo, Y. R. and S. W. Benson, 1997, J. Phys. Chem. A, 101, 3042-3044. Manion, J. A., 2002, J. Phys. Chem. Ref. Data, 31, 123-172. McGrath, M. P. and F. S. Rowland, 1994, J. Phys. Chem., 98, 1060-1067. McMillen, D. F. and D. M. Golden, 1982, Ann. Rev. Phys. Chem., 33, 493-532. Miller, C. E., J. I. Lynton, D. M. Keevil and J. S. Franscisco, 1999, J. Phys. Chem. A, 103, 1145111459. Millward, G. E., R. Hartig and E. Tschuikow-Roux, 1971, J. Phys. Chem., 75, 3195-3201. Misra, A. and P. Marshall, 1998, J. Phys. Chem. A, 102, 9056-9060. Miyokawa, K., S. Ozaki and T. Yano, 1996, Bull. Chem. Soc. Jpn., 69, 869-873. Miyokawa, K. and E. Tschuikow-Roux, 1990, J. Phys. Chem., 94, 715-717. Miyokawa, K. and E. Tschuikow-Roux, 1992, J. Phys. Chem., 96, 7328-7331. Miyokawa, K. and E. Tschuikow-Roux, 1999, Bull. Chem. Soc. Jpn., 72, 1-5. Moore, C. E. Atomic Energy Levels; NSRDS: Washington. DC, 1971; Vol. 1. Murrells, T. P., E. R. Lovejoy and A. R. Ravishankara, 1990, J. Phys. Chem., 94, 2381-2386. Nickolaisen, S. L., R. R. Friedl and S. P. Sander, 1994, J. Phys. Chem., 98, 155-169. Nicovich, J. M., K. D. Kreutter, C. A. van Dijk and P. H. Wine, 1992, J. Phys. Chem., 96, 25182528. Nicovich, J. M., K. D. Kreutter, C. A. van Dijk and P. H. Wine, 1992, J. Phys. Chem., 96, 25182528. Okabe, H., 1970, J. Chem. Phys., 53, 3507-3515. Orlando, J. J. and G. S. Tyndall, 1996, J. Phys. Chem., 100, 19398-19405. Paddison, S. J., Y. H. Chen and E. Tschuikow-Roux, 1994, Can. J. Chem., 72, 561-567. Papina, T. S. and V. P. Kolesov, 1985, Russ. J. Phys. Chem., 59, 1289-1292. Pedley, J. B. Thermochemicsl Data and Structures of Organic Compounds; Thermodynamics Data Center: College Station, TX, 1994. Pickard, J. M. and A. S. Rodgers, 1977, J. Amer. Chem. Soc., 99, 691-694. Pickard, J. M. and A. S. Rodgers, 1983, Int. J. Chem. Kinet., 15, 569-577. Pittam, D. A. and G. Pilcher, 1972, J. Chem. Soc. Farad. Trans 1, 68, 2224-2229. Poutsma, J. C., J. A. Paulino and R. R. Squires, 1997, J. Phys. Chem. A, 101, 5327-5336. Regimbal, J. M. and M. Mozurkewich, 1997, J. Phys. Chem. A, 101, 8822-8820. Rodgers, A. S., J. Chao, R. C. Wilhoit and B. J. Zwolinski, 1974, J. Phys. Chem. Ref. Data, 3, 117-140. Rodgers, A. S. and P. Jerus, 1988, Int. J. Chem. Kinet., 20, 565-575. Ruscic, B., D. Feller, D. A. Dixon, K. A. Peterson, L. B. Harding, R. L. Asher and A. F. Wagner, 2001, J. Phys. Chem A, 105, 1-4. A-9 88. 89. 90. 91. 92. 93. 94. 95. 96. 97. 98. 99. 100. 101. 102. 103. 104. 105. 106. Ruscic, B., M. Litorja and R. L. Asher, 1999, J. Phys. Chem. A, 103, 8625-8633. Ruscic, B., J. V. Michael, P. C. Redfern, L. A. Curtiss and K. Raghavachri, 1998, J. Phys. Chem. A, 102, 10889-10899. Schneider, W. F., B. I. Nance and T. J. Wallington, 1995, J. Amer. Chem. Soc., 117, 478-485. Schneider, W. F. and T. J. Wallington, 1994, J. Phys. Chem., 98, 7448-7451. Seetula, J. A., 1996, J. Chem. Soc. Faraday Trans., 92, 3069-3078. Seetula, J. A., 1998, J. Chem. Soc. Faraday Trans., 94, 891-898. Seetula, J. A., 1999, Phys. Chem. Chem. Phys., 1, 4727-4731. Seetula, J. A. and I. R. Slagel, 1997, J. Chem. Soc. Faraday Trans., 93, 1709-1719. Shapley, W. A. and G. B. Bacskay, 1999, J. Phys. Chem. A, 103, 4505-4513. Spiglanin, T. A., R. A. Pery and D. W. Chandler, 1986, J. Phys. Chem., 90, 6184-6189. Stull, D. R., E. F. Westrum and G. C. Sinke The Chemical Thermodynamics of Organic Compounds; John Wiley & Sons: New York, 1969. Tsang, W. Heats of formation of organic free radicals by kinetic methods. In Energetics of Free Radicals; Simoes, J. A. M., Greenberg, A., Liebman, J. F., Eds.; Blackie Academic & Professional: London, 1996; pp 22-58. Tschuikow-Roux, E. and S. Paddison, 1987, Int. J. Chem. Kinet., 19, 15-24. van den Bergh, H. and J. Troe, 1976, J. Chem. Phys., 64, 736-742. Wagman, D. D., W. H. Evans, V. B. Parker, R. H. Schumm, I. Halow, S. M. Bailey, K. L. Churney and R. L. Nutall, 1982, J. Phys. Chem. Ref. Data, 11, 392 pp. Wilmouth, D. M., T. F. Hanisco, N. M. Donahue and J. G. Anderson, 1999, J. Phys. Chem A, 103, 8935-8945. Wu, E. C. and A. S. Rodgers, 1974, J. Phys. Chem., 78, 2315-2317. Wu, E. C. and A. S. Rodgers, 1976, J. Amer. Chem. Soc., 98, 6112-6115. Yu, D., A. Rauk and D. A. Armstrong, 1994, J. Chem. Soc. Perkin Trans 2, 2207-2215. A-10 D. DATA USEFUL IN TEACHING KINETICS CALCULATIONS FOR STRATOSPHERIC CHEMISTRY Extracted from: Kinetics and Photochemical Data for Use in Stratospheric Modeling. #12 JPL Publication 97-4 2002. Solar Fluxes Temperature and Pressure Profiles Concentration Profiles Photolysis Rate Constants APPENDIX 3: SOLAR FLUXES AND SPECIES PROFILES Figures 6 and 7 show data for solar irradiances and fluxes. These were provided by Kenneth Minschwaner. The solar irradiances are from measurements by the Solar Ultraviolet Spectral Irradiance Monitor (SUSIM) for λ ≤ 400 nm (VanHoosier et al. [6]), and by Neckel and Labs [5] for 400 < λ ≤ 600 nm. The SUSIM measurements are spectrally degraded to 2 nm full width half-maximum to correspond to the resolution of the Neckel and Labs data. Additionally, a normalization factor that varies linearly from 1.17 at 400 nm to 1.0 at 440 nm has been applied to the Neckel and Labs irradiances in order to match SUSIM values at 400 nm. Irradiances from 110 to 120 nm are based on measurements by Mount and Rottman [4] and Woods and Rottman [8]. Values below 110 nm are not plotted. The solar fluxes are computed from the sum of the direct, attenuated solar beam plus angularly integrated scattered radiation. Fluxes at 0, 20, 30, 40, and 50 km are based on the solar irradiances, assuming a solar zenith angle of 30° and the U.S. Standard Atmosphere (1976). Molecular and aerosol scattering are taken into account; the latter process is appropriate for "moderate volcanic" conditions (Fenn et al. [2]). The surface albedo is 0.3. Ozone cross sections follow the recommendations herein; oxygen cross sections in the Herzberg continuum are taken from Yoshino et al. [9]; Schumann-Runge band absorption is determined using the high-resolution treatment of Minschwaner et al. [3], with fluxes spectrally degraded to 1.0 nm resolution. The species and "J" value profiles presented in Figures 8-16 were provided by Peter Connell. They were generated by the LLNL 2-D model of the troposphere and stratosphere. The temperature profile is an interpolation to climatological values. Surface source gas boundary conditions are those for the year 1990, as reported in chapter 6 of the WMO/UNEP report [7]. The equatorial tropopause source gas mixing ratios are: total chlorine 3.4 ppb, total fluorine 1.6 ppb, total bromine 18 ppt, methane 1.67 ppm, and nitrous oxide 309 ppb. The kinetic parameters used were consistent, to the extent possible, with the current recommendations. Representations of sulfate aerosol and polar stratospheric heterogeneous processes which were included are hydrolysis of nitrogen pentoxide and chlorine and bromine nitrate and reaction of hydrogen chloride with chlorine nitrate and hypochlorous acid. The model run represents a periodic steady-state atmosphere with 1990 surface abundances of source gases. The "J" values were calculated with a clear sky, two-stream radiative transfer model with wavelength binning of 5 nm above 310 nm and 500 cm-1 below. Surface reflectance includes the effect of average cloudiness on the albedo. Oxygen cross sections in the Schumann-Runge region were calculated by the method of Allen and Frederick [1], corrected for the Herzberg continuum values of Yoshino et al. [9]. The fluxes and profiles are given to provide "order of magnitude" values of important photochemical parameters. They are not intended to be standards or recommended values. References 1. 2. 3. 4. 5. 6. 7. 8. 9. Allen, M. and J.E. Frederick, 1982, J. Atmos. Sci., 39, 2066-2075. Fenn, W.R., S.A. Clough, W.O. Gallery, R.E. Good, F.X. Kneizys, J.D. Mill, L.S. Rothman, E.P. Shettle, and F.E. Volz, 1985, "Optical and Infrared Properties of the Atmosphere," Handbook of Geophysics and the Space Environment, Chapter 18, A.S. Jursa, Editor, Air Force Geophysics Laboratory, Bedford, MA. Minschwaner, K., G.P. Anderson, L.A. Hall, and K. Yoshino, 1992, J. Geophys. Res., 97, 10103-10108. Mount, G.H. and G.J. Rottman, 1985, J. Geophys. Res., 90, 13031-13036. Neckel, H. and D. Labs, 1984, Solar Physics, 90, 205-258. VanHoosier, M.E., J.-D.F. Bartoe, G.E. Brueckner, and D.K. Prinz, 1988, Astro. Lett. and Communications, 27, 163-168. WMO, Scientific Assessment of Ozone Depletion: 1994, World Meteorological Organization Global Ozone Research and Monitoring Project, Report No. 37, 1994, Geneva: National Aeronautics and Space Administration. Woods, T.N. and G.J. Rottman, 1990, J. Geophys. Res., 95, 6227-6236. Yoshino, K., A.S.C. Cheung, J.R. Esmond, W.H. Parkinson, D.E. Freeman, S.L. Guberman, A. Jenouvrier, B. Coquart, and M.F. Merienne, 1988, Planet. Space Sci., 36, 1469-1475. 258 Figure 6. Solar Irradiance 259 Figure 7. Actinic Flux at Several Altitudes 260 Altitude (km) 10 30 20 100 10 200 220 240 Temperature (K) 260 280 1018 261 Figure 8. Temperature and Density 40 Total Density (molecules/cm3) Pressure (mb) 50 TEMPERATURE (March 15 - 40 N) 1 1017 OXYGEN and HYDROGEN SPECIES (March 15 - Local Noon - 40 N) 50 H*108 1017 40 Total Density Altitude (km) O(3P) O3 30 O(1D)*108 CH4 1018 20 H2O H2O2 10 106 108 1010 Number Density (molecules/cm3) 1012 1014 262 OH Figure 9. Number Densities of Oxygen and Hydrogen Species HO2 NITROGEN SPECIES (March 15 - Local Noon - 40 N) 50 NO Total Density Altitude (km) HO2NO2 30 NO2 HNO3 N2O 1018 NO3*106 20 ClONO2 N2O5 10 106 107 108 109 1010 Number Density (molecules/cm3) 1011 1012 263 1017 40 Figure 10. Number Densities of Nitrogen Species N(4S)*108 CHLORINE SPECIES (March 15 - Local Noon - 40 N) 50 HCl Total Density Altitude (km) ClONO2 30 Cl ClO 1018 20 10 104 105 106 107 108 3 Number Density (molecules/cm ) 109 264 1017 40 Figure 11. Number Densities of Chlorine Species HF OTHER SPECIES (March 15 - Local Noon - 40 N) Total Density Altitude (km) 30 1018 CFCl3 20 10 106 107 CF2Cl2 108 109 1010 Number Density (molecules/cm3) CO 1011 1012 265 1017 40 Figure 12. Number Densities of CFCl 3, CF2Cl2, and CO 50 SELECTED "J" VALUES (March 15 - Local Noon - 40 N) 50 H2O 20 10 10-14 10-13 10-12 10-11 10-10 Photolysis rate coefficient (sec-1) 10-9 10-8 266 O2 30 Figure 13. J-Values for O2 and H 2O Altitude (km) 40 SELECTED "J" VALUES (March 15 - Local Noon - 40 N) 50 CFCl3 CCl4 20 10 10-10 10-9 10-8 10-7 10-6 Photolysis rate coefficient (sec-1) 10-5 10-4 267 Altitude (km) N2O 30 Figure 14. Selected J-Values CF2Cl2 40 SELECTED "J" VALUES (March 15 - Local Noon - 40 N) 50 H2O2 Altitude (km) HOBr 30 HNO3 ClONO2 20 HO2NO2 10 10-7 10-6 N2O5 10-5 10-4 Photolysis rate coefficient (sec-1) HOCl 10-3 10-2 268 Figure 15. Selected J-Values 40 E. DATA ON CLIMATE CHANGE Extracted from: 2002 Summary Report of the Intergovernmental Panel on Climate Change Indicators of the human influence on the atmosphere during the Industrial Era Global Warming Potentials The global mean radiative forcing of the climate system Variations of the Earth's surface temperature Simulated annual global mean surface temperatures Indicators of the human influence on the atmosphere during the Industrial Era Figure 2: Long records of past changes in (a) Global atmospheric concentrations of three well mixed greenhouse gases (a) shows changes in the atmospheric CO2 (ppm) 360 the influence of anthropogenic emissions. concentrations of carbon dioxide (CO2), methane (CH4), and nitrous oxide (N2O) over the past 1000 1.5 Carbon dioxide atmospheric composition provide the context for years. The ice core and firn data for several sites in Antarctica and Greenland (shown by different 340 1.0 symbols) are supplemented with the data from direct 320 atmospheric samples over the past few decades 0.5 300 280 (shown by the line for CO2 and incorporated in the curve representing the global average of CH4). The 0.0 estimated positive radiative forcing of the climate 260 0.5 0.4 1500 0.3 1250 0.2 1000 0.1 750 N2O (ppb) 310 0.0 Radiative forcing (Wm−2) Methane 1750 CH4 (ppb) Atmospheric concentration system from these gases is indicated on the right- 0.15 Nitrous oxide hand scale. Since these gases have atmospheric lifetimes of a decade or more, they are well mixed, and their concentrations reflect emissions from sources throughout the globe. All three records show effects of the large and increasing growth in anthropogenic emissions during the Industrial Era. (b) illustrates the influence of industrial emissions on atmospheric sulphate concentrations, which produce negative radiative forcing. Shown is the time history of the concentrations of sulphate, not in the atmosphere but in ice cores in Greenland (shown by 0.10 lines; from which the episodic effects of volcanic 290 0.05 eruptions have been removed). Such data indicate the local deposition of sulphate aerosols at the site, 0.0 270 reflecting sulphur dioxide (SO2) emissions at mid-latitudes in the Northern Hemisphere. This 250 1000 record, albeit more regional than that of the 1200 1400 1600 1800 2000 globally-mixed greenhouse gases, demonstrates the Year large growth in anthropogenic SO2 emissions during the Industrial Era. The pluses denote the relevant 200 Sulphur 50 100 25 0 1600 0 1800 Year 2000 SO2 emissions (Millions of tonnes sulphur per year) Sulphate concentration 6 (mg SO42– per tonne of ice) (b) Sulphate aerosols deposited in Greenland ice regional estimated SO2 emissions (right-hand scale). [Based upon (a) Chapter 3, Figure 3.2b (CO2); Chapter 4, Figure 4.1a and b (CH4) and Chapter 4, Figure 4.2 (N2O) and (b) Chapter 5, Figure 5.4a] Table 3: Direct Global Warming Potentials (GWPs) relative to carbon dioxide (for gases for which the lifetimes have been adequately characterised). GWPs are an index for estimating relative global warming contribution due to atmospheric emission of a kg of a particular greenhouse gas compared to emission of a kg of carbon dioxide. GWPs calculated for different time horizons show the effects of atmospheric lifetimes of the different gases. [Based upon Table 6.7] Gas a b Lifetime (years) Global Warming Potential (Time Horizon in years) 20 yrs 100 yrs 1 1 62 23 275 296 500 yrs 1 7 156 Carbon dioxide Methanea Nitrous oxide CO2 CH4 N2 O 12.0b 114 b Hydrofluorocarbons HFC-23 HFC-32 HFC-41 CHF3 CH2F2 CH3F 260 5.0 2.6 9400 1800 330 12000 550 97 10000 170 30 HFC-125 HFC-134 HFC-134a HFC-143 HFC-143a HFC-152 HFC-152a HFC-161 CHF2CF3 CHF2CHF2 CH2FCF3 CHF2CH2F CF3CH3 CH2FCH2F CH3CHF2 CH3CH2F 29 9.6 13.8 3.4 52 0.5 1.4 0.3 5900 3200 3300 1100 5500 140 410 40 3400 1100 1300 330 4300 43 120 12 1100 330 400 100 1600 13 37 4 HFC-227ea HFC-236cb HFC-236ea HFC-236fa HFC-245ca HFC-245fa HFC-365mfc HFC-43-10mee CF3CHFCF3 CH2FCF2CF3 CHF2CHFCF3 CF3CH2CF3 CH2FCF2CHF2 CHF2CH2CF3 CF3CH2CF2CH3 CF3CHFCHFCF2CF3 33 13.2 10 220 5.9 7.2 9.9 15 5600 3300 3600 7500 2100 3000 2600 3700 3500 1300 1200 9400 640 950 890 1500 1100 390 390 7100 200 300 280 470 Fully fluorinated species SF6 CF4 C2F6 C3F8 C4F10 c-C4F8 C5F12 C6F14 3200 50000 10000 2600 2600 3200 4100 3200 15100 3900 8000 5900 5900 6800 6000 6100 22200 5700 11900 8600 8600 10000 8900 9000 32400 8900 18000 12400 12400 14500 13200 13200 Ethers and Halogenated Ethers CH3OCH3 0.015 1 1 <<1 HFE-125 HFE-134 HFE-143a CF3OCHF2 CHF2OCHF2 CH3OCF3 150 26.2 4.4 12900 10500 2500 14900 6100 750 9200 2000 230 HCFE-235da2 HFE-245fa2 HFE-254cb2 HFE-7100 HFE-7200 H-Galden 1040x HG-10 HG-01 CF3CHClOCHF2 CF3CH2OCHF2 CHF2CF2OCH3 C4F9OCH3 C4F9OC2H5 CHF2OCF2OC2F4OCHF2 CHF2OCF2OCHF2 CHF2OCF2CF2OCHF2 2.6 4.4 0.22 5.0 0.77 6.3 12.1 6.2 1100 1900 99 1300 190 5900 7500 4700 340 570 30 390 55 1800 2700 1500 110 180 9 120 17 560 850 450 The methane GWPs include an indirect contribution from stratospheric H2O and O3 production. The values for methane and nitrous oxide are adjustment times, which incorporate the indirect effects of emission of each gas on its own lifetime. 47 The global mean radiative forcing of the climate system for the year 2000, relative to 1750 2 Halocarbons N2O Aerosols Warming CH4 1 CO2 Tropospheric ozone Black carbon from fossil fuel burning Mineral Dust Aviation-induced Solar Contrails Cirrus 0 Cooling Radiative forcing (Watts per square metre) 3 Stratospheric ozone −1 Organic carbon Biomass burning Sulphate from fossil fuel burning Landuse (albedo) only Aerosol indirect effect −2 High Medium Medium Low Very Low Very Low Very Very Low Low Very Low Very Low Very Very Low Low Level of Scientific Understanding Figure 3: Many external factors force climate change. These radiative forcings arise from changes in the atmospheric composition, alteration of surface reflectance by land use, and variation in the output of the sun. Except for solar variation, some form of human activity is linked to each. The rectangular bars represent estimates of the contributions of these forcings − some of which yield warming, and some cooling. Forcing due to episodic volcanic events, which lead to a negative forcing lasting only for a few years, is not shown. The indirect effect of aerosols shown is their effect on the size and number of cloud droplets. A second indirect effect of aerosols on clouds, namely their effect on cloud lifetime, which would also lead to a negative forcing, is not shown. Effects of aviation on greenhouse gases are included in the individual bars. The vertical line about the rectangular bars indicates a range of estimates, guided by the spread in the published values of the forcings and physical understanding. Some of the forcings possess a much greater degree of certainty than others. A vertical line without a rectangular bar denotes a forcing for which no best estimate can be given owing to large uncertainties. The overall level of scientific understanding for each forcing varies considerably, as noted. Some of the radiative forcing agents are well mixed over the globe, such as CO2, thereby perturbing the global heat balance. Others represent perturbations with stronger regional signatures because of their spatial distribution, such as aerosols. For this and other reasons, a simple sum of the positive and negative bars cannot be expected to yield the net effect on the climate system. The simulations of this assessment report (for example, Figure 5) indicate that the estimated net effect of these perturbations is to have warmed the global climate since 1750. [Based upon Chapter 6, Figure 6.6] 8 Figure 1: Variations of the Earth’s Variations of the Earth's surface temperature for: surface temperature over the last 140 years and the last millennium. (a) the past 140 years (a) The Earth’s surface temperature is Departures in temperature (°C) from the 1961 to 1990 average 0.8 shown year by year (red bars) and GLOBAL approximately decade by decade (black line, a filtered annual curve suppressing 0.4 fluctuations below near decadal time-scales). There are uncertainties in the annual data (thin black whisker bars represent the 95% confidence 0.0 range) due to data gaps, random instrumental errors and uncertainties, uncertainties in bias corrections in the −0.4 ocean surface temperature data and also in adjustments for urbanisation over Data from thermometers. −0.8 1860 the land. Over both the last 140 years and 100 years, the best estimate is that 1880 1900 1920 1940 1960 1980 2000 the global average surface temperature has increased by 0.6 ± 0.2°C. Year (b) Additionally, the year by year (blue curve) and 50 year average (black (b) the past 1,000 years curve) variations of the average surface temperature of the Northern Hemisphere NORTHERN HEMISPHERE for the past 1000 years have been 0.5 reconstructed from “proxy” data Departures in temperature (°C) from the 1961 to 1990 average calibrated against thermometer data (see list of the main proxy data in the diagram). The 95% confidence range in the annual data is represented by the 0.0 grey region. These uncertainties increase in more distant times and are always much larger than in the instrumental record due to the use of relatively sparse −0.5 proxy data. Nevertheless the rate and duration of warming of the 20th century has been much greater than in any of the previous nine centuries. Similarly, it −1.0 is likely7 that the 1990s have been the Data from thermometers (red) and from tree rings, corals, ice cores and historical records (blue). warmest decade and 1998 the warmest year of the millennium. 1000 1200 1400 1600 Year 1800 2000 [Based upon (a) Chapter 2, Figure 2.7c and (b) Chapter 2, Figure 2.20] 3 (a) Natural (b) Anthropogenic 1.0 1.0 Temperature anomalies (°C) Temperature anomalies (°C) Simulated annual global mean surface temperatures model observations 0.5 0.0 −0.5 −1.0 1850 1900 1950 0.5 0.0 −0.5 −1.0 1850 2000 model observations 1900 Year 1950 2000 Year Temperature anomalies (°C) (c) All forcings 1.0 model observations 0.5 0.0 −0.5 −1.0 1850 1900 1950 2000 Year Figure 4: Simulating the Earth’s temperature variations, and comparing the results to measured changes, can provide insight into the underlying causes of the major changes. A climate model can be used to simulate the temperature changes that occur both from natural and anthropogenic causes. The simulations represented by the band in (a) were done with only natural forcings: solar variation and volcanic activity. Those encompassed by the band in (b) were done with anthropogenic forcings: greenhouse gases and an estimate of sulphate aerosols, and those encompassed by the band in (c) were done with both natural and anthropogenic forcings included. From (b), it can be seen that inclusion of anthropogenic forcings provides a plausible explanation for a substantial part of the observed temperature changes over the past century, but the best match with observations is obtained in (c) when both natural and anthropogenic factors are included. These results show that the forcings included are sufficient to explain the observed changes, but do not exclude the possibility that other forcings may also have contributed. The bands of model results presented here are for four runs from the same model. Similar results to those in (b) are obtained with other models with anthropogenic forcing. [Based upon Chapter 12, Figure 12.7] 11 F. COMPARING THE STRATOSPHERIC CHEMISTRY OF THE HALOGENS h 10 20 25 30 40 50 T 222 215 218 223 240 268 [M] 8.50E+18 2.00E+18 9.00E+17 3.00E+17 1.00E+17 1.70E+16 [O2] 1.8E+18 4.2E+17 1.9E+17 6.3E+16 2.1E+16 3.6E+15 [O3] 1.5E+12 7.5E+12 7.0E+12 5.0E+12 7.0E+11 8.0E+10 [CH4] 2.0E+13 4.0E+12 2.0E+12 7.0E+11 7.0E+10 8.0E+09 h 10 20 25 30 40 50 T 222 215 218 223 240 268 [M] 8.50E+18 2.00E+18 9.00E+17 3.00E+17 1.00E+17 1.70E+16 [O2] 1.8E+18 4.2E+17 1.9E+17 6.3E+16 2.1E+16 3.6E+15 [O3] 1.5E+12 7.5E+12 7.0E+12 5.0E+12 7.0E+11 8.0E+10 [CH4] 2.0E+13 4.0E+12 2.0E+12 7.0E+11 7.0E+10 8.0E+09 [H2O] 1.E+15 1.0E+13 6.0E+12 3.0E+12 8.0E+11 2.0E+11 Atomic Chlorine k'O2 k'O3 6.4E+04 1.3E+01 3.7E+03 6.5E+01 7.4E+02 6.1E+01 8.0E+01 4.5E+01 7.9E+00 6.9E+00 1.9E-01 8.8E-01 k'CH4 4.0E-01 6.5E-02 3.5E-02 1.4E-02 2.3E-03 4.7E-04 Atomic Chlorine %O2 %O3 100% 0.0% 98% 1.7% 92% 7.6% 64% 36.2% 54% 46.4% 18% 81.9% Atomic Fluorine k'O2 k'O3 9.6E+04 1.2E+01 5.5E+03 5.7E+01 1.1E+03 5.3E+01 1.2E+02 3.9E+01 1.2E+01 5.9E+00 3.1E-01 7.5E-01 k'CH4 9.9E+02 1.9E+02 9.7E+01 3.5E+01 3.8E+00 4.9E-01 k'H2O 1.4E+04 1.4E+02 8.4E+01 4.2E+01 1.1E+01 2.8E+00 %O2 86% 93% 82% 51% 37% 7% This makes use of the JPL Data Evaluation and the material in Section D of this coursepack. %CH4 0.001% 0.002% 0.004% 0.012% 0.015% 0.044% Atomic Fluorine %O3 %CH4 0.0% 0.895% 1.0% 3.237% 4.0% 7.252% 16.7% 14.863% 17.9% 11.496% 17.2% 11.186% %H2O 13% 2% 6% 18% 34% 65% G. HETEROGENEOUS CHEMICAL KINETICS The rate, Rcoll, of collision of a gas phase species, A, with the surface of aerosol particles is given by Rcoll = vave × [A] × Surface area per unit volume 4 ...where vave (meters/sec) is the average gas kinetic speed of species A: vave = 8 RT πM R is the gas constant (8.314 Joules mol-1 K-1) M is the molecular mass in kg/mole [A] is the concentration of A in molecules cm-3. Surface area per unit volume is usually expressed in µm2 cm-3 air. So watch the units conversions! The rate, Rrxn, of reaction of a gas phase species, A, aerosol particles = γ × Rcoll =γ× vave × [A] × Surface area per unit volume 4 ...where γ is the reactive uptake coefficient (see Section 5 of the JPL Data Evaluation). The value of γ varies between 0 and 1, and represents the fraction of collisions that result in reaction. γ may be a complex function of composition and temperature of the particle, and is generally time dependent. The lifetime, τ, of A with respect to heterogeneous reaction is γ× vave × Surface area per unit volume 4 H. CONCENTRATION DATA IN THE TROPOSPHERE On an Alabama pine plantation in 1990. Part of the "Rural Ozone in the Southern Environment" study. Atlantic Ocean, 1997. Data over the range from 1-11 kilometers in altitude, not just ground level. H. Singh, et al., J. Geophys. Res. 2000. Boulder, Co, 1991. [n-butane] = 0-2 ppbv [m+p-xylene]= 0-0.8 ppbv [NOy] = 1-100 ppbv P. D. Goldan, et al., J. Geophys. Res. 1995. I. LIFETIMES WITH RESPECT TO OH, O3, NO3, AND PHOTOLYSIS Reactant concentrations used for lifetime calculations Species [OH] [O3] [NO3] Concentration molecules cm-3 mole fraction 1.60E+06 7.00E+11 30 ppbv 4.80E+08 20 pptv Description global daytime average global 24-hour average global night-time average Room temperature lifetimes (in hours, except where specified) Species n-butane 1-butene trans-2-butene benzene toluene Formaldehyde 1-butanol Acetone 2-methyl-1,3-butadiene (isoprene) α-pinene naphthalene dibenzo-p-dioxin 1,2,3,4-tetrachloro-dibenzop-dioxin OH 68 hrs 5.6 2.7 145 29 18 20 789 1.7 O3 41 hrs 2 31 NO3 48 hrs 1.5 8510 0.9 3.2 6.9 17.4 217.0 4.6 0.1 * * Rate of loss = - kobs [naphthalene][NO3][NO2] kobs ~ 10-28 cm6 molecule-2 s-1 hν 4 hrs 38 days - J. DEGRADATION PATHWAYS OF ALKANES, ALKENES, OXYGENATES, AND AROMATICS Reference: Atkinson, R. Gas Phase Tropospheric Chemistry of Organic Compounds, J. Phys. Chem. Reference Data Monograph 2, 1994. + . OH . . CH2 CH 1 and 2-butyl radical O2 O2 OO. OO . NO NO ONO2 . O + NO + CH3CH2 . O2 ONO2 butyl nitrate O. HOO . NO2 O2 O + butoxy radical + 2 O2 O 1 and 2-butylperoxy radical H. .. .. O + ... O HOO. . CH OH 2 O2 Section J Degradation Pathways page 1 of 4 + . OH . Alkenes : OH mostly adds to an sp2 hybridized carbon in preference to H-atom abstraction. The subsequent chemistry is similar to that of the radicals derived from abstraction, except for the presence of the hydroxyl group. The preferred site of abstraction is the one leading to the more stable radicals: tertiary > secondary > primary. OH O2 OO. OH NO . O ONO2 + NO2 OH OH O + . OH O2 O Section J Degradation Pathways + HOO page 2 of 4 Alcohols: OH mainly abstracts from the substituted site:, and the subsequent reaction with O2 does not yield a peroxy radical. OH + . OH CHOH . CH . O O2 OH + HOO OH O2 OO . OO . . CH OH OH O2 Aldehydes The preferred site of abstraction is the aldehylic hydrogen. The reaction of the subsequent peroxy radical with NO2 yields relative stable products, unlike the situation with other peroxy radicals . CH3CH=O . CH3 + + CO2 + NO2 CH3C(=O) OH NO Compare CH3C(=O)OO. O2 CH3C(=O)OO. (peroxyacetylnitrate = PAN) CH3C(=O)OONO2 NO2 lifetime ~1 hour CH CH OO. 3 Section J Degradation Pathways 2 NO2 CH3CH2OONO2 lifetime ~1 second page 3 of 4 Aromatics Reaction with OH dominated by OH addition, but subsequent mechanism (and many of the final products) of this path are not known.. The products of the H-atom abstraction path are established. H OH . OH O2 H ? (and para and meta isomers) I have not drawn all the resonance structures OH . CH CH2OO . 2 O2 Section J Degradation Pathways CH2O NO . CH=O O2 + HOO K. TROPOSPHERIC PHOTOCHEMISTRY HONO + hν → OH + NO (Major OH source in the a.m.) HONO2 + hν → OH + NO2 (very slow in comparison to deposition) N2O5 + hν → NO2 + NO3 (slow with respect to thermal decomposition) RONO2 + hν → RO + NO2 (τ = days to weeks, somewhat slower than RONO2 + OH) HOOH + hν → 2 OH (τ ~ ________ ) NO3 + hν → NO2 + O or NO + O2 (absorbs strongly in the red, τ = seconds) H2C=O + hν → H + HC=O (45%) (τ = hours) → H2 + CO (55%) RCH=O + hν → R + HC=O (τ = days) RC(=O)OONO2 + hν → RC(=O)OO + NO2 (slower than thermal dissociation) RC(=O)O + NO3