GEOS 470R/570R Volcanology L04, 26 January 2015 Handing out --Sojourner Truth
advertisement

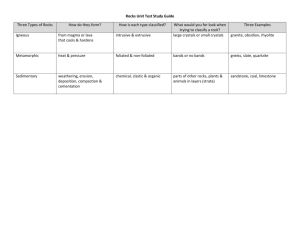
GEOS 470R/570R Volcanology L04, 26 January 2015 Handing out Today’s PowerPoint slides “It is the mind that rules the body.” --Sojourner Truth More on careers and geology “At the age of twenty, with a great deal of luck, I stumbled upon the profession of geology, and for nearly fifty years I have courted the earth, roaming its deserts and forests, its mountains and volcanoes. “I was often alone with the elements, extracting stories from the rocks, making new discoveries and building on those of my contemporaries and predecessors. “It has been the most enjoyable of occupations, solving unsolved mysteries of the earth and thinking of new ideas—the lifeblood of science.” --Richard V. Fisher, 1999, Out of the Crater: Chronicles of a volcanologist Study this diagram frequently for the lab, first field trip, and exams McPhie et al., 1993, Fig. 33 Superstition Mountains Readings from textbook For L04 from Lockwood and Hazlett (2010) Volcanoes—Global Perspectives Chapters 2 and 3 For L05 from Lockwood and Hazlett (2010) Volcanoes—Global Perspectives Chapters 6 and 9 Assigned reading For L04 today, 26 January 2015 Hildreth, W., 1981, Gradients in silicic magma chambers: Implications for lithospheric magmatism: Journal of Geophysical Research, v. 86, p. 10,15310,192. For L05, 30 January 2015 Metz, J. M., and Mahood, G. A., 1985, Precursors to the Bishop Tuff eruption: Glass Mountain, Long Valley, California: Journal of Geophysical Research, v. 90, p. 11,121-11,126. Last time Volatiles Undersaturation vs. saturation Solubility (saturation limit) Solubility controls H2O CO2 S Cl F Differences in proportions of volatiles as a function of magma composition Basaltic CO2 H2O Rhyolitic H2O SO2 F Cl Schmincke, 2004, Fig. 4.18 CO2 SO2 Solubility Maximum amount of a species or component that can be dissolved under a given set of conditions P T° X (melt composition) If melt contains less than maximum amount possible at given conditions, then the melt is undersaturated with respect to that volatile component Magmas are not necessarily volatile-saturated Dissolution of H2O forming OHgroups Dissolved oxygen forming hydroxyl ions Breaking O-Si-O polymers Reducing degree of polymerization of melt Predicted effect on viscosity? Best and Christiansen, 2001, Fig. 4.8 Concentration of water as a function of magma composition Schmincke, 2004, Table 4.1, after Fisher and Schmincke, 1984 Solubility of CO2 Bulk melt compositional effect for solution mechanism CO2 dissolves in rhyolitic melt as CO2 molecules CO2 dissolves in basaltic melt as a dissolved carbonate ion Intermediate compositions have both species Wallace and Anderson, 2000, Fig. 2 Sulfur solubility S more soluble at high oxygen fugacities Where dissolved sulfur occurs as sulfate Anhydrite present Solubility of sulfur increases with increasing temperature Under both oxidizing and reducing conditions Important temperature effect What is relationship to composition of magmas? Wallace and Anderson, 2000, Fig. 5 Summary: Volatiles Volatile: Element or compound that forms a gas at low P and T° Typically a multicomponent system in nature (mixed volatiles) Conditions: Undersaturated vs. saturated Solubility: Saturation limit Solution mechanisms differ widely; can be a function of Amount of volatile dissolved (H2O) Bulk silicate melt composition (CO2, S, Cl, F) Oxidation state / oxygen fugacity (S) Solubility of pure volatiles is a function of many factors Pressure (H2O) Bulk silicate melt composition, including other volatiles (H2O, CO2, S) Oxidation state / oxygen fugacity (S) Presence of separate sulfide phase (S) Lecture 04: Petrologic overview Chemical characterization and classification of volcanic rocks Individual rocks Normative mineralogy Silica content Silica saturation Alumina saturation Rock suites Volcanic rocks as samples of magma chambers Physical sorting in pyroclastic eruptions Types of zonation Compositional gaps Enrichment factors Silica content Ultramafic IUGS divisions commonly followed for ultramafic to andesite No agreement on terms for silicic rocks <45 wt% SiO2 Basalt 45 – 52% Basaltic andesite 52 – 57% Andesite 57 – 63% Dacite 63 – 68% Rhyodacite (quartz latite) 68 – 72% Rhyolite 72 – 75% High-silica rhyolite 75 – 77.5% IUGS has only two terms for SiO2 > 63 wt% (dacite and rhyolite) Many people who work on non-alkalic silicic rocks use a subdivision similar to what is at left Normative minerals Convert the chemical composition of a rock into hypothetical assemblage of water-free, standard minerals using a standard calculation scheme Normative composition, or CIPW norm Cross, Iddings, Pirsson, Washington Purpose Facilitate comparison of rocks using their chemical analyses Norms Hypothetical minerals based on simple molecular end members of complex solid solutions, e.g., Hy for hypersthene Di for diopside Q for quartz An for anorthite The minerals that would be present if there were no solid solutions Normative calculation emphasizes concentration of SiO2 relative to oxides of K, Na, Ca, Mg, and Fe You can use norms to classify rocks What solid solution series (minerals) contain the above normative minerals? Concept of degree of silica saturation SiO2 is most abundant component of igneous rocks Looking at system with only O, Si, Al, and Na Best and Christiansen, 2001, Fig. 2.14 Degree of silica saturation Silica-oversaturated (e.g., rhyolite, granite) Contain Q in norm Silica-saturated (e.g., andesite, diorite) Contain Hy but lack Q, Ne, or Ol Silica-undersaturated (e.g., phonolite, nepheline syenite) Contain Ol and possibly Ne These normative definitions have analogs in other types of classification schemes Concept of degree of alumina saturation Alumina (Al2O3) is the second most abundant component of igneous rocks Alumina saturation index Molecular ratio of Al2O3 / (K2O + Na2O + CaO) Ratio = 1 in feldspars and feldspathoids Any excess or deficiency must be accommodated in mafic or accessory minerals Important for classification of silicic rocks Alumina saturation Best and Christiansen, 2001, Fig. 2.15 Peraluminous rocks Alumina-oversaturated rocks Al2O3 / (K2O + Na2O + CaO) > 1 Excess alumina is accommodated in Micas (Al-rich biotite and muscovite) Other aluminous minerals, e.g., andalusite, cordierite, almandine-spessartine garnet, tourmaline After allocation of CaO for apatite, contain normative corundum, C Metaluminous rocks Alumina-undersaturated rocks that do not have excess alkalis Al2O3 / (K2O + Na2O + CaO) < 1 and Al2O3 / (K2O + Na2O) > 1 Deficiency in alumina is accommodated in minerals such as hornblende, Al-poor biotite, titanite After allocation of CaO for apatite, contain normative anorthite, An, and diopside, Di (or wollastonite, Wo) Peralkaline rocks Alumina-undersaturated rocks that have excess alkalis Al2O3 / (K2O + Na2O + CaO) < 1 and Al2O3 / (K2O + Na2O) < 1 Deficiency in alumina and excess in alkalis is accommodated in alkali mafic minerals such as aegirine, riebeckite, richterite, aenigmatite Mafic minerals with alkalis, Fe2O3, and TiO2 substitute for Al2O3 After allocation of CaO for apatite, contain normative acmite or sodium metasilicate, Ac or Ns, and lack normative anorthite, An Subdivision of peralkaline rocks Comenditic Al2O3 > 1.33*FeO + 4.4 Rhyolitic Comendite Trachytic Comenditic trachyte Pantelleritic (Fe-rich) Al2O3 < 1.33*FeO + 4.4 Le Maitre, 2002, Fig. 2.18; after Macdonald, 1974 Rhyolitic Pantellerite Trachytic Pantelleritic trachyte Rogers and Hawkesworth, 2000, Fig. 1 Alumina saturation Best and Christiansen, 2001, Fig. 2.15 Problems in practice Hard to analyze alkalis precisely Alkalis are easily removed by Hydration Vapor phase alteration Hydrothermal alteration TAS diagram Central portion divided into sodic and potassic suites sodic Le Maitre, 2002, Fig. 2.14 potassic TAS diagram Analyses of rocks from three suites of rocks Alkaline Tristan da Cunha oceanic island Subalkaline Andean continental arc Tongan island arc Best and Christiansen, 2001, Fig. 2.16 Suites of rocks Previous diagrams are used to classify individual rocks based on their chemical composition In contrast, the next few diagrams are used to To display compositions of suites To classify suites of rocks Harker variation diagram Plot of abundance of one chemical component against another Here, major elements shown for Crater Lake Danger: Plotting genetically unrelated rocks but interpreting them as if they were related Carmichael et al., 1974, Fig. 2-1 Harker variation diagram with trace elements Ta vs. Th of several large eruptive units P = Pantelleria DC = Tuff of Devine Canyon TT = Tala Tuff, Primavera LCT = Lava Creek Tuff, Yellowstone HRT = Huckleberry Ridge Tuff, Yellowstone BT = Bishop Tuff M = Mazama (Wineglass Tuff), Crater Lake K = Katmai (Valley of Ten Thousand Smokes) Hildreth, 1981, Fig. 2 AFM diagram F Presence or absence of iron enrichment trend Tholeiitic suite = Low-K suites (early iron enrichment) Calc-alkalic suite = High- and medium-K suites (no Fe enrichment) A M Best and Christiansen, 2001, Fig. 2.17 Alkali-lime index of Peacock (1931) Harker diagram, Na2O + K2O and CaO vs. SiO2 Vary inversely; crossing points <51% SiO2: Alkalic 51-56% SiO2: Alkali-calcic 56-61% SiO2: Calc-alkalic >61% SiO2: Calcic Should be used only for cogenetic rocks Hyndman, 1985, Fig. 43 TAS diagram Analyses of rocks from three suites of rocks Alkaline Tristan da Cunha oceanic island Subalkaline Andean continental arc Tongan island arc Would need to superimpose a plot with CaO for an alkalilime index Add to right axis Best and Christiansen, 2001, Fig. 2.16 Eruptive temperatures of prehistoric volcanic rocks: Mineral geothermometers Fe-Ti oxide geothermobarometer e.g., Ghiorso and Sack, 1991 Two pyroxenes e.g., Lindsley, 1983 Clinopyroxene e.g., Kretz, 1982 Olivine-ilmenite e.g., Andersen and Lindsley, 1981 Oxygen fugacity Concentration Adjusted concentrations to account for non-ideal behaviors Liquids: activity Gases: fugacity Fugacity: The partial pressure value needed to make a real (non-ideal) gas behave as an ideal gas Oxygen fugacity: A measure of the abundance of oxygen (adjusted “concentration”) May be present in exceptionally low concentrations, e.g., 10-20 The species O2 may not even be present, although reactions can be written as if it were Fe-Ti oxide geothermometer / oxygen barometer Uses compositions of two coexisting Fe-Ti oxide phases Cubic (spinel) phase: Magnetite-ulvöspinel solid solution Rhombohedral phase: Ilmenite-hematite solid solution Method is insensitive to total pressure Carmichael et al., 1974, Fig. 3-5, after Buddington and Lindsley, 1964 Applying Fe-Ti oxide geothermometer / oxygen barometer Highly useful, though many complexities (e.g., Ghiorso and Sack, 1991) Application requires Experimental data Means of extrapolation to experimentally inaccessible conditions (thermodynamic treatment) Accounting for “impurities” in the two phases No agreement yet on uniform method Oxygen fugacity and buffers Oxygen fugacity of geologically pertinent buffer assemblages varies with temperature Absolute value of f(O2), therefore, is not meaningful without corresponding T Buffer assemblages HM (MH): hematitemagnetite NNO: nickel-nickel oxide (bunsenite) FMQ (QFM): quartzfayalite-magnetite MW: magnetite-wüstite IW: Iron-wüstite Hildreth, 1981, Fig. 3 Oxidation state Oxidation state (relative oxygen fugacity): Position referenced to buffer assemblages at the same temperature Example: ΔNNO Hildreth, 1981, Fig. 3 Oxidation state Higher oxidation state—between NNO and MH Lamprophyric lavas Hornblende andesite lavas Basaltic andesites Lower oxidation state—between FMQ and IW Oceanic basalts Kilauea Reduced Oxidized Carmichael and Ghiorso, 1990, Fig. 2 Natural assemblages from intermediate to silicic volcanic rocks Assemblages with quartz, oxides, and ferromagnesian silicates Highest oxidation state Sphene (titanite)-quartz-clinopyroxene Hornblende-quartz-clinopyroxene Biotite-feldspar Orthopyroxene-quartz Olivine-quartz Lowest oxidation state Why might we care about the temperature and oxidation state? Why might we care? Probably telling us something about the source(s) of the magmas and processes that led to their generation Compositions and crustal history of the source(s) Alkaline vs. subalkaline basalts Assimilation of hematite-bearing redbeds and hydrothermally altered rocks vs. organic-rich or graphite-bearing shales and schists Tectonic setting of magmas Phenocryst species (and abundance) and oxidation state Note presence of fayalite (Fa), orthopyroxene (Op), ilmenite (I), magnetite (M), sphene/titanite (Sp) Also anorthoclase (Anor), chevkinite (Ch), aenigmatite (Aen), pyrrhotite (Po) Hildreth, 1981, Table 2 Volcanic rocks as samples of magma chambers Capturing products of a magma chamber at an instant in time Some characteristics probably are “quenched” in the bulk rock by the eruption Phenocryst content Others characteristics probably not preserved in the bulk rock Escape of volatiles from magma For these, need to look at compositions of fluid inclusions, melt inclusions, S content of apatite, etc. Chamber geometry versus eruptive sequence Inverted stratigraphy Material that is first out of vent winds up at stratigraphic base Material that is last out of vent winds up at stratigraphic top Initial assumption Single eruptive vent (or simultaneous vents tapping chamber at same level) “Bath tub like drawdown” of magma chamber Lots of complications imaginable Cashman et al., 2000, Fig. 1 Zonation of chamber (thermal) Total phenocryst abundance vs. SiO2 content for various eruptive units Late (bottom) SiO2 correlates with eruptive temperature Decreasing SiO2 corresponds to increasing temperature Note maximum phenocryst abundances Recall discussions of viscosity Early (top) Temperature Wohletz and Heiken, 1992, Fig. 1.7, adapted from Hildreth, 1981, Fig. 6 Zonation of chamber (chemical) Chemical zonation, Early/Late, for Bishop Tuff, Long Valley caldera, CA Compared to average granitic rocks Hildreth, 1981, Fig. 10 Fragmentation during eruption Cashman et al., 2000, Fig. 1 Physical sorting in pyroclastic eruptions During eruption, rock is fragmented “Pyro” + “clastic” Finest clasts/least dense materials most likely to go high in eruptive cloud and wind up in ash fall and be carried farthest away Fine glass shards from inflated melt Coarsest clasts/most dense materials most likely to wind up in pyroclastic flow Accidental lithic fragments, crystals (phenocrysts) Likelihood of fractionation of components based on size, density, etc. No longer in same proportions as in magma When sampling pyroclastic rocks Avoid sampling the whole rock--fractionated clasts If possible, sample pumice (but still have issue of volatiles!) Types and degrees of zonation Essentially unzoned Tapping chamber of uniform composition “Monotonous intermediates” Normal zonation Most silicic, least dense material generally at bottom of stratigraphy/top of magma chamber Most mafic, most dense material generally at top of stratigraphy/deeper in magma chamber Reverse zonation Opposite of normal Enrichment factors—A measure of the degree of zonation in the magma chamber Ratio of abundances of elements in two rocks Typically rocks from the same eruption, inferred to be from top of magma chamber and bottom of magma chamber Commonly ratio of most silicic rocks to least silicic rocks In simple case, corresponds to earliest rocks to latest rocks (Early/Late) Early/Late = Shallow/Deep (assumption) Different elements behave differently in one eruption Some enriched, others depleted toward roof Same elements may behave differently in different volcanoes Largely a function of different magma compositions Enrichment factors Bishop Tuff, Long Valley, CA Many others, too Ratio of abundances of elements in two rocks (especially trace elements) Typically rocks from the same eruption, inferred to be from top of magma chamber and bottom of magma chamber Wohletz and Heiken, Fig. 1.6, after Hildreth, 1981, Fig. 7 Hildreth, 1981, Fig. 10 Chemical analyses, Early/Late Hildreth, 1981, Table 1 Enrichment factors Enrichment factor diagram for Bishop Tuff, Long Valley caldera, CA: Example of strong zonation Enriched upward toward roof of magma chamber (No enrichment / depletion) Depleted upward (enriched downward toward floor of magma chamber) Wohletz and Heiken, Fig. 1.6, after Hildreth, 1981, Fig. 7 Plot of Ta vs. Th of several large eruptive units P = Pantelleria DC = Tuff of Devine Canyon TT = Tala Tuff, Primavera LCT = Lava Creek Tuff, Yellowstone HRT = Huckleberry Ridge Tuff, Yellowstone BT = Bishop Tuff M = Mazama (Wineglass Tuff), Crater Lake K = Katmai (Valley of Ten Thousand Smokes) Hildreth, 1981, Fig. 2 Laacher See Schmincke, 2004, Fig. 3.18 Laacher See Schmincke, 2004, Fig. 3.17 Compositional gaps Compositional gaps Inverted stratigraphy but with discontinuities in composition, e.g., zoned rhyolite, but skip to dacite In spite of thermal continuity indicated by geothermometry e.g., Katmai/Novarupta, AK, Valley of Ten Thousands Smokes Tuff Silica content Ultramafic IUGS divisions commonly followed for ultramafic to andesite No agreement on terms for silicic rocks <45 wt% SiO2 Basalt 45 – 52% Basaltic andesite 52 – 57% Andesite 57 – 63% Dacite 63 – 68% Rhyodacite (quartz latite) 68 – 72% Rhyolite 72 – 75% High-silica rhyolite 75 – 77.5% IUGS has only two terms for SiO2 > 63 wt% (dacite and rhyolite) Many people who work on non-alkalic silicic rocks use a subdivision similar to what is at left Ultramafic <45 wt% SiO2 Basalt 45 – 52% Basaltic andesite 52 – 57% Andesite 57 – 63% Dacite 63 – 68% Rhyodacite 68 – 72% Rhyolite 72 – 75% High- SiO2 rhyolite 75 – 77.5% Hildreth, 1981, Fig. 1 Eruptive types vs. SiO2 content Gaps Note Ranges of SiO2 contents Compositional gaps Gap Gaps Gap Hildreth, 1981, Fig. 1 Evolution of silicic magma chambers as a function of tectonic environment Time Basalt-rhyolite magmatism under crustal extension Bimodal magmatism Bimodal magmatism in areas of continental extension Early stage Wohletz and Heiken, 1992, Fig. 1.10, adapted from Hildreth, 1981, Fig. 15 Advanced stage Evolution of silicic magma chambers as a function of tectonic environment Time Tectonic extension, if any, is subordinate and shallow Abundant intermediate magmatism Island arcs, continental arcs, continental interior systems Wohletz and Heiken, 1992, Fig. 1.10, adapted from Hildreth, 1981, Fig. 15 Early stage Intermediate stage Mount St. Helens Mazama prior to formation of Crater Lake caldera Evolution of silicic magma chambers as a function of tectonic environment Evolutionary trend with time Time Hildreth, 1981, Fig. 15 Early stage Intermediate stage Hildreth, 1981, Fig. 12 Late stage Dynamic model of large, mature Cordilleran magmatic system Bishop Tuff, Long Valley caldera, CA Yellowstone, WY Can be reestablished over time Different volcanic centers in same volcanic field Multiple eruptions in same caldera complex, with repose times correlated directly to volumes of eruptions Hildreth, 1981, Fig. 12 Magma supply vs. percolation rate model Hildreth, 1981, Fig. 16 Ia Ib Ib IIb IIa IIa IIa IIa IIa IIa IIa Hildreth, 1981, Fig. 1 Magma supply vs. percolation rate model Note that “magma supply” to the lithosphere and eruption rate may differ by 1 to 3 orders of magnitude * = rapid transient injection of basalt at restricted crustal levels, resulting in generation and separation of rhyolite but few intermediates VIII VI IV V VII Ib Ia IIa IIb Hildreth, 1981, Fig. 16 III Multi-dimensional continuum of magma compositions Earth’s petrologic universe We will be exploring it the rest of the semester! Somewhat arbitrary subdivisions Given multiplicity of factors, might not expect there to be a perfect correlation of magma composition to tectonic setting Summary Chemical characterization of individual volcanic rocks Rock names (norms, TAS diagrams) Silica-saturation and alumina-saturation indices Chemical characterization of rock suites Variation diagrams Tholeiitic, calc-alkalic, shoshonitic suites Low-K, medium- and high-K, potassic suites Volcanic rocks represent nearly instantaneous samples of magma chambers (prior to their late crystallization histories) Beware of physical sorting during pyroclastic eruptions because of size and density differences (sample pumice, not whole rock) Types and degree of zonation measured by enrichment factors (magma chamber and inverted stratigraphy) Compositional gaps are common Next time: Silicic lava domes and flows