University of California, Irvine – Institutional Review Board
advertisement

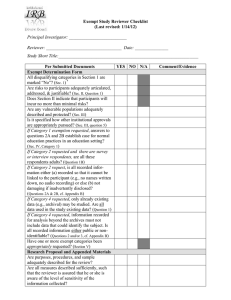
New Exempt Study Review - Internal Use Only University of California, Irvine – Institutional Review Board REVIEWER’S CHECKLIST – New Study Exempt Research Protocol HS#: eAPP#: Lead Researcher: Title: HRP ADMINISTRATIVE CHECKIST YES Required Signatures Received Consent Document(s) Received YES COMMENTS NO NO Waiting for Signatures ___ Consent(s) ___ Assent(s) ___ Study Info Sheet(s) Informed Consent Not Required – No Subject Contact Documented Informed Consent Not Required Appendix G: Use of Deception Appendix B: Pregnant Women / Neonates Appendix D: Children Appendix E: Cognitively Impaired / Medically Incapacitated YES NO YES NO YES NO YES NO Human Subjects section of Federal Grant Application/Proposal Received YES NO Permission Letters / Off-Site Research Agreement Received YES NO YES NO YES NO YES NO CTPRMC Approval/Exemption hSCRO Approval YES NO Appendix M: Storage of Data/Specimens for Future Research Appendix N: Genetic Testing Special Population(s) Identified Recruitment Material Received Data Collection Instrument Received Source of Funding Identified Specify: If Federally funded, a copy of the "Human Subjects” section of funding proposal is required Specify: PHI Accessed, Created or Disclosed Referred to COIOC Other Ancillary Committee Clearances Received All Appendices Received Appendix A: Non-UCI Site Appendix H: International Research Appendix I: Field Work Appendix T: Total Waiver of HIPAA Authorization Specify: HRP ADMINISTRATIVE COMMENTS **If you have any questions or would like assistance with this review, please feel free to contact me at (949) 824-XXXX or at XXXX@uci.edu. Thanks – XXXX v. 2013 HS# 1 of 4 New Exempt Study Review - Internal Use Only ADMINISTRATIVE QUESTIONS AND NOTES FOR THE IRB Notes for the IRB: Questions for the IRB: 1. Question X? YES NO (Please comment as necessary): 2. Appendix T: A partial waiver of HIPAA for recruitment purposes is requested. Please review Appendix T. In order to waive HIPAA Authorization, the IRB must determine that the study meets all of the following criteria: The use or disclosure of PHI involves no more than minimal risk Granting of the waiver will not adversely affect privacy rights and welfare of the individuals whose records will be used The project could not practicably be conducted without a waiver The project could not practicably be conducted without use of PHI The privacy risks are reasonable relative to the anticipated benefits of research An adequate plan to protect identifiers from improper use and disclosure is included in the research proposal An adequate plan to destroy the identifiers at the earliest opportunity, or justification for retaining identifiers, is included in the research proposal The project plan includes written assurances that PHI will not be re-used or disclosed for other purposes Whenever appropriate, the subjects will be provided with additional pertinent information after participation Is the request for a partial waiver acceptable? YES NO (Please comment as necessary): ADMINISTRATIVE COMMENTS FOR THE LEAD RESEARCHER (LR) UCI IRB REVIEWER’S CHECKLIST REVIEWERS: v. 2013 HS# 2 of 4 New Exempt Study Review - Internal Use Only A. Please specify whether you have a conflict of interest that would require you to be recused for the review of this protocol. If you do have a conflict, please contact HRP staff ASAP to arrange for reassigning this protocol. I DO NOT HAVE A CONFLICT OF INTEREST ON THIS PROTOCOL I DO HAVE A CONFLICT OF INTEREST ON THIS PROTOCOL B. Please review and specify if the research meets UCI’s Ethical Criteria outlined below by checking the corresponding box. Please document each concern you would like to be communicated to the LR in the corresponding comments box or in the open space below. CRITERIA FOR EXEMPT REGISTRATION BACKGROUND AND RESEARCH DESIGN RISK/BENEFIT ANALYSIS Statement of purpose/hypothesis is adequate Study personnel appear appropriate/qualified YES Risks are relatively non-existent Potential direct benefit to subjects or societal benefit included Acceptable risk/benefit relationship YES NO Comments: NO Comments: SUBJECT RECRUITMENT SUBJECT PROTECTION Selection of subjects is appropriate (inclusion/exclusion criteria) Selection of subjects is equitable Recruitment procedures are proper (undue influence or coercion is minimized, compensation is not coercive, recruitment materials are appropriate) YES NO Unanticipated Problem reporting is adequately addressed Provisions to protect subject privacy are adequate Provisions to maintain confidentiality are appropriate Addt’l Protections for Vulnerable Populations are addressed YES N/A Comments: NO Comments: INFORMED CONSENT Is an informed consent process appropriate? YES* NO, provide a rationale below N/A *If an informed consent process is appropriate, the researcher will disclose: That the activity involves research A description of the procedures That participation is voluntary There are adequate provisions to maintain privacy and confidentiality The name and contact information for the researcher YES NO Rationale/Comments: C. Risk Assessment: Virtually no risk [Exempt Registration] v. 2013 HS# 3 of 4 New Exempt Study Review - Internal Use Only If Virtually No Risk, indicate all corresponding Category(ies): No more than Minimal Risk [Expedited review] If Minimal Risk, indicate all corresponding Category(ies): Greater than Minimal Risk [Full Committee review] Please provide a rationale for any change in the risk assessment (e.g., from Exempt to Expedited or Full Committee). D. IRB Recommendation: E. Exempt Registration Confirmed Revisions/Clarifications Required Requires Expedited Review Requires Full Committee Review Reviewer Comments: Reviewer’s Signature Date Note: The information provided on this form may be preliminary and may not necessarily reflect the discussion and final decision and/or recommendation of the Committee. v. 2013 HS# 4 of 4